
Education Journalist | Study Abroad Strategy Lead
Phosphorus, an important element, was discovered in the 17th century and played a crucial role in the development of the concept of "element" in Chemistry.
- The initial discovery of phosphorus was in its most unstable form called white phosphorus.
- Since then, we have discovered that phosphorus can exist in different forms known as allotropes.
- Allotropy refers to the ability of certain chemical elements to exist in different forms while maintaining their similar physical properties.
- These different forms of the element are called allotropes.
- Phosphorus has multiple allotropes, and scientists have used quantum-chemical techniques to study and evaluate over 50 different crystalline structures of phosphorus allotropes.
These studies have contributed to our understanding of the stability and properties of different phosphorus allotropes, making phosphorus one of the most extensively studied elements on the periodic table.
Read More: Difference Between Allotropes and Isomers
| Table of Content |
Key Terms: Allotropes, Crystalline, Element, Furnace, Phosphorus, White Phosphorus, Black Phosphorus, Red Phosphorus
What is an Allotrope?
[Click Here for Sample Questions]
Allotropy is a fancy word for when an element can exist in different forms or shapes.
- These different forms of the same element are called allotropes.
- Many elements can have different allotropes, and each allotrope has its unique physical properties.
- However, their chemical properties are similar.
Read More: Law of multiple proportions
Allotropes of Phosphorus
[Click Here for Previous Year Questions]
The following are a few examples of phosphorus allotropes:
White Phosphorus
White phosphorus is a type of phosphorus that looks like wax and is see-through.
- It is fragile and requires careful handling.
- It doesn't dissolve in water but can dissolve in substances like carbon disulphide or carbon tetrachloride.
- When heated with a strong alkaline solution called caustic soda in the presence of air, it breaks down and forms sodium hypophosphite and phosphine.
Synthesis and Structure of White Phosphorous
In simple terms, phosphate rock is heated in an electric furnace along with carbon and silica.
- This process releases phosphorus as a vapor, which can be extracted using phosphoric acid.
- White phosphorus, which has a circular shape, is commonly used in weapons.
- The white phosphorus molecules are made up of three phosphorus atoms bonded together at a 60-degree angle.
- These molecules are attracted to each other by weak forces called Van Der Waals forces.


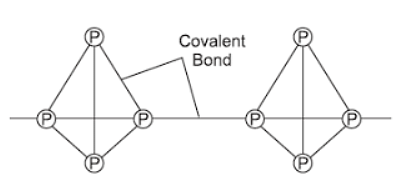
White Phosphorous
In the picture above, you can see the shape of white phosphorus.
- It looks like a pyramid with four sides.
- Each phosphorus particle is connected to three other phosphorus particles through strong bonds.
- Additionally, there are weak forces that hold these particles together.
- It's important to know that white phosphorus is highly reactive and dangerous.
Physical and Chemical Characteristics of White Phosphorus
White phosphorus, in its solid form, looks like wax and is see-through. It is very delicate and needs to be handled with care. It doesn't dissolve in water, but it can break down when in contact with certain substances.
- The solid form of white phosphorus is soft.
- When it is first made, it is almost colorless, but it gradually turns light yellow. This is why it is sometimes called yellow phosphorus.
- White phosphorus has a low melting point, so it can easily catch fire when exposed to air at temperatures slightly higher than room temperature.
- To prevent this, it is usually stored in water.
- When it burns, it produces phosphorus pentoxide.
- When white phosphorus comes into contact with moist air, it undergoes an oxidation process.
- This reaction creates a bright and sparkling light, making it glow without much effort.
- The molecular weight of white phosphorus is 30.97 g/mol & the melting point is 44°C.
- It is dangerous and very reactive.
Red Phosphorus
Red phosphorus is a great material to prevent fires, especially in things like plastic that can melt easily when they get hot.
- When red phosphorus combines with certain chemicals, it creates a special acid that helps stop flames.
- This acid, along with other materials, forms a protective layer that keeps the fire from spreading further.
Synthesis and Structure of Red Phosphorus
When white phosphorus is heated to a specific temperature in a special environment for a long time, it changes into red phosphorus.
- Red phosphorus is made up of many connected units and has a different structure compared to white phosphorus.
- It is less reactive than white phosphorus because of its unique structure.
Red Phosphorus
Physical and Chemical Characteristics of Red Phosphorus
Here are few properties of Red Phosphorus:
- Red phosphorus looks like a shiny, crystal-like substance and has a color similar to iron grey.
- It doesn't cause harm and doesn't have a bad smell. It doesn't break down when exposed to carbon tetrachloride or water.
- When red phosphorus is heated, it doesn't turn into boiling white phosphorus, which is a strong chemical.
- Instead, it breaks down when in contact with alcoholic potash.
- Normally, red phosphorus is stable and doesn't catch fire in the air. However, if we heat it to around 400°C, it will burn.
- When red phosphorus combines with oxygen at a temperature of 565 K, it forms phosphorus pentoxide.
- If red phosphorus combines with sulphur, it creates a substance called sulphides.
Black Phosphorus
There are two types of black phosphorus called α-black and β-black. When we heat red phosphorus in a closed tube at a temperature of 803K, it changes into the α-black form. This α-black form can turn into gas and disappear in the air.
- It has either rhombohedral crystals or an opaque monoclinic shape.
- It doesn't get affected by oxygen in the air.
- On the other hand, when we heat the white form of phosphorus under high pressure at a temperature of 473K, it turns into the β-black form.
- This β-black form doesn't catch fire in the air until it reaches a temperature of 673K.
- The structure of the black form of this substance is shown below.

