
Content Curator
Aluminum oxide is a compound made up of aluminum and oxygen atoms. The formula of the compound is Al2O3. Aluminum oxide filaments finer than a human hair may be made, making it suitable for DNA filtering. Aluminum oxide is extensively used in the mining and ceramic manufacturing industries.
| Table of Content |
Key Terms: Aluminium Oxide, Alumina, Bauxite, Electrolysis, Corundu, Aluminium, Ore, Minerals
What Is Aluminium Oxide?
[Click Here for Sample Questions]
When aluminum and oxygen combine, they form a compound called aluminum oxide. The most common form of aluminum oxide is aluminum (III) oxide. Corundum, the naturally occurring form of aluminum oxide, is generally in the crystalline polymorphic phase – Al2O3.
This aluminum oxide is found in many precious gemstones like ruby and sapphire. Due to its hardness and high melting point, Al2O3 is used to produce aluminum metal as an abrasive and refractory material. Aluminum oxide has a molar mass of roughly 101.96 g/mol.
Aluminum oxide is used in various lights, including sodium-vapour lamps, and as an electrical conductor in small circuits in the burgeoning nanotechnology sector.

Aluminum Oxide
Structure Of Aluminium Oxide
[Click Here for Previous year's Questions]
The structure of aluminum oxide are:
- Corundum is generally thermodynamically stable.
- The trivalent ions of the aluminum present in the compound are octahedral.
- The oxygen ions present in the compound are closely packed in hexagonal form with the aluminum ions.
Aluminum Oxide (Al2O3) has the following Lewis structure:
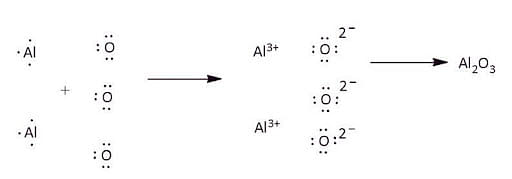
Structure of Aluminium Oxide (Al2O3)
Properties Of Aluminium Oxide
[Click Here for Sample Questions]
General Properties
Aluminum oxide is extraordinarily heavy; a 1-meter by 1-meter cube of aluminum oxide weighs approximately 7,200 lbs. Although it is non-toxic, airborne aluminum oxide dust can pose industrial dangers. Hence masks are suggested for extended exposure. Aluminum oxide is a white powdered material with no discernible odor.
Physical Properties
- Aluminum oxide has a density of 3.987g/cm3.
- It is insoluble in water and nearly all other solvents.
- Aluminum oxide appears as a white powder.
- Aluminum oxide has a relatively high melting point (2345K) and boiling point (3250K).
- While being an electrical insulator in ceramics, aluminum oxide possesses heat conductivity.
Also Check: Aluminium
Chemical Properties
In contrast to its relative aluminum, Al2O3 exhibits considerable electrical resistance. The degree of resistance varies with the purity of the substance. Despite being particularly reactive to chlorine trifluoride and ethylene oxide, most compounds do not react quickly with aluminum oxide.
A fire occurs when aluminum oxide is combined with one of these substances. Aluminum reacts with oxygen in the environment to form an aluminum oxide protective layer on its surface. This coating of aluminum oxide assists in the prevention of aluminum corrosion. Because of this, aluminum is referred to be a corrosion-resistant metal.
Industrial Properties
Aluminum oxide may be manufactured or cast into robust, wear-resistant composites for various engineering applications. Some examples of these applications may include metering devices, wire guides, machinery gaskets, and high-temperature electrical insulators.
Mechanical Properties
Aluminum oxide has a high wear resistance since it is an extremely hard material, nearly as hard as diamonds. Due to its high electrical resistance, aluminum oxide is often used as the dielectric in capacitors to keep charges separated. It possesses excellent corrosion resistance and temperature stability, low thermal expansion, and a good stiffness-to-weight ratio.
