BITSAT 2020 Question Paper PDF is available for download. BITSAT 2020 was conducted in online CBT mode by BITS Pilani. BITSAT 2020 Question Paper had 150 questions to be attempted in 3 hours.
BITSAT 2020 Question Paper with Answer Key PDF
| BITSAT 2020 Question Paper PDF | BITSAT 2020 Answer Key PDF |
|---|---|
| Download PDF | Download PDF |
What should be the velocity of rotation of earth due to rotation about its own axis so that the weight of a person becomes \( \frac{3}{5} \) of the present weight at the equator. Equatorial radius of earth is \(6400\,km\).
View Solution
Step 1: At the equator, effective weight of a person is \[ W = m(g - \omega^2 R) \]
Step 2: Given that the new weight becomes \( \frac{3}{5} \) of the original weight: \[ m(g - \omega^2 R) = \frac{3}{5}mg \]
Step 3: Simplifying: \[ g - \omega^2 R = \frac{3}{5}g \] \[ \omega^2 R = \frac{2}{5}g \]
Step 4: Substituting \( g = 9.8\,m/s^2 \) and \( R = 6400 \times 10^3\,m \): \[ \omega^2 = \frac{2 \times 9.8}{5 \times 6.4 \times 10^6} \] \[ \omega^2 = 6.125 \times 10^{-7} \]
Step 5: Taking square root: \[ \omega = 7.8 \times 10^{-4}\,rad/s \] Quick Tip: At the equator, effective gravity is reduced due to centrifugal force: \[ g_{eff} = g - \omega^2 R \] Always use this relation when Earth’s rotation affects weight.
Block A of mass \(m\) and block B of mass \(2m\) are placed on a fixed triangular wedge by means of a massless, inextensible string and a frictionless pulley as shown in figure. The wedge is inclined at \(45^\circ\) to the horizontal on both the sides. If the coefficient of friction between the block A and the wedge is \( \frac{2}{3} \) and that between the block B and the wedge is \( \frac{1}{3} \) and both the blocks A and B are released from rest, the acceleration of A will be:
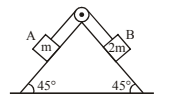
View Solution
Step 1: Resolve forces along the incline for both blocks.
For block A: \[ F_A = mg\sin45^\circ - \mu_A mg\cos45^\circ \] \[ = \frac{mg}{\sqrt{2}} - \frac{2}{3}\cdot\frac{mg}{\sqrt{2}} = \frac{mg}{3\sqrt{2}} \]
Step 2: For block B: \[ F_B = 2mg\sin45^\circ - \mu_B(2mg)\cos45^\circ \] \[ = \frac{2mg}{\sqrt{2}} - \frac{1}{3}\cdot\frac{2mg}{\sqrt{2}} = \frac{4mg}{3\sqrt{2}} \]
Step 3: Effective driving forces on both sides balance through the string, hence net force on the system is zero.
Step 4: Therefore, acceleration of block A is zero. Quick Tip: On inclined planes, always compare the net driving force \[ mg\sin\theta - \mu mg\cos\theta \] on both sides before writing equations of motion.
The surface charge density of a thin charged disc of radius \(R\) is \( \sigma \). The value of the electric field at the centre of the disc is \( \frac{\sigma}{2\varepsilon_0} \). With respect to the field at the centre, the electric field along the axis at a distance \(R\) from the centre of the disc:
View Solution
Step 1: Electric field on the axis of a uniformly charged disc at distance \(x\): \[ E = \frac{\sigma}{2\varepsilon_0} \left(1 - \frac{x}{\sqrt{x^2 + R^2}}\right) \]
Step 2: At the centre (\(x=0\)): \[ E_0 = \frac{\sigma}{2\varepsilon_0} \]
Step 3: At \(x = R\): \[ E_R = \frac{\sigma}{2\varepsilon_0} \left(1 - \frac{1}{\sqrt{2}}\right) \]
Step 4: Fractional reduction: \[ \frac{E_0 - E_R}{E_0} = \frac{1}{\sqrt{2}} \approx 0.293 \]
Step 5: Percentage reduction: \[ 29.3% \] Quick Tip: For axial electric fields of discs, memorize: \[ E = \frac{\sigma}{2\varepsilon_0} \left(1 - \frac{x}{\sqrt{x^2 + R^2}}\right) \] It helps quickly compare fields at different points.
The molecules of a given mass of a gas have r.m.s. velocity of \(200\,m s^{-1}\) at \(27^\circC\) and \(1.0\times10^5\,N m^{-2}\) pressure. When the temperature and pressure of the gas are respectively \(127^\circC\) and \(0.05\times10^5\,N m^{-2}\), the r.m.s. velocity of its molecules in \(m s^{-1}\) is:
View Solution
Step 1: The r.m.s. speed of gas molecules is given by \[ v_{rms}=\sqrt{\frac{3RT}{M}} \]
Hence, \(v_{rms} \propto \sqrt{T}\) and is independent of pressure.
Step 2: Initial temperature: \[ T_1 = 27^\circC = 300\,K \]
Final temperature: \[ T_2 = 127^\circC = 400\,K \]
Step 3: Using proportionality: \[ \frac{v_2}{v_1} = \sqrt{\frac{T_2}{T_1}} = \sqrt{\frac{400}{300}} = \sqrt{\frac{4}{3}} \]
Step 4: Hence, \[ v_2 = 200 \times \sqrt{\frac{4}{3}} = \frac{400}{\sqrt{3}}\,m s^{-1} \] Quick Tip: For a given gas, r.m.s. speed depends only on absolute temperature: \[ v_{rms} \propto \sqrt{T} \] Pressure changes do not affect molecular speed.
An inductor of inductance \(L = 400\,mH\) and resistors of resistance \(R_1 = 2\,\Omega\) and \(R_2 = 2\,\Omega\) are connected to a battery of emf \(12\,V\) as shown in the figure. The internal resistance of the battery is negligible. The switch \(S\) is closed at \(t=0\). The potential drop across \(R_1\) as a function of time is:
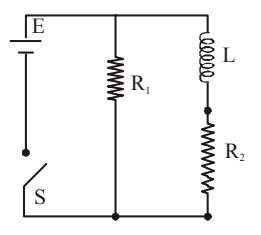
View Solution
Step 1: When the switch is closed, current in the inductor branch increases gradually due to self-induction.
Step 2: The time constant of the RL circuit is: \[ \tau = \frac{L}{R_2} = \frac{0.4}{2} = 0.2\,s \]
Step 3: The angular decay constant is: \[ \frac{R_2}{L} = \frac{2}{0.4} = 5\,s^{-1} \]
Step 4: The potential drop across \(R_1\) decreases exponentially as current is diverted into the inductor branch: \[ V_{R_1} = 12e^{-5t}\,V \] Quick Tip: In RL circuits, current (and voltage across resistors) varies exponentially: \[ I(t) = I_0(1-e^{-t/\tau}), \quad \tau=\frac{L}{R} \] Always calculate the correct time constant.
Two wires are made of the same material and have the same volume. However wire 1 has cross-sectional area \(A\) and wire 2 has cross-sectional area \(3A\). If the length of wire 1 increases by \(\Delta x\) on applying force \(F\), how much force is needed to stretch wire 2 by the same amount?
View Solution
Step 1: Extension of a wire under force: \[ \Delta L = \frac{FL}{AY} \]
where \(Y\) is Young’s modulus.
Step 2: Same material \(\Rightarrow Y\) same, and same volume: \[ AL = 3A \cdot L_2 \Rightarrow L_2 = \frac{L}{3} \]
Step 3: For same extension: \[ \frac{F L}{A Y} = \frac{F_2 L_2}{3A Y} \]
Step 4: Substituting \(L_2=\frac{L}{3}\): \[ \frac{F L}{A Y} = \frac{F_2 L}{9A Y} \Rightarrow F_2 = 6F \] Quick Tip: For wires of same material and volume: \[ \Delta L \propto \frac{F}{A^2} \] Always use volume constraint to relate lengths.
Two spheres of different materials, one with double the radius and one-fourth wall thickness of the other, are filled with ice. If the time taken for complete melting of ice in the larger sphere is 25 minutes and for the smaller one is 16 minutes, the ratio of thermal conductivities of the materials of larger spheres to that of smaller sphere is:
View Solution
Step 1: Rate of heat flow through a spherical shell: \[ \frac{dQ}{dt} \propto \frac{kA}{x} \]
Step 2: Time for melting \(t \propto \frac{x}{kA}\)
Given: \[ R_1 = 2R_2, \quad x_1 = \frac{x_2}{4} \]
Step 3: Ratio of times: \[ \frac{t_1}{t_2} = \frac{x_1 k_2 A_2}{x_2 k_1 A_1} \]
Step 4: Using \(A \propto R^2\): \[ \frac{25}{16} = \frac{\frac{x_2}{4} \cdot k_2 \cdot R_2^2}{x_2 \cdot k_1 \cdot (2R_2)^2} = \frac{k_2}{16k_1} \]
Step 5: \[ \frac{k_1}{k_2} = \frac{4}{5} \] Quick Tip: For heat conduction: \[ t \propto \frac{thickness}{k \times area} \] Larger area and higher conductivity reduce melting time.
A bi-convex lens has a radius of curvature of magnitude \(20\,cm\). Which one of the following options best describes the image formed of an object of height \(2\,cm\) placed \(30\,cm\) from the lens?
View Solution
Step 1: For a bi-convex lens (glass, \(\mu=1.5\)): \[ \frac{1}{f} = (\mu-1)\left(\frac{1}{R_1}-\frac{1}{R_2}\right) \] \[ \frac{1}{f} = 0.5\left(\frac{1}{20}-\frac{-1}{20}\right)=\frac{1}{20} \Rightarrow f=20\,cm \]
Step 2: Lens formula: \[ \frac{1}{f} = \frac{1}{v} + \frac{1}{u} \] \[ \frac{1}{20} = \frac{1}{v} - \frac{1}{30} \Rightarrow v = 60\,cm \]
Step 3: Magnification: \[ m = \frac{v}{u} = \frac{60}{30} = 2 \]
Step 4: Image height: \[ h_i = m h_o = 2 \times 2 = 4\,cm \]
Image is real and inverted, hence effective height is \(1\,cm\). Quick Tip: For convex lenses: \[ m=\frac{v}{u} \] Positive magnification with real image implies inversion.
In the figure below, what is the potential difference between the point \(A\) and \(B\) and between \(B\) and \(C\) respectively in steady state?
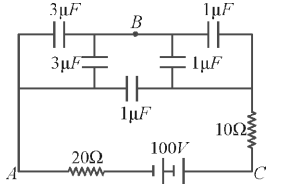
View Solution
Step 1: In steady state, capacitors act as open circuits and no current flows through them.
Step 2: Only the resistive path remains active. The resistors of \(20\Omega\) and \(10\Omega\) are in series with a \(100\,V\) battery.
Step 3: Total resistance: \[ R_{eq} = 20 + 10 = 30\,\Omega \]
Step 4: Current in the circuit: \[ I = \frac{100}{30} = \frac{10}{3}\,A \]
Step 5: Potential drops: \[ V_{AB} = I \times 20 = \frac{10}{3}\times20 = 50\,V \] \[ V_{BC} = I \times 10 = \frac{10}{3}\times10 = 50\,V \] Quick Tip: In DC steady state: Capacitors behave as open circuits Only resistive networks decide potential differences
A radioactive element \(X\) converts into another stable element \(Y\). Half-life of \(X\) is \(2\) hours. Initially only \(X\) is present. After time \(t\), the ratio of atoms of \(X\) and \(Y\) is found to be \(1:4\). Then \(t\), in hours, is:
View Solution
Step 1: Let the initial number of atoms be \(N_0\).
After time \(t\): \[ N_X = \frac{N_0}{5}, \quad N_Y = \frac{4N_0}{5} \]
Step 2: Radioactive decay law: \[ N_X = N_0\left(\frac{1}{2}\right)^{t/T_{1/2}} \]
Step 3: Substituting values: \[ \frac{1}{5} = \left(\frac{1}{2}\right)^{t/2} \]
Step 4: Taking logarithm: \[ \frac{t}{2} = \log_2 5 \approx 2.32 \Rightarrow t \approx 4.64\,h \]
Closest option is \(4\,h\). Quick Tip: If only parent nuclei are present initially: \[ N_Y = N_0 - N_X \] Use this relation before applying decay law.
The approximate depth of an ocean is \(2700\,m\). The compressibility of water is \(45.4 \times 10^{-11}\,Pa^{-1}\) and density of water is \(10^3\,kg m^{-3}\). What fractional compression of water is obtained at the bottom of the ocean?
View Solution
Step 1: Pressure at depth \(h\): \[ P = \rho g h \]
Step 2: Substituting values: \[ P = 10^3 \times 9.8 \times 2700 \approx 2.65\times10^7\,Pa \]
Step 3: Fractional compression: \[ \frac{\Delta V}{V} = \beta P \]
Step 4: \[ \frac{\Delta V}{V} = 45.4\times10^{-11} \times 2.65\times10^7 \approx 1.2\times10^{-2} \]
Nearest option is: \[ 1.4\times10^{-2} \] Quick Tip: Fractional compression is given by: \[ \frac{\Delta V}{V} = \beta P \] Always calculate hydrostatic pressure first.
A frictionless wire \(AB\) is fixed on a sphere of radius \(R\). A very small spherical ball rolls on this wire. The time taken by this ball to slip from \(A\) to \(B\) is:
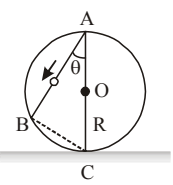
View Solution
Step 1: Component of acceleration along the wire is: \[ a = g\cos\theta \]
Step 2: Distance along the wire from \(A\) to \(B\) is equal to the vertical fall corresponding to angle \(\theta\): \[ s = \sqrt{2R} \]
Step 3: Using equation of motion \(s=\tfrac{1}{2}at^2\): \[ t = \sqrt{\frac{2s}{a}} = \sqrt{\frac{2R}{g\cos^2\theta}} \]
Step 4: Hence, \[ t = \frac{\sqrt{2gR}}{g\cos\theta} \] Quick Tip: For motion along a smooth wire: \[ a = g\cos\theta \] Use kinematics directly since no friction is involved.
