
Content Curator
Isomers of Butane are of two types, namely isobutane (2-methylpropane) and n-butane. In Chemistry, isomers are those compounds that have different chemical structures and the same molecular formula.
- Isomers have different chemical and physical properties. This is because of their distinct structures.
- Butane is considered an alkane.
- The chemical formula of Butane is C4H10.
- It consists of 4 carbon atoms and 10 Hydrogen atoms together.
- The structure of butane is in the form of a long straight chain.
Read More: Organic Compounds
| Table of Content |
Key Terms: Isomers, Atoms, Butane, Alkane, Ethane, Methane, Chain Isomerism, stereoisomerism
What are Isomers?
[Click Here for Sample Questions]
'Isomers' is derived from the Greek words i.e. 'Isos' and 'meros'. It means 'equal parts'. The word “isomer” is derived from the Greek words “isos” and “meros”, which mean “equal parts”.
- Isomers are chemical compounds which share the same chemical formula but vary in their chemical structures.
- Isomers have various chemical properties. So, it can be said that isomers are those compounds which exhibit the phenomenon of isomerism.
- In this phenomenon of isomerism, the structure varies due to the arrangements of atoms in the molecule.
Also Read:
| Concept-Related Articles | ||
|---|---|---|
| Isomerism in Coordination Compounds | Optical isomerism | Finkelstein reaction |
| Cis-Trans Isomerism | Alliphatic Hydrocarbons | Saturated solution |
Types of Isomers
[Click Here for Previous Year Questions]
On the basis of the structure of atoms, it is classified into various types. The chemical compounds like methane, butane, ethane, etc are structured in different ways.
Structural Isomerism
In this type of isomerism, the compounds are distributed in different directions and make varying structure. For example, Isobutane and n-butane.
Chain Isomerism
In this type of Isomerism, the compounds have different branches of Carbon atoms but the molecular formula is the same for it. For example - 2 methyl butane (Isopentane)

Isopentane
Functional Isomerism
In this type of Isomerism, the compounds have the same molecular formula but vary in their functional group. For example - Ethanol. Ethanol contains 'OH' as its functional group and the molecular formula is C2H6O.
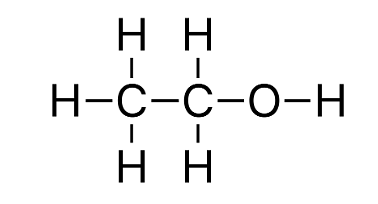
Ethanol
Stereoisomerism
In this type of isomerism , the compounds have molecular formulas with different spatial orders of atoms. This is known as stereoisomerism. For example: 1,2 - dichloroethene. The chemical formula is (C2H2Cl2). There are two types of stereoisomerism namely Optical isomerism and Geometric isomerism.