| Property | Value |
|---|---|
| Appearance | Solid |
| Density | 3.95 g/cm3 |
| Hardness | 15 – 19 GPa (9 on the Mohs scale) |
| Compressive strength | 2,000 – 4,000 MPa |
| Mechanical strength | 300 – 630 MPa |
| Boiling point | 2,977 °C (5,391 °F; 3,250 K) |
| Melting point | 2,072 °C (3,762 °F; 2,345 K) |
| Electrical resistivity | 1012 – 1013 Ωm |
| Thermal conductivity | 20 – 30 W/mK |
| Molecular mass | 101.96 g/mol |
Properties and their value
Preparation Of Aluminium Oxide
[Click Here for Previous year's Questions]
The following processes can be used for manufacturing aluminum oxide:
- Aluminum oxide is created when aluminum reacts with oxygen.
| 4Al + 3O2 → 2Al2O3 |
- Alumina (aluminum oxide) may be extracted from bauxite by leaching. Common impurities in the ores of aluminum oxide include silicon dioxide (SiO2), iron oxides, and titanium oxide (TiO2).
A NaOH solution is used to concentrate the bauxite ore at 473-523K and 35-36 bar pressure. As a consequence, sodium aluminate is formed (SiO2 is produced in solution as sodium silicate), leaving the impurities behind.
| Al2O3(s) + 2NaOH(aq) + 3H2O(I) → 2Na[Al(OH)4](aq) |
Carbon dioxide is pumped through the solution to neutralize the sodium aluminate. Hydrated aluminum oxide is formed as a result.
| 2Na[Al(OH)4](aq) + CO2(g) → Al2O3⋅xH2O(s) + 2NaHCO3(aq) |
The hydrated alumina is removed from the solution, keeping the sodium silicate aside. Pure Al2O3 is produced by drying and heating filtered hydrated alumina.
Chemical Reactions Of Aluminium Oxide
[Click Here for Sample Questions]
With Hydrochloric Acid
Aluminum oxide interacts with acids similarly to sodium and magnesium oxides because it contains oxide ions. When aluminum oxide reacts with hot dilute hydrochloric acid, an aluminum chloride solution is generated.
| Al2O3 + 6HCl → 2AlCl3 + 3H2O |
With Sulphuric Acid
Metal oxides are typically basic, although aluminum oxide is amphoteric. As a result, it functions as both an acid and a base. In this scenario, it serves as a foundation. This type of reaction is called a neutralization reaction.
| Al2O3 + H2SO4 → Al2(SO4)3 + H2O |
With Sodium Hydroxide
When aluminum oxide reacts with sodium hydroxide, sodium aluminate and water are generated. Temperatures ranging from 900 to 1100°C are involved in this process. When aluminum oxide works as an acid, it generates salt and water.
| Al2O3 + 2NaOH → 2NaAlO2 + H2O |
With Hydrofluoric Acid
When aluminum oxide reacts with hydrofluoric acid, aluminum fluoride salt and water are formed.
| Al2O3 + 6HF2 → AlF3 + 3H2O |
Uses Of Aluminium Oxide
[Click Here for Previous year's Questions]
- It can be present in birth control pills and hip replacement surgery.
- It is used in the manufacture of glass. Aluminum oxide is a frequent component of aluminosilicate glass. Aluminum oxide makes around 5-10% of its makeup.
- Aluminum oxide flakes are used in paints to add a reflective or ornamental appearance.
- In laboratories, it can be employed as a chromatographic medium.
- Because of its hardness and strength, aluminum oxide is commonly used as an abrasive. Aluminum oxide is used to make sandpaper, cutting tools, and other items in place of industrial diamonds.
- Transparent aluminum oxide is used in sodium vapour lamps and small fluorescent lights.
- Water may be removed from gas streams using aluminum oxide.
- It is used as a catalyst in several industrial processes.
- It is most frequently used as a ceramic material.
- It's prevalent in sunscreen, lipstick, blush, and nail polish.
- It's a type of plastic filler.
- Aluminum oxide is used in integrated circuits as an electrical insulator.
Also Check:
| Chapter Related Topics | ||
|---|---|---|
| Hydrochloric Acid | Sulphuric Acid | Sodium Hydroxide |
| Amphoteric | Polar Ionic Compound | Vsep |
Things To Remember
- Aluminum oxide is an amphoteric material that can act as an acid and a base.
- Al2O3 is a polar ionic compound.
- Sp2 hybridization takes place in Al2O3 molecules.
- Because Al2O3 has a VSEP number of 3, the molecule should adopt the shape of a Trigonal Planar.
- Aluminum is referred to be a corrosion-resistant metal.