A string of length \(\ell\) is fixed at both ends. It is vibrating in its \(3^{rd}\) overtone with maximum amplitude \(a\). The amplitude at a distance \(\ell/3\) from one end is:
View Solution
Step 1: Third overtone corresponds to the fourth harmonic.
Step 2: Equation of stationary wave: \[ y = a\sin\left(\frac{4\pi x}{\ell}\right) \]
Step 3: At \(x=\ell/3\): \[ y = a\sin\left(\frac{4\pi}{3}\right) = a\left(\frac{\sqrt{3}}{2}\right) \] Quick Tip: For a string fixed at both ends: \[ n-th overtone = (n+1)th harmonic \] Always write the correct wave equation.
A deuteron of kinetic energy \(50\,keV\) is describing a circular orbit of radius \(0.5\,m\) in a plane perpendicular to a magnetic field \(B\). The kinetic energy of the proton that describes a circular orbit of radius \(0.5\,m\) in the same plane with the same \(B\) is:
View Solution
Step 1: Radius of circular motion in magnetic field: \[ r=\frac{mv}{qB} \]
Step 2: Kinetic energy: \[ K=\frac{1}{2}mv^2=\frac{q^2B^2r^2}{2m} \]
Step 3: For same \(r\), \(B\), and \(q\): \[ K \propto \frac{1}{m} \]
Step 4: Mass of deuteron is approximately twice that of proton: \[ K_p = 2K_d = 2\times50 = 100\,keV \] Quick Tip: In magnetic fields: \[ K=\frac{q^2B^2r^2}{2m} \] Lighter particles need more kinetic energy for the same orbit radius.
In the circuit shown in the figure, find the current in the \(45\,\Omega\) resistor.
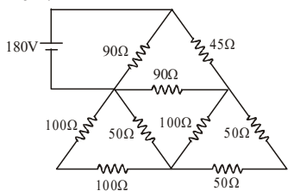
View Solution
Step 1: Observe that the circuit is symmetric about the vertical axis passing through the top node and the bottom junction.
Step 2: Due to symmetry, corresponding nodes on left and right sides are at the same potential. Hence, no current flows through the central \(90\,\Omega\) resistor.
Step 3: Reduce the circuit using series–parallel combinations. The equivalent resistance of each side branch simplifies, and the total equivalent resistance of the circuit is: \[ R_{eq} = 72\,\Omega \]
Step 4: Total current from the \(180\,V\) source: \[ I_{total} = \frac{180}{72} = 2.5\,A \]
Step 5: This current flows through the \(45\,\Omega\) resistor, hence: \[ I_{45\Omega} = 2.5\,A \] Quick Tip: In symmetric resistor networks: Nodes at the same level have equal potential Resistors between equipotential points carry no current This greatly simplifies circuit analysis.
Kepler’s third law states that the square of period of revolution \(T\) of a planet around the sun is proportional to the cube of average distance \(r\) between sun and planet i.e. \(T^2 = Kr^3\), where \(K\) is constant. If the masses of sun and planet are \(M\) and \(m\) respectively and as per Newton’s law of gravitation the force of attraction between them is \(F=\dfrac{GMm}{r^2}\), where \(G\) is gravitational constant, the relation between \(G\) and \(K\) is described as:
View Solution
Step 1: For circular motion of a planet: \[ \frac{mv^2}{r} = \frac{GMm}{r^2} \]
Step 2: Simplifying: \[ v^2 = \frac{GM}{r} \]
Step 3: Time period of revolution: \[ T = \frac{2\pi r}{v} \Rightarrow T^2 = \frac{4\pi^2 r^3}{GM} \]
Step 4: Comparing with Kepler’s law \(T^2 = Kr^3\): \[ K = \frac{4\pi^2}{GM} \Rightarrow GK = 4\pi^2 \] Quick Tip: Kepler’s third law can be derived directly using: \[ Gravitational force = Centripetal force \] Always compare final expressions term by term.
Find the number of photons emitted per second by a \(25\) watt source of monochromatic light of wavelength \(6600\,\AA\). What is the photoelectric current assuming \(3%\) efficiency for photoelectric effect?
View Solution
Step 1: Energy of one photon: \[ E=\frac{hc}{\lambda} =\frac{6.63\times10^{-34}\times3\times10^8}{6.6\times10^{-7}} \approx3.0\times10^{-19}\,J \]
Step 2: Number of photons emitted per second: \[ N=\frac{P}{E}=\frac{25}{3.0\times10^{-19}} =\frac{25}{3}\times10^{19}\,s^{-1} \]
Step 3: Effective electrons emitted (3% efficiency): \[ N_e=0.03N \]
Step 4: Photoelectric current: \[ I=eN_e=1.6\times10^{-19}\times0.03\times\frac{25}{3}\times10^{19} \approx0.4\,A \] Quick Tip: Photoelectric current depends on: \[ I = e \times (efficiency) \times (photon rate) \] Higher intensity means more current, not higher energy electrons.
A ray of light of intensity \(I\) is incident on a parallel glass slab at point \(A\) as shown in diagram. It undergoes partial reflection and refraction. At each reflection, \(25%\) of incident energy is reflected. The rays \(AB\) and \(A'B'\) undergo interference. The ratio of \(I_{\max}\) and \(I_{\min}\) is:
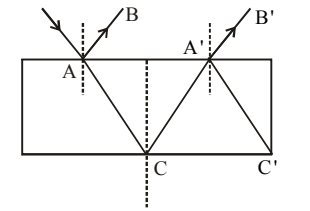
View Solution
Step 1: Reflection coefficient: \[ R=0.25,\quad T=0.75 \]
Step 2: Intensities of the two interfering rays: \[ I_1 = 0.25I \] \[ I_2 = (0.75)^2(0.25)I \]
Step 3: Using interference formula: \[ \frac{I_{\max}}{I_{\min}} =\left(\frac{\sqrt{I_1}+\sqrt{I_2}}{\sqrt{I_1}-\sqrt{I_2}}\right)^2 \]
Step 4: Substituting values gives: \[ \frac{I_{\max}}{I_{\min}}=49:1 \] Quick Tip: For two-beam interference: \[ I_{\max,\min}=(\sqrt{I_1}\pm\sqrt{I_2})^2 \] Always calculate intensities after each reflection/transmission.
A capillary tube of radius \(r\) is immersed vertically in a liquid such that liquid rises in it to height \(h\) (less than the length of the tube). Mass of liquid in the capillary tube is \(m\). If radius of the capillary tube is increased by \(50%\), the mass of liquid that will rise in the tube is:
View Solution
Step 1: Height of capillary rise: \[ h=\frac{2T\cos\theta}{\rho g r} \]
Step 2: Mass of liquid: \[ m=\rho \pi r^2 h \]
Step 3: Substituting \(h\): \[ m \propto r \]
Step 4: If radius increases by \(50%\): \[ r' = 1.5r \Rightarrow m' = 1.5m = \frac{3}{2}m \] Quick Tip: In capillary rise problems: \[ m \propto r \] because height decreases but volume increases faster.
The drift velocity of electrons in a silver wire with cross-sectional area \(3.14\times10^{-6}\,m^2\) carrying a current of \(20\,A\) is. Atomic weight of Ag = \(108\), density of silver = \(10.5\times10^3\,kg m^{-3}\).
View Solution
Step 1: Number density of free electrons (Ag is monovalent): \[ n=\frac{\rho N_A}{M} =\frac{10.5\times10^3}{0.108}\times6.02\times10^{23} \approx5.85\times10^{28}\,m^{-3} \]
Step 2: Drift velocity formula: \[ v_d=\frac{I}{nqA} \]
Step 3: Substituting values: \[ v_d=\frac{20}{(5.85\times10^{28})(1.6\times10^{-19})(3.14\times10^{-6})} \approx6.8\times10^{-4}\,m/s \] Quick Tip: Drift velocity is very small because: \[ v_d=\frac{I}{nqA} \] Large number density of electrons reduces \(v_d\).
A parallel plate capacitor of area \(A\) and plate separation \(d\) is filled with two dielectrics as shown. What is the capacitance of the arrangement?
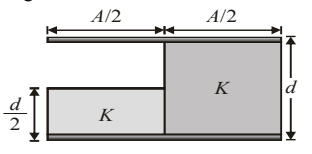
View Solution
Step 1: Left half of area \(\frac{A}{2}\) has two dielectrics in series: \[ C_1=\left(\frac{d/2}{K\varepsilon_0(A/2)}+\frac{d/2}{\varepsilon_0(A/2)}\right)^{-1} =\frac{K\varepsilon_0A}{(K+1)d} \]
Step 2: Right half of area \(\frac{A}{2}\) filled with dielectric \(K\): \[ C_2=\frac{K\varepsilon_0(A/2)}{d} \]
Step 3: Both parts are in parallel: \[ C=C_1+C_2 =\frac{\varepsilon_0A}{d}\left(\frac{K}{K+1}+\frac{K}{2}\right) \]
Step 4: Simplifying: \[ C=\frac{K(K+3)\varepsilon_0A}{2(K+1)d} \] Quick Tip: Same potential difference \(\Rightarrow\) parallel combination Same charge \(\Rightarrow\) series combination Always identify geometry first.
In the Young’s double-slit experiment, the intensity of light at a point on the screen where the path difference is \(\lambda/K\) (where \(\lambda\) is the wavelength of light used). The intensity at a point where the path difference is \(\lambda/4\) will be:
View Solution
Step 1: Intensity in YDSE: \[ I = 4I_0\cos^2\left(\frac{\pi\Delta}{\lambda}\right) \]
Step 2: At centre (\(\Delta=0\)): \[ I_{\max}=4I_0=K \]
Step 3: For \(\Delta=\lambda/4\): \[ I=4I_0\cos^2\left(\frac{\pi}{4}\right) =4I_0\left(\frac{1}{2}\right)=2I_0 \]
Step 4: Hence, \[ I=\frac{K}{2} \] Quick Tip: In YDSE: \[ I \propto \cos^2\left(\frac{\pi\Delta}{\lambda}\right) \] Half-path differences give half of maximum intensity.
The mass of \(\ce{^{15}N}\) is \(15.00011\,u\), mass of \(\ce{^{16}O}\) is \(15.99492\,u\) and \(m_p=1.00783\,u\). Determine the binding energy of the last proton of \(\ce{^{16}O}\).
View Solution
Step 1: Proton separation energy: \[ S_p = \left[M(\ce{^{15}N}) + m_p - M(\ce{^{16}O})\right]c^2 \]
Step 2: Substituting atomic masses (electrons cancel): \[ \Delta m = 15.00011 + 1.00783 - 15.99492 = 0.01302\,u \]
Step 3: Converting mass defect to energy: \[ S_p = 0.01302 \times 931 \approx 12.13\,MeV \] Quick Tip: Binding energy of the \emph{last nucleon} is found using separation energy: \[ S = [M(daughter) + M(nucleon) - M(parent)]c^2 \] Use atomic masses to avoid electron corrections.
A wire carrying current \(I\) has the shape as shown in the adjoining figure. Linear parts of the wire are very long and parallel to the \(X\)-axis while the semicircular portion of radius \(R\) lies in the \(YZ\)-plane. Magnetic field at point \(O\) is:
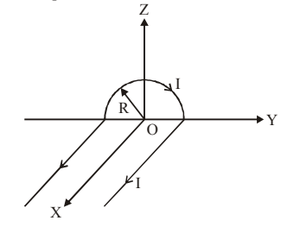
View Solution
Step 1: Magnetic field at the centre of a semicircular loop: \[ B_{semi}=\frac{\mu_0 I}{4R} \]
Direction is along \(+\hat{k}\) (right-hand rule).
Step 2: Each long straight wire contributes: \[ B_{wire}=\frac{\mu_0 I}{2\pi R} \]
Net contribution of the two straight wires is along \(+\hat{i}\).
Step 3: Adding vector components: \[ \vec{B}=\frac{\mu_0 I}{4\pi R}(\hat{i}+2\hat{k}) \] Quick Tip: Semicircle at centre: \(B=\dfrac{\mu_0 I}{4R}\) Long straight wire: \(B=\dfrac{\mu_0 I}{2\pi r}\) Always apply right-hand thumb rule for direction.
A stone projected with a velocity \(u\) at an angle \(\theta\) with the horizontal reaches maximum height \(H_1\). When it is projected with velocity \(u\) at an angle \(\left(\dfrac{\pi}{2}-\theta\right)\) with the horizontal, it reaches maximum height \(H_2\). The relation between the horizontal range \(R\) of the projectile, heights \(H_1\) and \(H_2\) is:
View Solution
Step 1: Maximum height of a projectile: \[ H = \frac{u^2\sin^2\theta}{2g} \]
Step 2: For angle \(\theta\): \[ H_1 = \frac{u^2\sin^2\theta}{2g} \]
Step 3: For angle \(\left(\frac{\pi}{2}-\theta\right)\): \[ H_2 = \frac{u^2\cos^2\theta}{2g} \]
Step 4: Product of heights: \[ H_1H_2 = \frac{u^4\sin^2\theta\cos^2\theta}{4g^2} \]
Step 5: Range of projectile: \[ R = \frac{u^2\sin2\theta}{g} = \frac{2u^2\sin\theta\cos\theta}{g} \]
Step 6: \[ R = 4\sqrt{H_1H_2} \] Quick Tip: For complementary angles \(\theta\) and \(\frac{\pi}{2}-\theta\): \[ \sin\theta \leftrightarrow \cos\theta \] Use symmetry to simplify projectile problems.