1,2 - dichloroethene
Constitutional Iomers of Butane
In this type of isomerism, the chemical compounds have the same molecular formula with different structural formula.
- The atoms in molecules are connected in a different form.
- In order to determine the constituency of the atoms, the number of each atom is counted in both molecules.
This can be understood with an example.
- In Isobutane, the carbon atoms are arranged with a side chain because the molecules have different connectivity of atoms.
- Whereas in case of N-butane, the connectivity of carbon atoms differ. They are arranged in the form of a straight chain.
- But both these isomers have same molecular formula i.e. C4H10.
Conformational Isomers of Butane
This type of conformational isomerism can be seen in alkanes. This is because of the presence of C-C bonds. This can be understood with an example based on the rotation around the sigma bond.
- The stereoisomers change their form by rotation.
- When the molecules of butane are rotated at the axis of Carbon bond, then the Conformational Isomers of butane is obtained.
- In this type of isomerism, the identical groups are seen that makes the bond unstable.
- The rotation leads to the spatial arrangements of the hydrogen atoms with Carbon.
- This is known as conformational isomers of butane.
- Example: Butane, there are three conformers attached with the two methyl (CH3) groups.
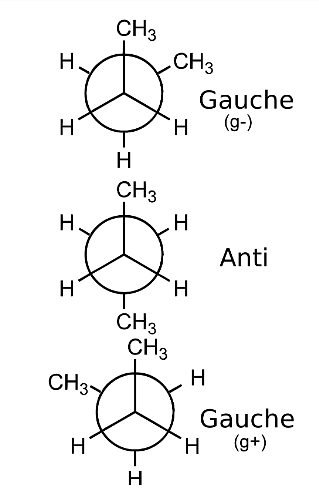
Conformational Isomers of Butane
Staggered Conformation of Butane
A conformer which has a dihedral angle of 60 degrees, wherein the H-atoms of C-1 are close enough to the H-atoms of C-2 are known as staggered conformation.
- The staggered conformer is known to be more stable than the eclipsed conformer.
- This is due to the H-atoms which are separated by an adequate distance.
- Butane staggered conformers are of two types: anti and gauche conformers.
- When one carbon atom in butane is fixed, while spinning the other, three types of conformers can be obtained: eclipsed, anti, and gauche.
- In anti-form, both methyl groups of butane can be found at anti position. It is considered to be one of the most stable butane forms. The other type is known as gauche or skew conformation.
Isomers of Butane
[Click Here for Sample Questions]
Isomers are composed of more than two molecules. That's why their structure as well as their properties are different. The isomers of Butane are: Isobutane (2-methylpropane) and n-butane.
Thus, some of the Properties of Isomers of Butane are:
- Those isomers who form branched- chain have low melting and boiling points. Its example is isobutane.
- The melting point of isobutane is -160° C and boiling point is -12° C
- The properties of N - butane are different. The boiling point is 0 °C and melting point is -138 °C. The lower the boiling and melting points.Thus,it shows if there is low melting and boiling point of the compound, the more branched chain can be formed.
- Butane is highly inflammable in nature. So, it can be used for heating purposes easily.
Structural Isomers of Butane
[Click Here for Previous Year Questions]
Butane is a chemical compound which consists of 4 carbon alkane and 10 atoms of hydrogen.
- The chemical formula of Butane is C4H10. It constitutes two different types of structures.
- It simply means, the structures are straight as well as branched chain forms.
- The main aim of the structure is to complete the valency of the carbon atom in order to form the four bonds.
Butane consists of two structural/constitutional isomers. They are N-butane and Isobutane. According to the IUPAC name, they are known as butane and 2 - methylpropane.
The structure of N-butane is present in the form of a straight chain because of the arrangement of all Carbon atoms. Whereas isobutane has the side chain.
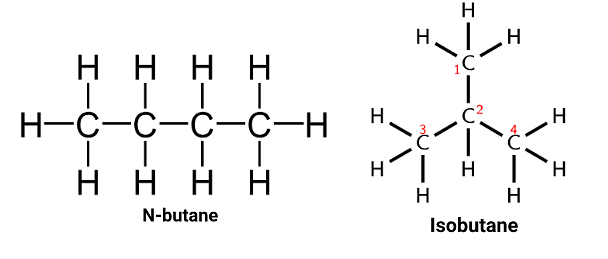
Structural Isomers of Butane
Things to Remember
- Alkanes are chemical compounds that exhibit the phenomenon of isomerism.
Example - Methane (CH4), ethane (C2H6), propane (C3H8) and butane (C4H10).
- Isomers are those compounds that have the same molecular formula but their chemical structure is different.
- Butane is an example of alkane and the chemical formula of Butane is C4H10.
- There are two isomers of Butane and they are known as N-butane and Isobutane. Their structures are formed in a ring chain and in a straight chain.
- There are two types of isomerism. They are known as structural or constitutional isomerism and stereoisomerism/spatial isomerism.
Previous Year Questions
- Which of the following releases maximum energy on combustion….[AMUEEE 2011]
- In the following reaction, major product obtained is [AMUEEE 2010]
- Which of the following is aromatic compounds? [AMUEEE 2016]
- Which one of the following mono-enes does not exhibit geometric isomerism? [JKCET 2008]
- For an electrophilic aromatic substitution reaction [JKCET 2012]
- 2-Hexyne gives trans-2-hexene on treatment with [JEE Advanced 2004]
- 2-phenyl propene on acidic hydration, gives [JEE Advanced 2004]
- Ethylene can be converted into alcohol by treatment of [JKCET 2014]
- Which one of the following is not aromatic? [JKCET 2011]
Sample Questions
Ques. What type of bonds are present in butane? (1 mark)
Ans. Butane is an alkane having 4 carbon atoms consisting of covalent bonds connecting the molecules. There are around 13 single covalent bonds present in Butane.
Ques. Is methylpropane known to be the isomer of butane? (1 mark)
Ans. Yes, as per the isomerism properties of butane, methylpropane is known to be the isomer of butane.
Ques. Why does butane not possess the geometric type of isomers? (1 mark)
Ans. Due to the single bonds between the carbons atoms in butane, it fails to form geometric isomers.
Ques. Why type of isomerism properties do butane possess? (1 mark)
Ans. Butane is said to possess chain or nuclear isomerism in it. It is highly inflammable by nature. The boiling and melting point is different because of their structure.
Ques. From which source Butane can be obtained? (1 mark)
Ans. We get Butane from different processes that take place in the refinery. It can be achieved from some environmental sources like natural gas plants and steam crackers.
Ques. Define conformational isomers. (1 mark)
Ans. Stereoisomers that allow to be converted into one another by rotation around a single bond are known as conformational isomers
Ques. What is isomerism? (2 marks)
Ans. Isomerism is the phenomenon in which the chemical compounds possess the same chemical formulas but have different structural atom arrangements. Isomers that fall under the category of organic substances. Butane is a chemical compound having two isomers. They are isobutane and n-butane.
Ques. State the types of isomerism. (2 marks)
Ans. Based on their structure, they are classified into two different types and further they are subdivided.
- Structural or Constitutional isomerism.
- Stereoisomerism / Spatial isomerism.
Ques. What is Newman’s projection formula? (2 marks)
Ans. The conformation of a chemical bond shows from front to back. The front atom can be represented as a dot, while the back carbon as a circle. This is visualized by a Newman projection, which can be very useful in alkane stereochemistry. The unique dihedral angle that can be found between the proximal and distal atoms can be shown by this form of representation.
Ques. What are the disadvantages of Butane? (3 marks)
Ans. The disadvantages of Butane are given.
- Inhalation of Butane is harmful for human health.
- It can cause severe problems like drowsiness, asphyxia, etc.
- On burning of butane, it releases toxic gas i.e. Nitrogen.
Ques. How many isomers does Butane have? Show their structural formulas. (5 marks)
Ans. Butane can be expressed as an alkane that has four carbon atoms. Its molecular formula is C4H10.
- Butane has two isomers, namely, n-butane and isobutane.
- In the n-butane isomer, it consists of straight chain compounds having four carbon atoms connected together with a single covalent bond.
- Isobutane or 2-methylpropane, where three carbon atoms which come from the parent chain and one carbon atom is placed as the side chain at C-2 of the parent chain.
The structural formula of n-butane and isobutane are:

Do Check Out:






Comments