Important PYQs Based On Aluminium Oxide
- The Enthalpy Of Sublimation Of Aluminium Is 330 KJ…. [BITSAT 2014]
- Aluminum Reacts With Caustic Soda To Form….. [MU OET 2005]
- Bombardment Of Aluminium By A particle Leads To Its… [JEE Advance 2011]
- An Aluminium Rod And A Copper Rod Are Taken Such…. [DUET 2007]
- 45 G Of Aluminium At Mass 27 Amu Is Deposited At… [NEET 2005]
- Aluminum Chloride In Acidified Aqueous Solution… [NEET 2019]
- Purification Of Aluminium By Electrolytic Refining…. [NEET 1999]
- In The Electrolytic Method Of Obtaining Aluminium… [KCET 2004]
- Aluminum Displaces Hydrogen From Dilute HCl Where…. [KCET 2004]
- An Aluminium Ring B Faces An Electromagnet A The…. [KCET 2001]
- Aluminium Chloride Is A…. [KCET 2000]
- Aluminium Atomic Mass 27 Crystallizes In A Cubic… [KEAM]
- During Extraction Of Aluminium From Bauxite…
- Aluminium Is Usually Found In 3 Oxidation Stage… [JEE Main 2019]
- In The HallHeroult Process Aluminium Is Formed At…. [JEE Main 2019]
- Lithium Aluminium Hydride Reacts With Silicon… [JEE Main 2018]
- In The Aluminothermite Process Aluminium Acts As… [JEE Advanced 1983]
- Electrolytic Reduction Of Alumina To Aluminium By…. [JEE Advanced 2000]
- When Zeolite Is Hydrated Sodium Aluminium…. [JEE Advanced 1990]
- Bauxite The Ore Of Aluminium Is Purified By Which…
Sample Questions
Ques. Find out the hybridization of Al2O3. (4 Marks)
Ans. The hybridization formula is provided as follows:
Number of sigma () bonds on central atom + lone pair on center atom = hybridization
Al2O3 hybridization determination:
The number of sigma bonds on the core atom is three.
The number of lone pairs on the central atom is equal to zero.
As a result, Hybridization = 3 + 0 = 3, indicating sp2 hybridization.
As a result, Al2O3 molecules exhibit sp2 hybridization.
Trigonal hybridization is another name for sp2 hybridization.
The hybrid orbital created by combining one s and two p-orbitals has trigonal symmetry and is kept at a 120° angle.
Ques. What is the hardness of aluminum oxide? (1 Mark)
Ans. The hardness of aluminum oxide is Alumina 1800, Unit HV.
Ques. What is the polarity of Al2O3? (3 Marks)
Ans. In the instance of Al2O3, it is produced by the passage of an electron from the aluminum atom to the oxygen atom.
Since the oxygen atom is much more electronegative, it draws the electron pair in the bonding, resulting in a negative charge.
In contrast, aluminum is electropositive and loses its valence electrons to oxygen, leading to a positive charge.
Al2O3 is, therefore, a polar ionic compound.
Ques. What is the basicity of aluminum oxide? (2 Marks)
Ans. According to the latest study, the basicity of aluminum oxide is less than 0.200 mmol g −1
Ques. Explain the uses of aluminum oxide in different industries. (5 Marks)
Ans. The following industries make use of aluminum oxide:
-Medicine Industry
Because of its hardness, bio-inertness, and chemical properties, aluminum oxide is an excellent material for bearing surfaces in hip replacements, such as prostheses, bionic implantation, artificial eye substitutes, tissue reinforcements, tooth crowns, girders, bridges, as well as other dental work. It's also found in laboratories' plants and machinery, including crucibles, burners, and other labware.
- Military and protective equipment
The hardness and lightness qualities of aluminum oxide aid in improving protective vests, such as suits of armour and car and aviation shields, which is its principal market. Aluminum oxide can also be used in ballistics and an unbreakable glass of synthesized sapphire.
- Manufacturing of electrical and electronic components
Because of its extreme melting and boiling temperatures and remarkable heat capacity, aluminium oxide is utilized in the manufacture of high-temperature furnace insulating layers and surface coatings. Alumina films are also critical elements in semiconductor devices.