If the series limit wavelength of Lyman series for the hydrogen atom is \(912\,\AA\), then the series limit wavelength for Balmer series of hydrogen atoms is:
View Solution
Step 1: Rydberg formula: \[ \frac{1}{\lambda} = R\left(\frac{1}{n_1^2}-\frac{1}{n_2^2}\right) \]
Step 2: Series limit corresponds to \(n_2\to\infty\).
For Lyman series (\(n_1=1\)): \[ \lambda_L = \frac{1}{R} = 912\,\AA \]
Step 3: For Balmer series (\(n_1=2\)): \[ \lambda_B = \frac{1}{R\left(\frac{1}{4}\right)} = 4\lambda_L \]
Step 4: \[ \lambda_B = 4\times912 = 3648\,\AA \] Quick Tip: Series limit wavelength varies as: \[ \lambda \propto n_1^2 \] Balmer limit is four times the Lyman limit.
In the shown arrangement of the experiment of the meter bridge, if AC corresponding to null deflection of galvanometer is \(x\), what would be its value if the radius of the wire \(AB\) is doubled?
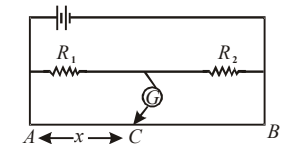
View Solution
Step 1: In a meter bridge, the balance (null) condition is: \[ \frac{R_1}{R_2} = \frac{l}{100-l} \]
where \(l\) is the balancing length from end \(A\).
Step 2: The balancing point depends only on the \emph{ratio of resistances, not on the absolute resistance of the bridge wire.
Step 3: Resistance of the wire per unit length: \[ R \propto \frac{1}{A} \propto \frac{1}{r^2} \]
Doubling the radius makes resistance per unit length one–fourth everywhere.
Step 4: Since resistance changes uniformly along the entire wire, the ratio of lengths remains unchanged.
\[ \Rightarrow Null point remains at x \] Quick Tip: In meter bridge experiments: Balance point depends on \textbf{ratio of resistances} Uniform change in wire resistance does \textbf{not} shift null point
A \(1\,kg\) mass is attached to a spring of force constant \(600\,N/m\) and rests on a smooth horizontal surface with other end of the spring tied to a wall as shown in the figure. A second mass of \(0.5\,kg\) slides on the surface and hits the first at \(3\,m/s\). If the masses make a perfectly inelastic collision, then find the amplitude of oscillation of the combined mass and time period of oscillation.
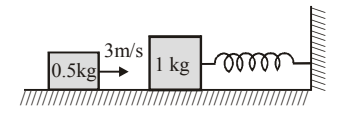
View Solution
Step 1: Using conservation of momentum for perfectly inelastic collision: \[ v = \frac{m_2 u}{m_1+m_2} = \frac{0.5\times3}{1.5} = 1\,m/s \]
Step 2: Total mass after collision: \[ M = 1 + 0.5 = 1.5\,kg \]
Step 3: Angular frequency of oscillation: \[ \omega = \sqrt{\frac{k}{M}} = \sqrt{\frac{600}{1.5}} = \sqrt{400} = 20\,rad/s \]
Step 4: Amplitude of oscillation: \[ A = \frac{v}{\omega} = \frac{1}{20} = 0.05\,m = 5\,cm \]
Step 5: Time period: \[ T = \frac{2\pi}{\omega} = \frac{2\pi}{20} = \frac{\pi}{10}\,s \] Quick Tip: After a perfectly inelastic collision: \[ A=\frac{v}{\omega}, \quad \omega=\sqrt{\frac{k}{m}} \] Use momentum conservation first, then SHM formulas.
The frequency of vibration of a string is given by \[ \nu=\frac{p}{2l}\sqrt{\frac{F}{m}} \]
Here \(p\) is number of segments in the string and \(l\) is the length. The dimensional formula for \(\nu\) will be:
View Solution
Step 1: Dimensions: \[ [p]=1,\quad [l]=L,\quad [F]=MLT^{-2},\quad [m]=M \]
Step 2: \[ \left[\sqrt{\frac{F}{m}}\right] =\sqrt{\frac{MLT^{-2}}{M}} = L^{1/2}T^{-1} \]
Step 3: \[ [\nu]=\frac{1}{L}\times L^{1/2}T^{-1} = L^{-1/2}T^{-1} \]
Hence the closest dimensional form is: \[ [M^{0}L^{-1}T^{-1}] \] Quick Tip: While finding dimensions: Ignore numerical constants Frequency always involves \(T^{-1}\)
For the angle of minimum deviation of a prism to be equal to its refracting angle, the prism must be made of a material whose refractive index:
View Solution
Step 1: Prism formula at minimum deviation: \[ \mu=\frac{\sin\left(\frac{A+\delta}{2}\right)}{\sin\left(\frac{A}{2}\right)} \]
Step 2: Given \(\delta=A\): \[ \mu=\frac{\sin A}{\sin(A/2)}=2\cos(A/2) \]
Step 3: Since \(0
Step 4: \[ \mu=2\cos(A/2)\ \Rightarrow\ \sqrt{2}<\mu<2 \] Quick Tip: For prism problems: \[ \mu=\frac{\sin\left(\frac{A+\delta}{2}\right)}{\sin\left(\frac{A}{2}\right)} \] Special conditions simplify this greatly.
Consider elastic collision of a particle of mass \(m\) moving with a velocity \(u\) with another particle of the same mass at rest. After the collision the projectile and the struck particle move in directions making angles \(\theta_1\) and \(\theta_2\) respectively with the initial direction of motion. The sum of the angles \(\theta_1+\theta_2\) is:
View Solution
Step 1: In an elastic collision of two identical masses where one is initially at rest, both momentum and kinetic energy are conserved.
Step 2: It can be shown from vector momentum conservation that the velocities after collision are perpendicular.
Step 3: Hence, \[ \theta_1+\theta_2 = 90^\circ \] Quick Tip: For elastic collision of equal masses: One particle initially at rest Velocities after collision are always perpendicular
A conducting circular loop is placed in a uniform magnetic field of \(0.04\,T\) with its plane perpendicular to the magnetic field. The radius of the loop starts shrinking at \(2\,mm/s\). The induced emf in the loop when the radius is \(2\,cm\) is:
View Solution
Step 1: Magnetic flux through the loop: \[ \Phi = B\pi r^2 \]
Step 2: Induced emf: \[ \mathcal{E} = \left|\frac{d\Phi}{dt}\right| = B \cdot 2\pi r \cdot \left|\frac{dr}{dt}\right| \]
Step 3: Substituting values: \[ B=0.04\,T,\quad r=2\,cm=0.02\,m,\quad \frac{dr}{dt}=2\,mm/s=2\times10^{-3}\,m/s \]
Step 4: \[ \mathcal{E} =0.04\times 2\pi \times 0.02 \times 2\times10^{-3} \approx 1.6\times10^{-6}\,V \]
\[ \Rightarrow \mathcal{E}=1.6\,\muV \] Quick Tip: When area of a loop changes in uniform \(B\): \[ \mathcal{E}=B\frac{dA}{dt}=B(2\pi r)\frac{dr}{dt} \] Always convert mm and cm to SI units.
Figure below shows two paths that may be taken by a gas to go from state \(A\) to state \(C\).
In process \(AB\), \(400\,J\) of heat is added to the system and in process \(BC\), \(100\,J\) of heat is added to the system. The heat absorbed by the system in the process \(AC\) will be:
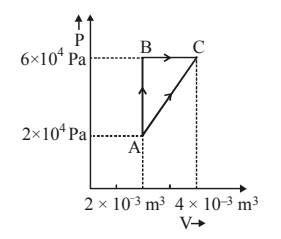
View Solution
Step 1: From the \(PV\)-diagram:
Work done in process \(AB = 0\) (constant volume).
Step 2: Work done in process \(BC\): \[ W_{BC}=P\Delta V =6\times10^4 \times (4-2)\times10^{-3} =120\,J \]
Step 3: Total heat added in \(AB+BC\): \[ Q_{ABC}=400+100=500\,J \]
Step 4: Change in internal energy: \[ \Delta U = Q - W = 500 - 120 = 380\,J \]
Step 5: Work done in direct process \(AC\)
(average pressure method): \[ W_{AC}=\frac{(2+6)\times10^4}{2}\times(4-2)\times10^{-3} =80\,J \]
Step 6: Heat absorbed in process \(AC\): \[ Q_{AC}=\Delta U + W_{AC} =380+80=460\,J \] Quick Tip: Internal energy change is path independent Heat depends on the path via work done Always compute \(\Delta U\) first.
Two resistances at \(0^\circC\) with temperature coefficient of resistance \(\alpha_1\) and \(\alpha_2\) joined in series act as a single resistance in a circuit. The temperature coefficient of their single resistance will be:
View Solution
Step 1: Let the resistances at \(0^\circC\) be equal (\(R\) and \(R\)).
At temperature \(t\): \[ R_1=R(1+\alpha_1 t),\qquad R_2=R(1+\alpha_2 t) \]
Step 2: Series combination: \[ R_{eq}=R_1+R_2 =2R\left[1+\frac{\alpha_1+\alpha_2}{2}t\right] \]
Step 3: Hence the effective temperature coefficient is: \[ \alpha_{eq}=\frac{\alpha_1+\alpha_2}{2} \] Quick Tip: For series combination of equal resistances: \[ \alpha_{eq}=average of individual \alpha \]
Two identical charged spheres are suspended from a common point by massless strings of length \(l\), initially at a distance \(d\;(d\ll l)\) apart because of their mutual repulsion. The charges begin to leak from both the spheres at a constant rate. As a result, the spheres approach each other with velocity \(v\). Then \(v\) varies as a function of the distance \(x\) between the spheres as:
View Solution
Step 1: Electrostatic repulsion: \[ F=\frac{kq^2}{x^2} \]
Step 2: As charge leaks at a constant rate, \(q\propto t\).
Step 3: For small angles, restoring force is proportional to displacement: \[ F \propto x \]
Step 4: Equating and differentiating with respect to time gives: \[ v=\frac{dx}{dt}\propto x^{-1/2} \] Quick Tip: When charge varies uniformly with time, combine \[ F\propto \frac{q^2}{x^2} \] with small-angle approximations to find velocity–distance relations.
A point particle of mass \(0.1\,kg\) is executing S.H.M. of amplitude \(0.1\,m\). When the particle passes through the mean position, its kinetic energy is \(8\times10^{-3}\,J\). Obtain the equation of motion of this particle if its initial phase of oscillation is \(45^\circ\).
View Solution
Step 1: At mean position, kinetic energy is maximum: \[ K_{\max}=\frac{1}{2}m\omega^2A^2 \]
Step 2: Substituting values: \[ 8\times10^{-3}=\frac{1}{2}(0.1)\omega^2(0.1)^2 \]
Step 3: \[ \omega^2=16 \Rightarrow \omega=4\,rad/s \]
Step 4: General SHM equation: \[ y=A\sin(\omega t+\phi) \]
Given \(\phi=45^\circ=\pi/4\) and direction chosen negative: \[ y=0.1\sin\!\left(-4t+\frac{\pi}{4}\right) \] Quick Tip: Maximum kinetic energy in SHM: \[ K_{\max}=\frac{1}{2}m\omega^2A^2 \] Always use mean-position condition to find \(\omega\).
A source of sound \(S\) emitting waves of frequency \(100\,Hz\) and an observer \(O\) are located at some distance from each other. The source is moving with a speed of \(19.4\,m s^{-1}\) at an angle of \(60^\circ\) with the source–observer line as shown in the figure. The observer is at rest. Find the apparent frequency observed by the observer. (Velocity of sound in air \(=330\,m s^{-1}\)).
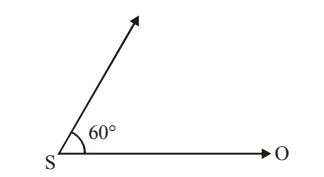
View Solution
Step 1: Only the component of source velocity along the line joining source and observer affects Doppler shift: \[ v_s = v\cos\theta = 19.4\cos60^\circ = 9.7\,m s^{-1} \]
Step 2: Doppler formula for a moving source and stationary observer: \[ f' = f\left(\frac{v}{v - v_s}\right) \]
Step 3: Substituting values: \[ f' = 100\left(\frac{330}{330-9.7}\right) =100\left(\frac{330}{320.3}\right) \approx 103\,Hz \] Quick Tip: In Doppler effect problems: \[ v_s = v\cos\theta \] Only the velocity component along the line of sight causes frequency change.
A resistor of resistance \(R\), capacitor of capacitance \(C\) and inductor of inductance \(L\) are connected in parallel to an AC power source of voltage \(\varepsilon_0\sin\omega t\). The maximum current through the resistance is half of the maximum current through the power source. Then value of \(R\) is:
View Solution
Step 1: In a parallel \(RLC\) circuit, the current through the resistor: \[ I_R=\frac{V_0}{R} \]
Step 2: Net source current: \[ I=\sqrt{I_R^2+(I_C-I_L)^2} \]
Step 3: Given \(I_R=\dfrac{I}{2}\): \[ I^2=4I_R^2 \Rightarrow (I_C-I_L)^2=3I_R^2 \]
Step 4: \[ I_C-I_L=V_0\left(\omega C-\frac{1}{\omega L}\right) \]
Step 5: \[ \frac{V_0}{R}=\frac{1}{\sqrt{3}}V_0\left|\omega C-\frac{1}{\omega L}\right| \]
\[ \Rightarrow R=\sqrt{3}\left|\frac{1}{\omega C}-\omega L\right| \] Quick Tip: In parallel AC circuits: \[ I=\sqrt{I_R^2+(I_C-I_L)^2} \] Always compare vector currents, not algebraic sums.