Black Phosphorous
Physical and Chemical Properties of Black Phosphorus
Here are few properties of Black Phosphorus:
- Black phosphorus has a 416 degree celsius melting point.
- The black phosphorus has a specific gravity of 2.69.
- Black phosphorus has a precise mass of 30.973762 g/mol.
- Black phosphorus has a density of 2.34 g/cm3.
- Black phosphorus dissolves 0.3 g/l in water (H2O).
- Black phosphorus has a molecular mass of 30.97.
- There are crystalline and amorphous forms of black phosphorus.
Read More: Potassium Hypochlorite Formula
Difference Between Black Phosphorus, White Phosphorus, and Red Phosphorus
[Click Here for Sample Questions]
The basic differences are mentioned in the table below.
| Basis | Black Phosphorus | White Phosphorus | Red Phosphorus |
|---|---|---|---|
| Color | Black | yellowish | Red |
| Stability | Most stable among all | Least stable among all | Moderate stable among all |
| Reactivity | Least Reactive among all | Most Reactive among all | Moderate Reactive among all |
Also Read:
| Related Articles | ||
|---|---|---|
| Atomic mass of elements | Mole concept | Mendeleev's Periodic Table |
Things to Remember
- White phosphorus exhibits chemiluminescence, but red phosphorus does not.
- Phosphates, which are substances with the phosphate symbol (PO43-), are the main sources of phosphorus.
- Phosphates are found in ATP, DNA, RNA, and phospholipids, all of which are critical components of living things.
- Both bone ash and human urine were used as early phosphate symbol sources.
- The oldest known source of the fundamental phosphate symbol was human urine.
- The white form of phosphorus is heated to 473 K under great pressure, where it turns into black phosphorus.
Previous Year Questions
- Extraction of metal from the ore cassiterite involves...[JEE Advanced 2011]
- Commonly used vectors for human genome sequencing are...[NEET UG 2014]
- Interfascicular cambium and cork cambium are formed due to..
- Pneumotaxic centre is present in...[UP CPMT 2007]
- Reaction of HBr with propene in the presence of peroxide gives….[NEET UG 2004]
- Assuming the expression for the pressure exerted by the gas on the walls of the container, it can be shown that pressure is...[MHT CET 2016]
- Which among the following is the strongest acid?...[TS EAMCET 2017]
- Isopropyl alcohol on oxidation forms..
- A vector is not changed if..
- Which of the following arrangements does not represent the correct order of the property stated against it?...[JEE Main 2013]
Sample Questions
Ques: What are allotropes? (2 Marks)
Ans: Allotropes are different forms of the same element that exhibit distinct physical and chemical properties. In the case of phosphorus, it exists in several allotropes with varying structures and properties.
Ques: How many allotropes of phosphorus are there? (2 Marks)
Ans: Phosphorus has several known allotropes, but the most common ones are white phosphorus, red phosphorus, black phosphorus, and violet phosphorus.
Ques: What is white phosphorus? (2 Marks)
Ans: White phosphorus is the most reactive and unstable allotrope of phosphorus. It is a translucent, waxy solid that emits a faint glow in the dark. It is highly flammable and reacts vigorously with air and water.
Ques: What is red phosphorus? (2 Marks)
Ans: Red phosphorus is a more stable allotrope of phosphorus. It is a dark red powder that is less reactive than white phosphorus. It is not flammable and does not ignite spontaneously like white phosphorus.
Ques: What is black phosphorus? (2 Marks)
Ans: Black phosphorus is another allotrope of phosphorus that exists in multiple forms, including α-black and β-black. It has a layered structure and appears as a dark-colored solid. It is less reactive than white phosphorus but more reactive than red phosphorus.
Ques: What are the properties of black phosphorus? (2 Marks)
Ans: Black phosphorus is a semiconductor material with unique electronic properties. It has high anisotropy, meaning its properties vary depending on the direction in which it is measured. It can exhibit different crystalline structures and has potential applications in electronics and optoelectronics.
Ques: Is black phosphorus stable? (2 Marks)
Ans: Black phosphorus is relatively stable compared to white phosphorus. However, it can undergo degradation over time, particularly in the presence of oxygen and moisture. Care must be taken to handle and store black phosphorus properly to maintain its stability.
Ques: What is violet phosphorus? (2 Marks)
Ans: Violet phosphorus is a recently discovered allotrope of phosphorus. It has a unique crystal structure and appears as a dark violet or purple solid. It is even less reactive than black phosphorus and has potential applications in electronic devices.
Ques: Are all allotropes of phosphorus dangerous? (2 Marks)
Ans: White phosphorus is considered highly toxic and flammable, requiring careful handling. However, red, black, and violet phosphorus are relatively stable and less hazardous. It is still important to follow safety precautions when working with any form of phosphorus.
Ques: What are the applications of phosphorus allotropes? (2 Marks)
Ans: The different allotropes of phosphorus find various applications. White phosphorus has been used in matches, munitions, and chemical synthesis. Red phosphorus is commonly used as a flame retardant and in the production of safety matches. Black phosphorus has potential uses in electronic devices, while violet phosphorus is still being explored for its unique properties.
For Latest Updates on Upcoming Board Exams, Click Here: https://t.me/class_10_12_board_updates
Check-Out:






Comments