- Gemstone industry
Aluminum oxide is a key ingredient in the creation of rubies and sapphires. In its crystalline form, Corundum serves as the basis for these exquisite gems. Chromium impurities cause the deep red color of rubies, but the various colors of sapphires are caused by iron and titanium residues.
Ques. Is aluminum oxide a neutral compound? (3 Marks)
Ans. The overall charge of a compound should be equal to zero which is neutral. In Al2O3, we require 2 aluminum atoms and 3 oxygen atoms to balance the charge and to make the compound neutral. This means the chemical formula for aluminum oxide is Al2O3.
Ques. Why is aluminum oxide amphoteric by nature? (3 Marks)
Ans. Aluminum oxides are amphoteric and may function as either an acid or a base in an acid-base reaction. Amphoteric compounds are those that can react with both acids and bases. For example, aluminum oxide reacts with hydrochloric acid to form water and aluminum chloride. In this reaction, the base is aluminum oxide. When aluminum oxide reacts with basic sodium hydroxide (NaAlO3), water and sodium aluminate can be produced. Aluminum oxide acts as the acid in this process.
Ques. How is aluminum oxide produced from aluminum? (3 Marks)
Ans. Aluminum metal interacts with atmospheric oxygen during air exposure to generate aluminum oxide. As a result, aluminum is passivated against oxidation once the aluminum oxide layer forms and aluminum is completely passivated against oxidation.
First, prepare a very fine aluminum powder in an inert environment before gradually introducing oxygen to obtain a high yield of aluminum oxide. It would also work to expose the powder to air, but this would be exceedingly hazardous. You could dissolve it in acid and oxidize it in solution, but you risk sparking a fire. And it will be burning on top of a strong acid solution.
Ques. In what way does cryolite lower the melting point of aluminum oxide? (3 Marks)
Ans. Because aluminum oxide has an exceptionally high melting point (about 2,000°C), melting it would be costly. It is instead dissolved in molten cryolite, an aluminum compound with a lower melting point than aluminum oxide. The use of cryolite minimizes some of the energy expenses associated with aluminum extraction. On the other hand, the cryolite dissolves the aluminum oxide rather than lowering its melting point. When aluminum oxide ions are liberated from molten cryolite, they can be electrolyzed (melting point at 1273K).
Ques. Is aluminum oxide acidic basic or amphoteric? (2 Marks)
Ans. Aluminum oxide is an amphoteric substance.
Ques. Why is it necessary to melt aluminum oxide during electrolysis? (2 Marks)
Ans. Ions must be free to migrate in electrolysis to reach the proper electrode (in the case of metal ions, the cathode). Ions in liquids may flow freely but not in solids. They would be free to move if the aluminum oxide was dissolved in an aqueous solution (through acid addition); but, because hydrogen ion is quicker to reduce than aluminum ion, the electrolysis would create hydrogen gas rather than aluminum metal.
Ques. Why can't aluminum oxide conduct electricity as a solid? (2 Marks)
Ans. Aluminum oxide cannot conduct electricity as a solid because the electrons are securely bound to the atoms in the combination and are not free to move. Aluminum is a highly reactive metal that readily reacts with oxygen to form aluminum oxide.
This is a very stable chemical in which the electron orbitals of the atoms are filled with shared electrons. It's relatively inert since the electrons are in very stable structures and don't easily come free to conduct electricity. Electrons in aluminum metal, like those in other basic metals, are much more loosely bonded and may be easily moved by an electric charge.
Ques. How can aluminum oxide get reduced? (1 Mark)
Ans. Using electrolysis, aluminum oxide is converted to aluminum metal. In an electrolytic cell, bauxite ore is heated to melting with particular flux chemicals, sometimes aluminum fluoride. A large current is sent across the melt, producing aluminum metal on carbon electrodes. This is how almost all aluminum is created.
Ques. Is Al2O3 harmful to human health? (2 Marks)
Ans. Aluminum oxide is an insoluble aluminum compound that does not generate a dangerous reaction right away. Although continuous exposure to aluminum oxide is almost non-existent in today's company, it can cause health discomfort and more serious health concerns. Apart from skin irritation, one element that makes aluminum oxide a feasible and popular choice in the manufacturing of contemporary ceramics is its lack of acute toxicity and overall low chronic toxicity.
Also Read:






Comments