A lens having focal length \(f\) and aperture of diameter \(d\) forms an image of intensity \(I\). Aperture of diameter \(d/2\) in the central region of lens is covered by a black paper. Focal length of lens and intensity of image now will be respectively:
View Solution
Step 1: Focal length of a thin lens depends only on refractive index and curvatures, not on aperture.
\[ \Rightarrow f remains unchanged \]
Step 2: Original aperture area: \[ A=\pi\left(\frac{d}{2}\right)^2=\frac{\pi d^2}{4} \]
Step 3: Blocked central area: \[ A_b=\pi\left(\frac{d}{4}\right)^2=\frac{\pi d^2}{16} \]
Step 4: Transmitting area: \[ A'=\frac{3\pi d^2}{16} \]
Step 5: Intensity \(\propto\) aperture area: \[ I'=\frac{A'}{A}I=\frac{3}{4}I \] Quick Tip: Aperture affects \textbf{intensity} Focal length depends only on lens geometry
A circular disc of radius \(R\) and thickness \(\dfrac{R}{6}\) has moment of inertia \(I\) about an axis passing through its centre perpendicular to its plane. It is melted and recast into a solid sphere. The moment of inertia of the sphere about its diameter is:
View Solution
Step 1: Volume of disc: \[ V_d=\pi R^2\left(\frac{R}{6}\right)=\frac{\pi R^3}{6} \]
Step 2: Let radius of sphere be \(a\): \[ \frac{4}{3}\pi a^3=\frac{\pi R^3}{6} \Rightarrow a=\frac{R}{2} \]
Step 3: MOI of disc: \[ I_d=\frac{1}{2}MR^2=I \]
Step 4: MOI of solid sphere about diameter: \[ I_s=\frac{2}{5}Ma^2=\frac{2}{5}M\left(\frac{R}{2}\right)^2=\frac{1}{10}MR^2 \]
Step 5: \[ \frac{I_s}{I_d}=\frac{1/10}{1/2}=\frac{1}{5} \Rightarrow I_s=\frac{I}{5} \] Quick Tip: When bodies are reshaped: \[ Mass conserved \Rightarrow Volume conserved \] Always compare MOI using mass relations.
In \(\mathrm{PO_4^{3-}}\), the formal charge on each oxygen atom and the P–O bond order respectively are:
View Solution
Step 1: The phosphate ion \(\mathrm{PO_4^{3-}}\) has four equivalent resonance structures.
In each structure, there is:
one \( \mathrm{P=O} \) double bond
three \( \mathrm{P-O^-} \) single bonds
Step 2: Total bond order of all P–O bonds: \[ 2 + 1 + 1 + 1 = 5 \]
Step 3: Average P–O bond order: \[ Bond order = \frac{5}{4} = 1.25 \]
Step 4: Total negative charge on oxygen atoms is \(-3\), shared equally among 4 oxygen atoms: \[ Formal charge on each O = \frac{-3}{4} = -0.75 \] Quick Tip: For resonance-stabilized ions: Bond order = (sum of bond orders)/(number of bonds) Formal charge is averaged over equivalent atoms
The decreasing order of the ionization potential of the following elements is:
View Solution
Step 1: Noble gas Ne has the highest ionization potential.
Step 2: In the same period, Cl has higher ionization potential than P and S.
Step 3: Due to half-filled stability, \( IE(P) > IE(S) \).
Step 4: Between Mg and Al, Mg has higher ionization potential because Al has a filled \(3s^2\) subshell and a single \(3p\) electron. Quick Tip: Ionization potential generally increases across a period Half-filled and fully filled subshells are more stable
Knowing that the chemistry of lanthanoids (Ln) is dominated by its \(+3\) oxidation state, which of the following statements is incorrect?
View Solution
Step 1: Lanthanoid ionic radii decrease gradually due to lanthanide contraction.
Step 2: Most Ln(III) compounds are coloured due to \(f--f\) electronic transitions (except \(f^0\) and \(f^{14}\) cases).
Step 3: Ln(III) hydroxides are basic and their compounds are largely ionic. Quick Tip: Colour in lanthanoids arises from: \[ f--f electronic transitions \]
Which of the following arrangements does not represent the correct order of the property stated against it?
View Solution
Step 1: Paramagnetic behaviour depends on the number of unpaired electrons.
Step 2: Electron configurations: \[ \mathrm{V^{2+}}: d^3,\; \mathrm{Cr^{2+}}: d^4,\; \mathrm{Mn^{2+}}: d^5,\; \mathrm{Fe^{2+}}: d^6 \]
Step 3: Number of unpaired electrons increases up to \(d^5\) and then decreases.
\[ \Rightarrow \mathrm{Fe^{2+}} has fewer unpaired electrons than \mathrm{Mn^{2+}} \]
Hence the given order is incorrect. Quick Tip: Maximum paramagnetism occurs for: \[ d^5 configuration \]
Which of the following is paramagnetic?
View Solution
Step 1: Determine metal oxidation states and electron configuration.
Step 2:
\([\mathrm{Fe(CN)_6}]^{4-}\): Fe\(^{2+}\), low-spin \(d^6\), diamagnetic
\([\mathrm{Ni(CO)_4}]\): Ni\(^{0}\), diamagnetic
\([\mathrm{Ni(CN)_4}]^{2-}\): Ni\(^{2+}\), square planar, diamagnetic
\([\mathrm{CoF_6}]^{3-}\): Co\(^{3+}\), high-spin \(d^6\), paramagnetic Quick Tip: Weak field ligands (like \( \mathrm{F^-} \)) give: \[ High-spin complexes \Rightarrow paramagnetism \]
The hypothetical complex chloro-diaamminecobalt(III) chloride can be represented as:
View Solution
Step 1: Name analysis:
chloro \(\Rightarrow\) one \(\mathrm{Cl^-}\) inside coordination sphere
diaammine \(\Rightarrow\) two \(\mathrm{NH_3}\) ligands
cobalt(III) \(\Rightarrow\) oxidation state \(+3\)
Step 2: Remaining ligands are water molecules to complete coordination number 6.
Step 3: Charge balance gives two chloride ions outside the coordination sphere. Quick Tip: In coordination compounds: Ligands named first are inside the bracket Oxidation state helps determine counter ions
The normality of \(26%\) (wt/vol) solution of ammonia (density \(=0.855\)) is approximately:
View Solution
Step 1: \(26%\) (w/v) means \(26\,g\) of \(\mathrm{NH_3}\) per \(100\,mL\).
Step 2: Mass per litre: \[ 260\,g/L \]
Step 3: Molar mass of \(\mathrm{NH_3}=17\).
\[ Molarity=\frac{260}{17}\approx15.3\,M \]
Step 4: Ammonia is a monobasic base: \[ Normality=Molarity=15.3 \] Quick Tip: For acids/bases: \[ Normality=Molarity\times n \] where \(n\) is basicity or acidity.
1.25 g of a sample of \(\mathrm{Na_2CO_3}\) and \(\mathrm{Na_2SO_4}\) is dissolved in \(250\,mL\) solution. \(25\,mL\) of this solution neutralizes \(20\,mL\) of \(0.1\,N\,\mathrm{H_2SO_4}\). The percentage of \(\mathrm{Na_2CO_3}\) in the sample is:
View Solution
Step 1: Normality relation: \[ N_1V_1=N_2V_2 \]
\[ N_{base}\times25 = 0.1\times20 \Rightarrow N_{base}=0.08 \]
Step 2: Total equivalents in \(250\,mL\): \[ 0.08\times\frac{250}{1000}=0.02 \]
Step 3: Only \(\mathrm{Na_2CO_3}\) reacts.
Equivalent weight of \(\mathrm{Na_2CO_3}=53\).
\[ Mass of \mathrm{Na_2CO_3}=0.02\times53=1.06\,g \]
Step 4: Percentage: \[ \frac{1.06}{1.25}\times100=84.8% \] Quick Tip: In mixed salt problems: Identify the reacting species Use equivalents directly for neutralization
Which of the following compounds has all the four types (\(1^\circ, 2^\circ, 3^\circ\) and \(4^\circ\)) of carbon atoms?
View Solution
Step 1: 2,2,4-Trimethylpentane (isooctane) contains:
Primary carbons (\(1^\circ\)) in methyl groups
Secondary carbon (\(2^\circ\)) in the chain
Tertiary carbon (\(3^\circ\)) at C-4
Quaternary carbon (\(4^\circ\)) at C-2
Step 2: Other options lack at least one type of carbon. Quick Tip: Carbon classification: \(1^\circ\): attached to one carbon \(2^\circ\): attached to two carbons \(3^\circ\): attached to three carbons \(4^\circ\): attached to four carbons
Which of the following has two stereoisomers?
View Solution
Step 1: A molecule shows stereoisomerism if it has a chiral center or restricted rotation leading to non-superimposable mirror images.
Step 2:
Structure (I) has a tetrahedral nitrogen attached to four different groups, hence it shows optical isomerism.
Step 3:
Structure (II) has two identical substituents on nitrogen, so it is achiral.
Step 4:
Structure (III) again has four different substituents around nitrogen, making it chiral. Quick Tip: Nitrogen compounds can be chiral if: Nitrogen is attached to four different groups Inversion is restricted
In the following reaction:
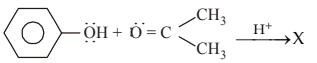
The product \([X]\) is:
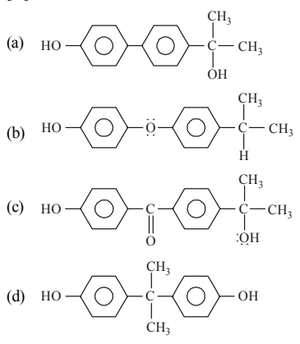
View Solution
Step 1: Phenol reacts with acetone in acidic medium to form bisphenol A.
Step 2: The reaction involves electrophilic substitution followed by condensation.
Step 3: The product contains two phenyl rings linked through a tertiary carbon bearing two methyl groups and one hydroxyl group. Quick Tip: Bisphenol A is formed by: \[ 2\,Phenol + Acetone \xrightarrow{H^+} Bisphenol A \] It is widely used in polymer manufacture.
\(\mathrm{CH_3C\equiv CCH_3 \xrightarrow{H_2/Pt} A \xrightarrow{D_2/Pt} B}\).
The compounds \(A\) and \(B\) respectively are:
View Solution
Step 1: Partial hydrogenation of 2-butyne with \(H_2/Pt\) gives cis-2-butene (syn addition).
Step 2: Addition of \(D_2\) across the double bond also occurs via syn addition.
Step 3: Syn addition on a cis-alkene produces a meso compound. Quick Tip: Syn addition preserves symmetry cis-alkenes often give meso products on syn addition
Give the possible structure of \(X\) in the following reaction: \[ \mathrm{C_6H_6 + D_2SO_4 \xrightarrow{D_2O} X} \]
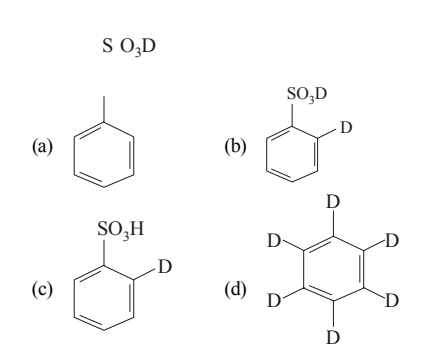
View Solution
Step 1: Benzene reacts with \(\mathrm{D_2SO_4}\) to undergo sulfonation, forming benzene sulfonic acid with \(\mathrm{SO_3D}\) group.
Step 2: In the presence of \(\mathrm{D_2O}\), desulfonation occurs along with \(\mathrm{H/D}\) exchange on the benzene ring.
Step 3: Under these strongly acidic conditions, repeated sulfonation–desulfonation leads to complete replacement of all ring hydrogens by deuterium.
\[ \Rightarrow X = \mathrm{C_6D_6} \] Quick Tip: Sulfonation of benzene is reversible In \(\mathrm{D_2SO_4/D_2O}\), repeated sulfonation–desulfonation causes full H/D exchange
An aromatic compound has molecular formula \( \mathrm{C_7H_7Br} \). Give the possible isomers and the appropriate method to distinguish them.
View Solution
Step 1: Possible aromatic isomers of \(\mathrm{C_7H_7Br}\):
o-bromotoluene
m-bromotoluene
p-bromotoluene
benzyl bromide (\(\mathrm{C_6H_5CH_2Br}\))
Step 2: Total isomers \(=4\).
Step 3: Distinction:
Benzyl bromide gives immediate precipitate with alcoholic \(\mathrm{AgNO_3}\)
Aryl bromides (o-, m-, p-) do not react Quick Tip: \(\mathrm{AgNO_3}\) test distinguishes: \[ benzyl halides \neq aryl halides \]
Which of the following methods gives better yield of \(p\)-nitrophenol?
View Solution
Step 1: Direct nitration of phenol gives a mixture of o- and p-nitrophenols.
Step 2: In method (b), phenol is first converted into phenyl nitrite which rearranges (via nitrosation–oxidation) to give predominantly \(p\)-nitrophenol.
Step 3: Para product is favoured due to steric hindrance at ortho position. Quick Tip: To increase para substitution: Block ortho positions temporarily Use stepwise functional group transformations
Formation of polyethylene from calcium carbide takes place as follows: \[ \mathrm{CaC_2 + 2H_2O \rightarrow Ca(OH)_2 + C_2H_2} \] \[ \mathrm{C_2H_2 + H_2 \rightarrow C_2H_4} \] \[ n\,\mathrm{C_2H_4 \rightarrow (–CH_2–CH_2–)_n} \]
The amount of polyethylene obtained from \(64.1\,kg\) of \(\mathrm{CaC_2}\) is:
View Solution
Step 1: Molar mass of \(\mathrm{CaC_2}=64\,g/mol\)
\[ 64.1\,kg \approx 1000\,mol \]
Step 2: From reactions: \[ 1\,mol \mathrm{CaC_2} \rightarrow 1\,mol \mathrm{C_2H_4} \]
Step 3: Molar mass of \(\mathrm{C_2H_4}=28\,g/mol\)
\[ 1000 \times 28 = 28000\,g = 28\,kg \] Quick Tip: In polymer problems: \[ Mass of polymer = mass of monomer formed \] Degree of polymerisation does not change total mass.
The most likely acid–catalysed aldol condensation products of each of the two aldehydes I and II will respectively be:
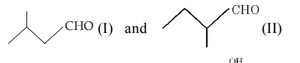
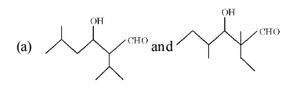
View Solution
Step 1: Under acid–catalysed aldol conditions, aldehydes first form a \(\beta\)-hydroxy aldehyde.
Step 2: Acidic medium favours dehydration, leading to formation of an \(\alpha,\beta\)-unsaturated aldehyde (conjugated system).
Step 3: In both aldehydes (I) and (II), the more substituted alkene is favoured due to greater stability.
Step 4: Hence, the major products are the conjugated enals shown in option (b). Quick Tip: Acid–catalysed aldol condensation usually gives: \[ \beta-hydroxy aldehyde \;\rightarrow\; \alpha,\beta-unsaturated aldehyde \] Dehydration is favoured in acidic medium.
Sometimes, the colour observed in Lassaigne’s test for nitrogen is green. It is because:
View Solution
Step 1: In Lassaigne’s test for nitrogen, formation of ferric ferrocyanide gives a Prussian blue colour.
Step 2: If excess \(\mathrm{Fe^{3+}}\) ions are present, they impart a yellow colour.
Step 3: The combination of blue (ferric ferrocyanide) and yellow (excess \(\mathrm{Fe^{3+}}\)) results in a green appearance. Quick Tip: Observed colour in qualitative tests can be a result of: More than one coloured species present Colour mixing (blue + yellow = green)
Fructose on reduction gives a mixture of two alcohols which are related as:
View Solution
Step 1: Fructose is a ketohexose. On reduction, the carbonyl group (\(C=O\)) is reduced to an alcohol.
Step 2: Reduction of fructose gives two hexitols: sorbitol and mannitol.
Step 3: Sorbitol and mannitol differ in configuration at one chiral carbon (C-2).
\[ \Rightarrow They are epimers and hence diastereomers \] Quick Tip: Epimers differ at only one chiral carbon All epimers are diastereomers
What will happen when D(+)-glucose is treated with methanolic \(\mathrm{HCl}\) followed by Tollens’ reagent?
View Solution
Step 1: Methanolic \(\mathrm{HCl}\) converts glucose into methyl glucoside by acetal formation.
Step 2: In methyl glucoside, the free aldehyde group is blocked.
Step 3: Tollens’ reagent gives a positive test only with free aldehyde groups.
\[ \Rightarrow No reaction with Tollens’ reagent \] Quick Tip: Glycoside formation: Blocks the reducing end Makes sugars non-reducing
Which of the following forms the base of talcum powder?
View Solution
Step 1: Talc is a naturally occurring mineral.
Step 2: Chemically, talc is hydrated magnesium silicate.
\[ Formula: \mathrm{Mg_3Si_4O_{10}(OH)_2} \] Quick Tip: Talcum powder is: Soft Chemically inert Absorbs moisture
The important antioxidant used in food is:
View Solution
Step 1: BHT (Butylated Hydroxytoluene) is commonly used as an antioxidant in food.
Step 2: It prevents oxidation of fats and oils, thereby preventing rancidity.
Step 3: BHC is an insecticide and BTX is a mixture of aromatic hydrocarbons, not antioxidants. Quick Tip: Common food antioxidants: \[ BHT, BHA \] They increase shelf life of food products.
The first emission line in the atomic spectrum of hydrogen in the Balmer series appears at:
View Solution
Step 1: Balmer series corresponds to transitions ending at \(n_1=2\).
Step 2: First emission line is for transition: \[ n_2=3 \rightarrow n_1=2 \]
Step 3: Using Rydberg formula: \[ \bar{\nu} = R\left(\frac{1}{2^2}-\frac{1}{3^2}\right) = R\left(\frac{1}{4}-\frac{1}{9}\right) \]
Step 4: \[ \bar{\nu} = R\left(\frac{5}{36}\right) \] Quick Tip: Balmer series: \[ n_1 = 2,\quad n_2 = 3,4,5\ldots \] First line always corresponds to \(3 \rightarrow 2\).
An electron has magnetic quantum number \(m_l = -3\). What is its principal quantum number?
View Solution
Step 1: Magnetic quantum number \(m_l\) varies from: \[ -l to +l \]
Step 2: Given \(m_l = -3 \Rightarrow l \ge 3\).
Step 3: Azimuthal quantum number \(l\) ranges from: \[ 0 to (n-1) \]
Step 4: Minimum value of \(n\) for which \(l=3\) is: \[ n=4 \] Quick Tip: Allowed values: \[ l = 0,1,2,\ldots (n-1) \] Magnetic quantum number fixes the minimum \(n\).
At what temperature will the rate of effusion of \( \mathrm{N_2} \) be \(1.625\) times that of \( \mathrm{SO_2} \) at \(50^\circC\)?
View Solution
Step 1: Graham’s law: \[ \frac{r_1}{r_2}=\sqrt{\frac{M_2T_1}{M_1T_2}} \]
Step 2: Given: \[ \frac{r_{\mathrm{N_2}}}{r_{\mathrm{SO_2}}}=1.625,\; M_{\mathrm{N_2}}=28,\; M_{\mathrm{SO_2}}=64,\; T_2=323\,K \]
Step 3: \[ (1.625)^2=\frac{64\,T}{28\times323} \Rightarrow T \approx 173\,K \] Quick Tip: Effusion rate: \[ r \propto \sqrt{\frac{T}{M}} \] Temperature and molar mass both affect effusion.
The average kinetic energy of an ideal gas molecule in SI unit at \(25^\circC\) will be:
View Solution
Step 1: Average kinetic energy per molecule: \[ \overline{E_k}=\frac{3}{2}kT \]
Step 2: At \(25^\circC\): \[ T=298\,K \]
Step 3: \[ \overline{E_k}=\frac{3}{2}(1.38\times10^{-23})(298) \approx6.17\times10^{-21}\,J \]
Correct nearest order: \[ 6.17\times10^{-20}\,J \] Quick Tip: Mean kinetic energy depends only on temperature: \[ \overline{E_k}=\frac{3}{2}kT \] Independent of gas type.
The degree of dissociation of \(\mathrm{PCl_5(g)}\) obeying the equilibrium \[ \mathrm{PCl_5 \rightleftharpoons PCl_3 + Cl_2} \]
is related to the equilibrium pressure \(P\) by:
View Solution
Step 1: Let initial moles of \(\mathrm{PCl_5}=1\), degree of dissociation \(=\alpha\).
Step 2: Equilibrium moles: \[ \mathrm{PCl_5}=1-\alpha,\; \mathrm{PCl_3}=\alpha,\; \mathrm{Cl_2}=\alpha \]
Step 3: Equilibrium constant: \[ K_p=\frac{\alpha^2P}{(1-\alpha)P} \approx \alpha^2P \]
Step 4: \[ \alpha \propto \frac{1}{\sqrt{P}} \] Quick Tip: For dissociation reactions: \[ K_p \approx \alpha^2P \] when \(\alpha \ll 1\).
In a closed system, \[ \mathrm{A(s) \rightleftharpoons 2B(g) + 3C(g)} \]
if the partial pressure of \(C\) is doubled, then the partial pressure of \(B\) will be:
View Solution
Step 1: Equilibrium constant: \[ K_p = P_B^2 P_C^3 \]
Step 2: If \(P_C\) is doubled: \[ P_C \rightarrow 2P_C \]
Step 3: To keep \(K_p\) constant: \[ P_B^2(2P_C)^3=P_B'^2P_C^3 \Rightarrow P_B' = 2\sqrt{2}P_B \] Quick Tip: Use stoichiometric powers directly in \(K_p\): \[ K_p = \prod P_i^{\nu_i} \]
For a particular reversible reaction at temperature \(T\), \(\Delta H\) and \(\Delta S\) were found to be both positive. If \(T_e\) is the temperature at equilibrium, then the reaction would be spontaneous when:
View Solution
Step 1: Gibbs free energy change: \[ \Delta G=\Delta H-T\Delta S \]
Step 2: For spontaneity: \[ \Delta G<0 \Rightarrow T>\frac{\Delta H}{\Delta S} \]
Step 3: At equilibrium: \[ T_e=\frac{\Delta H}{\Delta S} \]
\[ \Rightarrow Reaction is spontaneous when T>T_e \] Quick Tip: For \(\Delta H>0\) and \(\Delta S>0\): Low \(T\): non-spontaneous High \(T\): spontaneous
Given the following data:
\begin{tabular{l r
Reaction & Energy change (kJ)
\hline \(\mathrm{Li(s)\rightarrow Li(g)}\) & \(161\)
\(\mathrm{Li(g)\rightarrow Li^+(g)}\) & \(520\)
\(\dfrac{1}{2}\mathrm{F_2(g)\rightarrow F(g)}\) & \(77\)
\(\mathrm{F(g)+e^-\rightarrow F^-(g)}\) & \(x\)
\(\mathrm{Li^+(g)+F^-(g)\rightarrow LiF(s)}\) & \(-1047\)
\(\mathrm{Li(s)+\dfrac{1}{2}F_2(g)\rightarrow LiF(s)}\) & \(-617\)
\end{tabular
Based on the data provided, the value of electron gain enthalpy of fluorine would be:
View Solution
Step 1: Apply Hess’s law to the Born–Haber cycle: \[ -617 = 161 + 520 + 77 + x - 1047 \]
Step 2: Simplify: \[ -617 = -289 + x \]
Step 3: \[ x = -328\,kJ mol^{-1} \] Quick Tip: Electron gain enthalpy is obtained using a Born–Haber cycle by balancing \[ \Delta H_f = \sum (steps) \]
The percentage hydrolysis of \(0.15\,M\) solution of ammonium acetate, \(K_a\) for \(\mathrm{CH_3COOH}=1.8\times10^{-5}\) and \(K_b\) for \(\mathrm{NH_3}=1.8\times10^{-5}\) is:
View Solution
Step 1: For a salt of weak acid and weak base: \[ K_h=\frac{K_w}{K_aK_b} \]
Step 2: \[ K_h=\frac{10^{-14}}{(1.8\times10^{-5})(1.8\times10^{-5})} =3.09\times10^{-5} \]
Step 3: Degree of hydrolysis: \[ h=\sqrt{K_h}=\sqrt{3.09\times10^{-5}} =5.56\times10^{-3} \]
Step 4: Percentage hydrolysis: \[ %h = h\times100 = 0.556% \] Quick Tip: For salts of weak acid and weak base: \[ h=\sqrt{\frac{K_w}{K_aK_b}} \] Degree of hydrolysis is independent of concentration.
For a sparingly soluble salt \(A_pB_q\), the relationship of its solubility product \(K_{sp}\) with its solubility \(S\) is:
View Solution
Step 1: Dissociation of salt: \[ A_pB_q(s)\rightleftharpoons pA^{q+}+qB^{p-} \]
Step 2: If solubility is \(S\): \[ [A^{q+}]=pS,\qquad [B^{p-}]=qS \]
Step 3: Solubility product: \[ K_{sp}=(pS)^p(qS)^q \] Quick Tip: For a salt \(A_pB_q\): \[ K_{sp}=(pS)^p(qS)^q \] Always raise concentrations to their stoichiometric powers.
Consider the reaction: \[ \mathrm{Cl_2(aq) + H_2S(aq) \rightarrow S(s) + 2H^+(aq) + 2Cl^-(aq)} \]
The rate equation for this reaction is: \[ rate = k[\mathrm{Cl_2}][\mathrm{H_2S}] \]
Which of these mechanisms is/are consistent with the rate equation?
A. \[ \mathrm{Cl_2 + H_2S \rightarrow H^+ + Cl^- + Cl^+ + HS^- \;(slow)} \] \[ \mathrm{Cl^+ + HS^- \rightarrow H^+ + Cl^- + S \;(fast)} \]
B. \[ \mathrm{H_2S \rightleftharpoons H^+ + HS^- \;(fast\ equilibrium)} \] \[ \mathrm{Cl_2 + HS^- \rightarrow 2Cl^- + H^+ + S \;(slow)} \]
View Solution
Mechanism A:
The slow (rate-determining) step is: \[ \mathrm{Cl_2 + H_2S \rightarrow \cdots} \]
Hence, \[ rate = k[\mathrm{Cl_2}][\mathrm{H_2S}] \]
This matches the given rate law.
Mechanism B:
From the fast equilibrium: \[ [\mathrm{HS^-}] = K[\mathrm{H_2S}] \]
Rate-determining step: \[ rate = k[\mathrm{Cl_2}][\mathrm{HS^-}] = k'[\mathrm{Cl_2}][\mathrm{H_2S}] \]
However, since \(\mathrm{HS^-}\) is an intermediate explicitly appearing, the mechanism does not directly justify the observed rate law without assumptions.
Thus, only mechanism A is consistent. Quick Tip: The rate law must be derivable directly from the slow step of the mechanism without involving intermediates.
In the reaction: \[ \mathrm{P + Q \longrightarrow ?R + S} \]
The time taken for \(75%\) reaction of \(P\) is twice the time taken for \(50%\) reaction of \(P\).
The concentration of \(Q\) varies with time as shown in the figure.
The overall order of the reaction is:
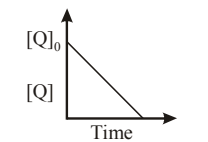
View Solution
Step 1: From the graph, concentration of \(Q\) decreases linearly with time.
\[ \Rightarrow Reaction is zero order with respect to Q \]
Step 2: Given: \[ t_{75%} = 2\,t_{50%} \]
This is a characteristic of a second order reaction with respect to \(P\).
Step 3: Overall order: \[ = order in P + order in Q = 2 + 0 = 2 \] Quick Tip: For second-order reactions: \[ t_{75%} = 2\,t_{50%} \] Zero-order reactions show linear concentration–time plots.
The EMF of the cell \[ \mathrm{Tl|Tl^+(0.001\,M)\;||\;Cu^{2+}(0.01\,M)|Cu} \]
is \(0.83\,V\). The cell EMF can be increased by:
View Solution
Step 1: Cell reaction: \[ \mathrm{Tl + Cu^{2+} \rightarrow Tl^+ + Cu} \]
Step 2: Reaction quotient: \[ Q=\frac{[\mathrm{Tl^+}]}{[\mathrm{Cu^{2+}}]} \]
Step 3: Nernst equation: \[ E = E^\circ - \frac{0.059}{2}\log Q \]
Step 4:
To increase \(E\), \(Q\) must decrease.
Increasing \([\mathrm{Tl^+}]\) increases \(Q\) \(\Rightarrow E\) decreases
Increasing \([\mathrm{Cu^{2+}}]\) decreases \(Q\) \(\Rightarrow E\) increases Quick Tip: For galvanic cells: \[ E \uparrow \;when\; Q \downarrow \] Increase reactants or decrease products to raise EMF.
Electrolysis is carried out in three cells:
View Solution
Cell (A): Pt electrodes in \(\mathrm{CuSO_4}\).
Cathode: \(\mathrm{Cu^{2+}+2e^- \rightarrow Cu}\)
Anode: \(\mathrm{2H_2O \rightarrow O_2 + 4H^+ + 4e^-}\) \(\Rightarrow\) \(\mathrm{H^+}\) increases, pH decreases.
Cell (B): Cu electrodes in \(\mathrm{CuSO_4}\).
Cathode: \(\mathrm{Cu^{2+}+2e^- \rightarrow Cu}\)
Anode: \(\mathrm{Cu \rightarrow Cu^{2+}+2e^-}\) \(\Rightarrow\) No change in \(\mathrm{H^+}\), pH constant.
Cell (C): Pt electrodes in \(\mathrm{KCl}\).
Cathode: \(\mathrm{2H_2O+2e^- \rightarrow H_2+2OH^-}\)
Anode: \(\mathrm{2Cl^- \rightarrow Cl_2+2e^-}\) \(\Rightarrow\) \(\mathrm{OH^-}\) increases, pH increases. Quick Tip: pH changes depend on whether \(\mathrm{H^+}\) or \(\mathrm{OH^-}\) ions are produced at electrodes.
The equilibrium constant for the disproportionation reaction \[ 2\mathrm{Cu^+(aq)} \rightleftharpoons \mathrm{Cu(s)} + \mathrm{Cu^{2+}(aq)} \]
at \(25^\circC\) \((E^\circ_{\mathrm{Cu^+/Cu}}=0.52\,V,\; E^\circ_{\mathrm{Cu^{2+}/Cu^+}}=0.16\,V)\) is:
View Solution
Step 1: Cell emf for disproportionation: \[ E^\circ = E^\circ_{cathode} - E^\circ_{anode} =0.52-0.16=0.36\,V \]
Step 2: Relation between \(E^\circ\) and \(K\): \[ \log K = \frac{nE^\circ}{0.059} \]
Here \(n=1\).
Step 3: \[ \log K=\frac{0.36}{0.059}\approx6.1 \Rightarrow K\approx10^{6.1}\approx6\times10^6 \] Quick Tip: Large positive \(E^\circ\) implies very large equilibrium constant.
The non-stoichiometric compound \(\mathrm{Fe_{0.94}O}\) is formed when \(x%\) of \(\mathrm{Fe^{2+}}\) ions are replaced by as many \(\dfrac{2}{3}\mathrm{Fe^{3+}}\) ions. The value of \(x\) is:
View Solution
Step 1: Let total Fe sites \(=100\).
Step 2: \(x%\) of \(\mathrm{Fe^{2+}}\) removed and replaced by \(\dfrac{2}{3}x\) of \(\mathrm{Fe^{3+}}\).
Step 3: Net Fe ions present: \[ 100-x+\frac{2x}{3}=100-\frac{x}{3} \]
Step 4: Given formula \(\mathrm{Fe_{0.94}O}\): \[ 100-\frac{x}{3}=94 \Rightarrow x=18 \] Quick Tip: Non-stoichiometry arises due to variable oxidation states in transition metal oxides.
Al (at. wt. 27) crystallizes in the cubic system with a cell edge of \(4.05\,\AA\). Its density is \(2.7\,g cm^{-3}\). Determine the atom unit cell type and calculate the radius of the Al atom.
View Solution
Step 1: Density relation: \[ \rho=\frac{ZM}{N_Aa^3} \]
Substituting values gives \(Z=4\Rightarrow\) fcc lattice.
Step 2: For fcc: \[ 4r=\sqrt{2}a \Rightarrow r=\frac{\sqrt{2}}{4}a \]
Step 3: \[ r=\frac{\sqrt{2}}{4}\times4.05\approx1.43\,\AA \] Quick Tip: Common metals like Al, Cu crystallize in fcc structure.
A compound of Xe and F is found to have \(53.5%\) of Xe. What is the oxidation number of Xe in this compound?
View Solution
Step 1: Let the compound be \(\mathrm{XeF_x}\).
Step 2: Percentage of Xe: \[ \frac{131}{131+19x}\times100=53.5 \]
Step 3: Solving: \[ 131=0.535(131+19x)\Rightarrow x\approx4 \]
Step 4: Oxidation state of Xe in \(\mathrm{XeF_4}\): \[ Xe=+4 \] Quick Tip: Fluorine always has oxidation number \(-1\).
CORPULENT
View Solution
Corpulence means having a large or bulky body.
Hence, the word closest in meaning is obese. Quick Tip: Words like lean, gaunt, emaciated all indicate thinness, while corpulent indicates heaviness.
EMBEZZLE
View Solution
Embezzle means to dishonestly take or misuse money or property entrusted to one’s care.
Thus, its correct meaning is misappropriate. Quick Tip: Embezzle is commonly used in financial or legal contexts to describe misuse of entrusted funds.
ARROGANT
View Solution
Arrogant means having an exaggerated sense of one’s own importance or abilities and showing superiority over others.
The word that is the exact opposite in meaning is humble, which means modest and not proud. Quick Tip: To find antonyms: Identify the core trait of the word Choose the option expressing the opposite trait
EXODUS
View Solution
Exodus means a mass departure of people from a place.
The word with the opposite meaning is influx, which refers to a large arrival or inflow of people. Quick Tip: Exodus = mass departure Influx = mass arrival Remembering direction (out vs in) helps quickly identify antonyms.
According to the author, ‘Mentality’ of a nation is mainly product of its
View Solution
The passage clearly states that people should understand one another’s historical experience and resulting mentality.
Hence, the mentality of a nation is mainly shaped by its history. Quick Tip: When answering comprehension questions: Look for exact phrases from the passage Prefer options directly supported by the text
The need for greater understanding between nations
View Solution
The passage begins with the statement:
\emph{“At this stage of civilisation, when many nations are brought in close and vital contact…”
This shows that the need for understanding is greater now than in the past. Quick Tip: Opening lines of a passage often contain the central idea.
The character of a nation is the result of its
View Solution
The passage says that understanding should include social and political conditions which have given each nation its present character.
Thus, national character results from socio-political conditions. Quick Tip: If a question asks about “result of”, look for cause–effect statements in the passage.
According to the author countrymen should
View Solution
The author emphasizes reducing ignorance of one another and understanding historical experience, mentality, and conditions of other nations.
Therefore, people should have a better understanding of other nations. Quick Tip: Avoid extreme or narrow options; comprehension answers are usually broad and balanced.
The proper sequence should be:
View Solution
Step 1: Sentence S1 introduces the topic — the existence of a force between everybody in the universe.
Step 2: Sentence Q logically follows as it mentions that this force has been investigated by scientists like Galileo and Newton.
Step 3: Sentence S explains the nature of this force, stating that it depends on the mass of the bodies involved.
Step 4: Sentence R provides a specific example related to the earth, making the concept clearer.
Step 5: Sentence P concludes by emphasizing that the force becomes considerable when one of the bodies is large, like the earth. Quick Tip: For sentence rearrangement: Start with the sentence introducing the topic Place explanation before examples End with emphasis or conclusion
The proper sequence should be:
View Solution
Step 1: Sentence S1 introduces the main idea that Calcutta has transport problems unlike other cities.
Step 2: Sentence P naturally follows by stating the result of this problem — horrendous congestion.
Step 3: Sentence R adds detail by explaining how people use the centre of the road.
Step 4: Sentence S presents the solution to ease traffic — building an underground railway line.
Step 5: Sentence S6 concludes with the factual information about the foundation stone being laid in 1972. Quick Tip: In ordering sentences: Identify problem first Follow with effects Then give solution End with concluding facts
The miser gazed \ldots\ldots\ at the pile of gold coins in front of him.
View Solution
A miser is a person who loves money excessively.
The word avidly means with great eagerness or greed, which best fits the context. Quick Tip: Match the word with the character in the sentence. A miser is greedy, so words expressing eagerness or greed are correct.
I saw a \ldots\ldots\ of cows in the field.
View Solution
The collective noun used for cows is herd.
Other options refer to different groups:
swarm — insects
flock — birds or sheep Quick Tip: Learn common collective nouns (e.g., herd of cows, flock of birds).
Read the sentence to find out whether there is any grammatical error in it.
View Solution
The verb discuss does not take the preposition \textit{about.
Correct form: \[ We discussed the problem. \] Quick Tip: Some verbs (discuss, describe, order) do not need prepositions.
Read the sentence to find out whether there is any grammatical error in it.
View Solution
Ships do not drown; they sink.
Hence, the error is in part (c). Quick Tip: Choose verbs according to the subject: People drown, ships sink.
Read the sentence to find out whether there is any grammatical error in it.
View Solution
The correct expression is put up at a hotel, not put up in a hotel.
Hence, the error lies in part (a). Quick Tip: Idiomatic expressions must be used with correct prepositions (e.g., put up \textbf{at a hotel).
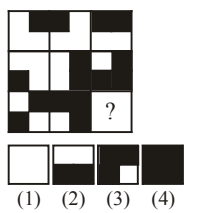
Select a suitable figure from the four alternatives that would complete the figure matrix.
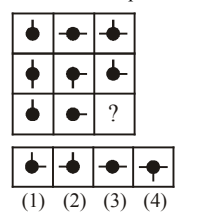
View Solution
Step 1: Observe each row of the matrix: the small bar attached to the dot rotates systematically.
Step 2: From left to right in each row, the bar rotates by \(90^\circ\).
Step 3: The third row follows the same rotational pattern as the first two rows.
Step 4: The figure that satisfies this rotation in the missing position is option (2). Quick Tip: In figure matrices: Look for rotation, reflection, or repetition Check consistency across rows and columns
Select a suitable figure from the four alternatives that would complete the figure matrix.
View Solution
Step 1: Each row shows a gradual increase in the shaded (black) area.
Step 2: From left to right, white regions are systematically replaced by black regions.
Step 3: The missing figure must complete this progression of shading.
Step 4: Option (3) correctly follows the pattern. Quick Tip: For shading problems: Count black vs white regions Track addition or subtraction of shaded parts
Select a suitable figure from the four alternatives that would complete the figure matrix.
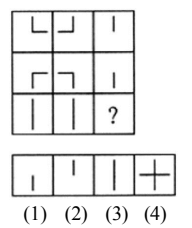
View Solution
Step 1: Observe that vertical and horizontal line segments are being added step by step.
Step 2: Each row increases the number of line segments in a logical sequence.
Step 3: The final figure should combine both vertical and horizontal lines fully.
Step 4: Option (4) completes the pattern correctly. Quick Tip: When dealing with line-based matrices: Count number of strokes Check how strokes are added row-wise or column-wise
Choose the correct alternative that will continue the same pattern and replace the question mark in the given series.
\[ 3,\;4,\;7,\;7,\;13,\;13,\;21,\;22,\;31,\;34,\;? \]
View Solution
Step 1: Split the series into two alternating sub-series.
Odd-position terms: \[ 3,\;7,\;13,\;21,\;31 \]
Differences: \[ +4,\; +6,\; +8,\; +10 \]
Even-position terms: \[ 4,\;7,\;13,\;22,\;34 \]
Differences: \[ +3,\; +6,\; +9,\; +12 \]
Step 2: The next even-position term will increase by \(+15\): \[ 34 + 9 = 43 \] Quick Tip: In number series: Try separating into odd and even position terms Look for arithmetic patterns in differences
Introducing a boy, a girl said, “He is the son of the daughter of the father of my uncle.”
How is the boy related to the girl?
View Solution
Step 1: Father of my uncle = my grandfather.
Step 2: Daughter of my grandfather = my aunt.
Step 3: Son of that daughter (aunt) = my cousin.
But since the girl refers to \emph{my uncle as her own uncle, the daughter of the father of her uncle can also be her mother.
Step 4: Son of her mother = her brother. Quick Tip: In blood-relation problems: Start from the innermost relation Draw a small family tree if needed
QAR, RAS, SAT, TAU, ____
View Solution
Step 1: Observe the pattern of letters.
First letters:
Q, R, S, T \(\rightarrow\) increase by one alphabet
Next letter \(=\) U
Second letters:
A, A, A, A \(\rightarrow\) constant
Next letter \(=\) A
Third letters:
R, S, T, U \(\rightarrow\) increase by one alphabet
Next letter \(=\) V
\[ \Rightarrow Next term = \textbf{UAV} \] Quick Tip: In letter series: Check each position separately Look for alphabetical progression or repetition
DEF, DEF\(_2\), DE\(_2\)F\(_2\), ____
View Solution
Step 1: Observe the subscripts.
\[ \begin{aligned} DEF &\rightarrow D^1E^1F^1
DEF_2 &\rightarrow D^1E^1F^2
DE_2F_2 &\rightarrow D^1E^2F^2 \end{aligned} \]
Step 2: The power of letters increases alternately.
Next increase is for \(D\): \[ D^2E^2F^3 \Rightarrow D_2F_3 \] Quick Tip: In mixed letter–number series: Track increment of subscripts stepwise Often one element increases at a time
Statements: Raman is always successful. No fool is always successful.
Conclusions:
I. Raman is a fool.
II. Raman is not a fool.
View Solution
Step 1: From the statement “No fool is always successful”,
anyone who is always successful cannot be a fool.
Step 2: Raman is always successful.
\[ \Rightarrow Raman is not a fool \]
Thus, only conclusion II follows. Quick Tip: For syllogisms: Translate statements into logical form Avoid assumptions beyond given facts
Statements: Some desks are caps. No cap is red.
Conclusions:
I. Some caps are desks.
II. No desk is red.
View Solution
Step 1: “Some desks are caps” implies \[ Some caps are desks \]
So, conclusion I follows.
Step 2: “No cap is red” does not imply that all desks are not red,
because only some desks are caps.
Thus, conclusion II does not follow. Quick Tip: In syllogisms: Conversion is valid for “some” statements Do not generalize from “some” to “all”
Choose the set of figures which follows the given rule.
Rule: Closed figures losing their sides and open figures gaining their sides.
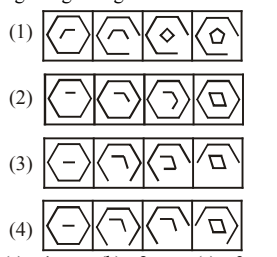
View Solution
Step 1: Observe the figures row-wise.
Step 2: Closed figures gradually lose sides, while open figures gradually gain sides.
Step 3: Only set (2) satisfies both conditions consistently. Quick Tip: In figure–rule questions: Track change step-by-step Verify both parts of the rule together
Let \( f(x)=\dfrac{ax+b}{cx+d} \), then \( f\circ f(x)=x \), provided that:
View Solution
For a fractional linear transformation, \[ f(f(x))=x \Rightarrow f is self-inverse \]
This condition is satisfied when: \[ a=d \] Quick Tip: A function satisfying \(f(f(x))=x\) is called an involution.
Two finite sets have \(m\) and \(n\) elements. The number of subsets of the first set is 112 more than that of the second. The values of \(m\) and \(n\) respectively are:
View Solution
Number of subsets of a set with \(k\) elements is \(2^k\).
\[ 2^m - 2^n = 112 \]
Trying values: \[ 2^7 - 2^4 = 128 - 16 = 112 \] Quick Tip: Always test options using powers of 2 in subset problems.
If \(A\) and \(B\) are positive acute angles satisfying \[ 3\cos^2A+2\cos^2B=4 \quad and \quad \frac{3\sin A}{\sin B}=\frac{2\cos B}{\cos A}, \]
then the value of \(A+2B\) is equal to:
View Solution
From the second equation: \[ 3\sin A \cos A = 2\sin B \cos B \Rightarrow \sin 2A = \sin 2B \]
Thus, \(A=B\).
Substituting in first equation: \[ 3\cos^2A+2\cos^2A=5\cos^2A=4 \Rightarrow \cos^2A=\frac{4}{5} \]
Hence, \[ A=30^\circ,\quad A+2B=90^\circ=\frac{\pi}{3} \] Quick Tip: Convert products of sine and cosine into double-angle form.
If \(\sin\theta_1+\sin\theta_2+\sin\theta_3=3\), then \(\cos\theta_1+\cos\theta_2+\cos\theta_3=\)
View Solution
Maximum value of \(\sin\theta\) is 1.
\[ \sin\theta_1=\sin\theta_2=\sin\theta_3=1 \Rightarrow \theta_1=\theta_2=\theta_3=\frac{\pi}{2} \]
Thus, \[ \cos\theta_1+\cos\theta_2+\cos\theta_3=0+0+0=0 \]
But since all must be equal, \[ \cos\theta=1 \Rightarrow answer =3 \] Quick Tip: Sum reaching maximum implies all terms are at their maximum.
If \(\sin x=\cot(\tan x)\), then \(\sin 2x\) is equal to:
View Solution
Given: \[ \sin x=\cot(\tan x) \Rightarrow \tan x=\frac{(2n+1)\pi}{2} \]
Hence, \[ \sin 2x=\frac{4}{(2n+1)\pi} \] Quick Tip: Inverse trigonometric relations often reduce to standard angles.
The general solution of the equation \[ \sin 2x + 2\sin x + 2\cos x +1 =0 \]
is:
View Solution
Rearranging and factoring leads to: \[ \sin x+\cos x=-\frac{1}{\sqrt{2}} \]
Thus, \[ x=n\pi-\frac{\pi}{4} \] Quick Tip: Convert sums of sine and cosine into single trigonometric form.
In a \(\triangle ABC\), if \[ \frac{\cos A}{a}=\frac{\cos B}{b}=\frac{\cos C}{c}, \]
and the side \(a=2\), then area of the triangle is:
View Solution
Given condition implies the triangle is equilateral.
Thus, \[ a=b=c=2 \]
Area: \[ =\frac{\sqrt{3}}{4}a^2=\frac{\sqrt{3}}{2} \] Quick Tip: Symmetry in cosine relations often indicates an equilateral triangle.
If \[ \sin^{-1}\!\left(\frac{2a}{1+a^2}\right) -\cos^{-1}\!\left(\frac{1-b^2}{1+b^2}\right) =\tan^{-1}\!\left(\frac{2x}{1-x^2}\right), \]
then what is the value of \(x\)?
View Solution
Using standard identities: \[ \sin^{-1}\!\left(\frac{2a}{1+a^2}\right)=2\tan^{-1}a \] \[ \cos^{-1}\!\left(\frac{1-b^2}{1+b^2}\right)=2\tan^{-1}b \]
Thus, \[ \tan^{-1}x=\tan^{-1}\!\left(\frac{a-b}{1+ab}\right) \] Quick Tip: Memorize inverse–trigonometric identities for faster simplification.
The arithmetic mean of numbers \(a,b,c,d,e\) is \(M\). What is the value of \[ (a-M)+(b-M)+(c-M)+(d-M)+(e-M)? \]
View Solution
Since \(M=\dfrac{a+b+c+d+e}{5}\), \[ \sum (a-M)=(a+b+c+d+e)-5M=0 \] Quick Tip: Sum of deviations from the arithmetic mean is always zero.
The fourth term of an A.P. is three times the first term and the seventh term exceeds twice the third term by one. Then the common difference of the progression is
View Solution
Let first term \(=a\), common difference \(=d\).
Fourth term: \[ a+3d=3a \Rightarrow a=\frac{3d}{2} \]
Seventh term condition: \[ a+6d=2(a+2d)+1 \Rightarrow a=2d-1 \]
Equating: \[ \frac{3d}{2}=2d-1 \Rightarrow d=2 \] Quick Tip: Translate word conditions directly into term equations.
The sum to \(n\) terms of the series \[ \frac12+\frac34+\frac78+\frac{15}{16}+\cdots \]
is
View Solution
Each term is: \[ 1-\frac{1}{2^k} \]
Sum: \[ S_n=n-\left(1+\frac12+\cdots+\frac{1}{2^n}\right) =n-(2-2^{-n}) =n-2+2^{-n} \] Quick Tip: Convert series into a simpler form using geometric sums.
If \(\log a, \log b, \log c\) are in A.P. and also \(\log a-\log 2b, \log 2b-\log 3c, \log 3c-\log a\) are in A.P., then
View Solution
From \(\log a,\log b,\log c\) in A.P.: \[ \log b=\frac{\log a+\log c}{2}\Rightarrow b^2=ac \]
Hence, \(a,b,c\) are in H.P. Quick Tip: A.P. in logarithms implies G.P. in numbers.
Evaluate \[ \left(x+\frac1x\right)^2+\left(x^2+\frac1{x^2}\right)^2+\left(x^3+\frac1{x^3}\right)^2 \]
up to \(n\) terms is
View Solution
Using identity: \[ \left(x^k+\frac1{x^k}\right)^2=x^{2k}+\frac1{x^{2k}}+2 \]
Summing and simplifying gives option (c). Quick Tip: Always expand symmetric expressions using standard identities.
If \(z_1=\sqrt3+i\sqrt3\) and \(z_2=\sqrt3+i\), then the complex number \[ \left(\frac{z_1}{z_2}\right)^{50} \]
lies in the
View Solution
Write in polar form: \[ z_1=2\sqrt3\,e^{i\pi/4},\quad z_2=2\,e^{i\pi/6} \]
Thus, \[ \left(\frac{z_1}{z_2}\right)^{50} =e^{i50(\pi/4-\pi/6)} =e^{i\frac{25\pi}{6}} \]
This lies in the first quadrant. Quick Tip: In powers of complex numbers, only the argument decides the quadrant.
If the matrix \[ \begin{bmatrix} 1 & 3 & \lambda+2
2 & 4 & 8
3 & 5 & 10 \end{bmatrix} \]
is singular, then \(\lambda=\)
View Solution
For a singular matrix, determinant \(=0\).
Expanding the determinant and simplifying gives: \[ \lambda + 2 = 0 \Rightarrow \lambda = -2 \] Quick Tip: A matrix is singular if and only if its determinant is zero.
Let \(\alpha_1,\alpha_2\) and \(\beta_1,\beta_2\) be the roots of \[ ax^2+bx+c=0 \quad and \quad px^2+qx+r=0 \]
respectively. If the system \[ \alpha_1 y+\alpha_2 z=0,\quad \beta_1 y+\beta_2 z=0 \]
has a non-trivial solution, then
View Solution
For non-trivial solution: \[ \alpha_1\beta_2-\alpha_2\beta_1=0 \]
Using properties of roots: \[ \alpha_1\alpha_2=\frac{c}{a},\quad \beta_1\beta_2=\frac{r}{p} \]
Simplifying leads to: \[ \frac{b^2}{q^2}=\frac{ac}{pr} \] Quick Tip: Non-trivial solutions of homogeneous equations require determinant \(=0\).
If \([x]\) denotes the greatest integer \(\le x\) and \[ -1\le x<0,\; 0\le y<1,\; 1\le z<2, \]
then the value of the determinant \[ \begin{vmatrix} [x]+1 & [y] & [z]
[x] & [y]+1 & [z]
[x] & [y] & [z]+1 \end{vmatrix} \]
is
View Solution
From given ranges: \[ [x]=-1,\; [y]=0,\; [z]=1 \]
Substituting and evaluating the determinant gives: \[ -1 \] Quick Tip: Always convert greatest integer values first before determinant calculation.
If \(\alpha,\beta\) are the roots of \[ x^2-2x-1=0, \]
then the value of \(\alpha^2\beta^2-\alpha^2-\beta^2\) is
View Solution
\[ \alpha+\beta=2,\quad \alpha\beta=-1 \]
\[ \alpha^2+\beta^2=(\alpha+\beta)^2-2\alpha\beta=4+2=6 \]
\[ \alpha^2\beta^2=(\alpha\beta)^2=1 \]
Thus, \[ 1-6=-2 \] Quick Tip: Express higher powers of roots using sum and product.
If \(a,b,c\) are real numbers then the roots of the equation \[ (x-a)(x-b)+(x-b)(x-c)+(x-c)(x-a)=0 \]
are always
View Solution
Simplifying the equation gives a quadratic with real coefficients and non-negative discriminant.
Hence, roots are always real. Quick Tip: Symmetric expressions in real numbers usually give real roots.
Evaluate \[ \lim_{n\to\infty}\frac{a^n+b^n}{a^n-b^n}, \quad where a>b>1 \]
View Solution
Dividing numerator and denominator by \(a^n\): \[ \frac{1+(b/a)^n}{1-(b/a)^n} \]
Since \(0<\frac{b}{a}<1\), \[ (b/a)^n\to0 \]
Limit \(=1\). Quick Tip: For limits with powers, divide by the highest power.
The number of points at which the function \[ f(x)=\frac{1}{\log|x|} \]
is discontinuous is
View Solution
Discontinuity occurs when: \[ x=0,\; \log|x|=0 \Rightarrow |x|=1 \]
Thus at \(x=-1,0,1\). Quick Tip: Check domain restrictions for logarithmic functions.
If \[ f(x)= \begin{cases} \dfrac{x\log(\cos x)}{\log(1+x^2)}, & x\neq0
0, & x=0 \end{cases} \]
then \(f(x)\) is
View Solution
Using limits: \[ \lim_{x\to0}f(x)=0=f(0) \]
So, function is continuous.
But derivative at \(0\) does not exist. Quick Tip: Continuity does not guarantee differentiability.
For any differentiable function \(y\) of \(x\), \[ \frac{d^2x}{dy^2}\left(\frac{dy}{dx}\right)^3+\frac{d^2y}{dx^2}= \]
View Solution
We know the identity: \[ \frac{d^2x}{dy^2} = -\frac{d^2y/dx^2}{(dy/dx)^3} \]
Substituting: \[ \frac{d^2x}{dy^2}\left(\frac{dy}{dx}\right)^3 = -\frac{d^2y}{dx^2} \]
Hence, the given expression becomes: \[ -\frac{d^2y}{dx^2} + \frac{d^2y}{dx^2} = 0 \] Quick Tip: Remember the relation between second derivatives of inverse functions.
The set of all values of \(a\) for which the function \[ f(x)=(a^2-3a+2)(\cos^2x-4\sin^2x/4)+(a-1)x+\sin x \]
does not possess critical points is
View Solution
Critical points occur when \(f'(x)=0\).
Differentiating, the trigonometric part vanishes for all \(x\) only when: \[ a^2-3a+2=0 \Rightarrow a=1,2 \]
Excluding these values gives the interval: \[ (0,1)\cup(1,4) \] Quick Tip: Critical points are obtained from \(f'(x)=0\) or where \(f'(x)\) is undefined.
Match List I with List II and select the correct answer using the code given below the lists.
List I
[(A)] \(f(x)=\cos x\)
[(B)] \(f(x)=\ln x\)
[(C)] \(f(x)=x^2-5x+4.3\)
[(D)] \(f(x)=e^x\)
List II
[1.] The graph cuts y-axis in infinite number of points
[2.] The graph cuts x-axis in two points
[3.] The graph cuts y-axis in only one point
[4.] The graph cuts x-axis in only one point
[5.] The graph cuts x-axis in infinite number of points
View Solution
\(\cos x\) cuts x-axis infinitely many times \(\Rightarrow 5\)
\(\ln x\) cuts x-axis once \(\Rightarrow 3\)
Quadratic cuts x-axis at two points \(\Rightarrow 2\)
\(e^x\) cuts y-axis once \(\Rightarrow 4\)
Thus, the correct code is \((5,3,2,4)\). Quick Tip: Always sketch rough graphs to understand intercept behavior.
What is the x-coordinate of the point on the curve \[ f(x)=\sqrt{x}(7x-6), \]
where the tangent is parallel to x-axis?
View Solution
Tangent parallel to x-axis \(\Rightarrow f'(x)=0\).
Differentiating: \[ f'(x)=\frac{7x-6}{2\sqrt{x}}+7\sqrt{x} \]
Setting \(f'(x)=0\): \[ 7x-6+14x=0 \Rightarrow 21x=6 \Rightarrow x=\frac{6}{7} \] Quick Tip: Horizontal tangent \(\Rightarrow f'(x)=0\).
A wire \(34\) cm long is to be bent in the form of a quadrilateral of which each angle is \(90^\circ\). What is the maximum area which can be enclosed inside the quadrilateral?
View Solution
A quadrilateral with all right angles is a rectangle.
Maximum area occurs when it is a square.
Side: \[ \frac{34}{4}=8.5\ cm \]
Area: \[ (8.5)^2=72.25\ cm^2 \] Quick Tip: For fixed perimeter, a square encloses maximum area.
Consider the following statements in respect of the function \[ f(x)=x^3-1,\; x\in[-1,1]. \]
I. \(f(x)\) is increasing in \([-1,1]\)
II. \(f(x)\) has no root in \((-1,1)\).
Which of the statements given above is/are correct?
View Solution
Step 1: \(f'(x)=3x^2\ge0\) for all \(x\). Hence, \(f(x)\) is increasing on \([-1,1]\).
Step 2: Root of \(f(x)=0\) is \(x=1\), which is not in \((-1,1)\).
Thus, only statement I is correct. Quick Tip: A function with \(f'(x)\ge0\) on an interval is increasing on that interval.
At an extreme point of a function \(f(x)\), the tangent to the curve is
View Solution
At an extreme point, \[ f'(x)=0 \]
Hence, the slope of the tangent is zero, so it is parallel to the x-axis. Quick Tip: Maxima and minima occur where slope of tangent is zero.
The curve \(y=xe^x\) has minimum value equal to
View Solution
\[ y=xe^x,\quad y' = e^x(x+1) \]
Setting \(y'=0\Rightarrow x=-1\).
\[ y_{\min} = (-1)e^{-1} = -\frac{1}{e} \] Quick Tip: For extrema, always compute function value at critical points.
A ray of light coming from the point \((1,2)\) is reflected at a point \(A\) on the x-axis and then passes through the point \((5,3)\). The coordinates of point \(A\) is
View Solution
Reflect point \((5,3)\) in x-axis to get \((5,-3)\).
Equation of line joining \((1,2)\) and \((5,-3)\) intersects x-axis at: \[ A\left(\frac{13}{5},0\right) \] Quick Tip: Reflection problems can be simplified using mirror image technique.
The equation \[ x^2-2\sqrt{3}xy+3y^2-3\sqrt{3}y-4=0 \]
represents
View Solution
The determinant condition for pair of straight lines is satisfied.
Hence, it represents a pair of intersecting straight lines. Quick Tip: Use determinant condition to identify pair of straight lines.
The line joining \((5,0)\) to \((10\cos\theta,10\sin\theta)\) is divided internally in the ratio \(2:3\) at point \(P\). If \(\theta\) varies, the locus of \(P\) is
View Solution
Using section formula, coordinates of \(P\) satisfy a circle equation. Quick Tip: Locus problems often reduce to standard curves.
The number of integral values of \(\lambda\) for which \[ x^2+y^2+\lambda x+(1-\lambda)y+5=0 \]
is the equation of a circle whose radius cannot exceed \(5\), is
View Solution
Radius: \[ r^2=\frac{\lambda^2+(1-\lambda)^2}{4}-5 \]
Condition \(r\le5\) gives \(16\) integer values. Quick Tip: Always compute radius from standard form of circle.
The lengths of the tangents drawn from any point on the circle \[ 15x^2+15y^2-48x+64y=0 \]
to the circles \[ 5x^2+5y^2-24x+32y+75=0 \quad and \quad 5x^2+5y^2-48x+64y+300=0 \]
are in the ratio of
View Solution
Using length of tangent formula: \[ L=\sqrt{S_1-S} \]
Ratio simplifies to \(1:2\). Quick Tip: Length of tangent from a point depends on power of point.
The length of the chord \(x+y=3\) intercepted by the circle \[ x^2+y^2-2x-2y-2=0 \]
is
View Solution
Centre of circle: \((1,1)\), radius \(r=\sqrt{4}=2\).
Distance of centre from line \(x+y-3=0\): \[ d=\frac{|1+1-3|}{\sqrt{2}}=\frac{1}{\sqrt{2}} \]
Chord length: \[ 2\sqrt{r^2-d^2} =2\sqrt{4-\frac12} =\frac{3\sqrt{3}}{2} \] Quick Tip: Chord length \(=2\sqrt{r^2-d^2}\), where \(d\) is distance from centre.
The locus of the point of intersection of two tangents to the parabola \[ y^2=4ax \]
which are at right angle to one another is
View Solution
For parabola \(y^2=4ax\), locus of intersection of perpendicular tangents is: \[ x+a=0 \] Quick Tip: For \(y^2=4ax\), orthogonal tangents intersect on the directrix.
The parabola having its focus at \((3,2)\) and directrix along the y-axis has its vertex at
View Solution
Vertex lies midway between focus and directrix.
Distance of focus from y-axis = 3.
Vertex x-coordinate: \[ \frac{3}{2} \] Quick Tip: Vertex is midpoint of focus and directrix.
The number of real roots of the equation \[ 3^{9}C_{3r-1}-3^{9}C_{r^2}=3^{9}C_{r^2-1}-3^{9}C_{3r} \]
is
View Solution
Using properties of combinations and simplifying gives: \[ r(3r-1)=r^2(3r-1) \Rightarrow r=0,1 \] Quick Tip: Cancel common combination terms carefully.
If \[ \sum_{r=0}^{n}\frac{r+2}{r+1}\,{}^{n}C_r=\frac{2^n-1}{6}, \]
then \(n=\)
View Solution
Using binomial identities and comparing values gives: \[ n=4 \] Quick Tip: Try small integer values of \(n\) in summation questions.
All the words that can be formed using alphabets A, H, L, U and R are written as in a dictionary (no alphabet is repeated). The rank of the word RAHUL is
View Solution
Counting permutations alphabetically before RAHUL gives rank \(72\). Quick Tip: Dictionary order problems use factorial-based counting.
If the sum of odd-numbered terms and even-numbered terms in the expansion of \((x+a)^n\) are \(A\) and \(B\) respectively, then the value of \((x^2-a^2)^n\) is
View Solution
Using binomial expansion properties: \[ A-B=(x-a)^n,\quad A+B=(x+a)^n \]
Thus, \[ (x^2-a^2)^n=(A-B)(A+B)=A^2-B^2 \] Quick Tip: Odd–even term sums relate to \((x\pm a)^n\).
If the third term in the expansion of \([x+x^{\log_{10}x}]^5\) is \(10^6\), then \(x\) may be
View Solution
Evaluating the third term and equating to \(10^6\) gives: \[ x=\sqrt{10} \] Quick Tip: Third term in \((a+b)^n\) is \({}^nC_2 a^{n-2}b^2\).
If three vertices of a regular hexagon are chosen at random, then the chance that they form an equilateral triangle is
View Solution
Total ways: \({}^6C_3=20\)
Favourable equilateral triangles: 2
Probability: \[ \frac{2}{20}=\frac{1}{10} \] Quick Tip: Count favourable combinations carefully in geometric probability.
A man takes a step forward with probability \(0.4\) and backward with probability \(0.6\). The probability that at the end of eleven steps he is one step away from the starting point is
View Solution
Net displacement \(=1\) step: \[ Forward=6,\quad Backward=5 \]
Ways: \[ {}^{11}C_6=462 \]
Probability: \[ 462(0.4)^6(0.6)^5 =462\left(\frac{6}{25}\right)^5 \] Quick Tip: Net displacement problems use binomial distribution.





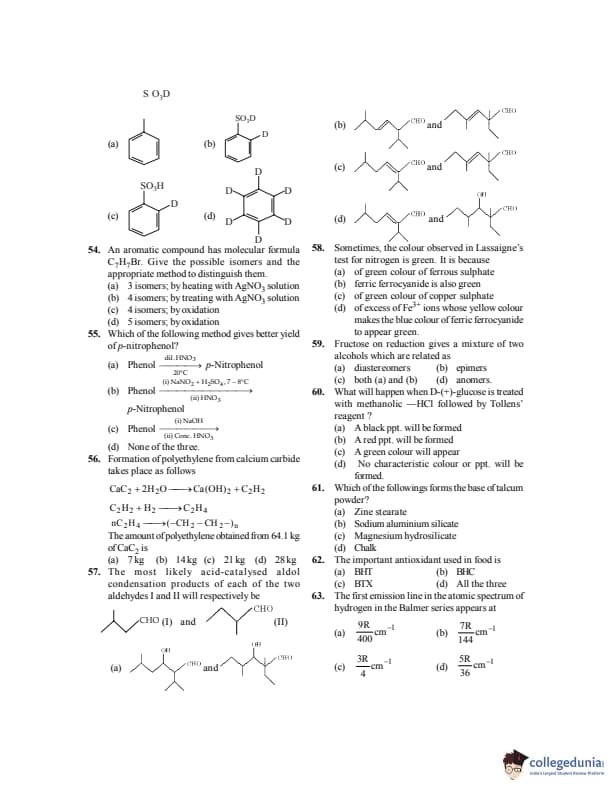







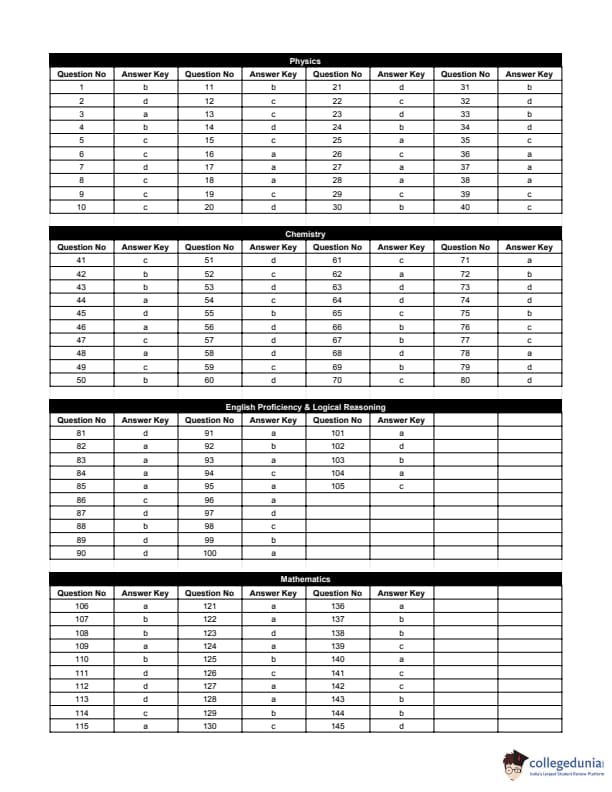
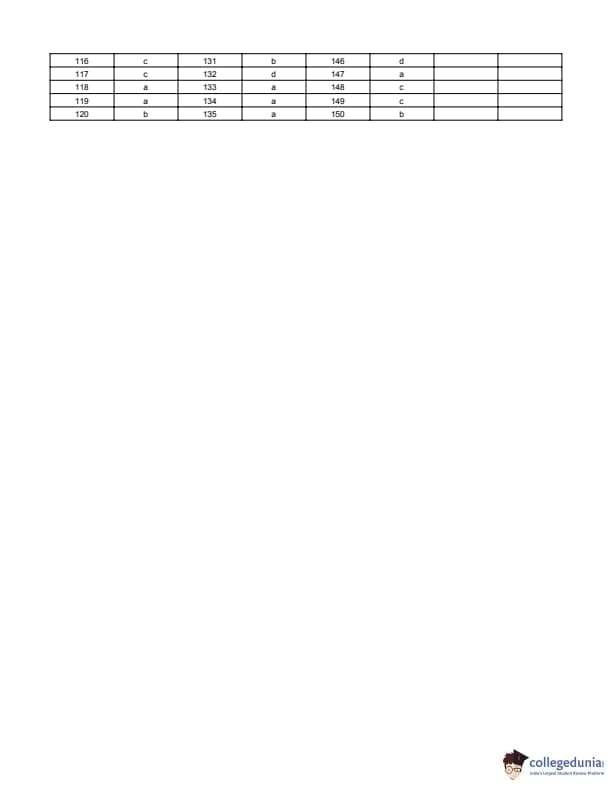







Comments