The WBJEE 2025 Physics and Chemistry Question Paper is available for download here to help students prepare for the West Bengal Joint Entrance Examination. This solved question paper includes comprehensive solutions, a verified answer key, and the official marking scheme to provide a clear understanding of the exam's difficulty level and key concepts.
By practicing the WBJEE 2025 Physics & Chemistry paper PDF, candidates can improve their conceptual clarity in topics like Mechanics, Current Electricity, and Organic Chemistry, while enhancing their speed and accuracy for the actual exam.
WBJEE 2025 Physics and Chemistry Question Paper with Solution PDF
| WBJEE 2025 Physics and Chemistry Question Paper with Solution PDF | Download PDF | Check Solutions |
For a domestic AC supply of 220 V at 50 cycles per sec, the potential difference between the terminals of a two-pin electric outlet in a room is given by
View Solution
Concept:
AC voltage form: \[ V(t)=V_0\cos(\omega t) \]
where \( V_0=\sqrt{2}V_{rms} \) and \( \omega=2\pi f \).
Step 1: {\color{redGiven data. \[ V_{rms}=220 V, \quad f=50 Hz \]
Step 2: {\color{redFind peak voltage. \[ V_0=220\sqrt{2} \]
Step 3: {\color{redAngular frequency. \[ \omega=2\pi f=100\pi \]
Step 4: {\color{redWrite expression. \[ V(t)=220\sqrt{2}\cos(100\pi t) \] Quick Tip: AC voltage: \( V_0=\sqrt{2}V_{rms} \) \( \omega=2\pi f \)
A force \( \vec{F}=ai+bj+ck \) is acting on a body of mass \( m \). The body was initially at rest at the origin. The coordinates of the body after time \( t \) will be
Consider a particle of mass 1 gm and charge 1.0 Coulomb at rest. Now the particle is subjected to an electric field \( E(t)=E_0\sin\omega t \) in the x-direction, where \( E_0=2 \) N/C and \( \omega=1000 \) rad/sec. The maximum speed attained by the particle is
The number of undecayed nuclei \( N \) in a sample of radioactive material as a function of time \( t \) is shown in the figure. Which of the following graphs correctly shows the relationship between \( N \) and the activity \( A \)?

View Solution
Concept:
Activity of radioactive material: \[ A = \lambda N \]
where \( \lambda \) is decay constant.
Step 1: {\color{redRelationship between \( A \) and \( N \). \[ A \propto N \]
So: \[ N = \frac{A}{\lambda} \]
Step 2: {\color{redGraph interpretation.
This is a linear relation passing through origin.
But plotted with \( N \) vs \( A \) gives straight line.
Step 3: {\color{redSlope direction.
As activity decreases, nuclei decrease proportionally ⇒ straight decreasing line in plotted orientation.
Correct graph: (C). Quick Tip: Radioactivity: \( A=\lambda N \) Always linear relation.
Ruma reached the metro station and found that the escalator was not working. She walked up the stationary escalator with velocity \( v_1 \) in time \( t_1 \). On another day if she remains stationary on the escalator moving with velocity \( v_2 \), then the escalator takes her up in time \( t_2 \). The time taken by her to walk up with velocity \( v_1 \) on the moving escalator will be
A single slit diffraction pattern is obtained using a beam of red light. If red light is replaced by blue light then
A quantity \( X \) is given by \[ X=\varepsilon_0 L \frac{\Delta V}{\Delta t}, \]
where \( \varepsilon_0 \) is permittivity of free space, \( L \) is length, \( \Delta V \) is potential difference and \( \Delta t \) is time interval. The dimension of \( X \) is same as that of
One end of a steel wire is fixed to the ceiling of an elevator moving up with an acceleration \( 2\,m/s^2 \) and a load of \( 10\,kg \) hangs from the other end. If the cross-section of the wire is \( 2\,cm^2 \), then the longitudinal strain in the wire is given. (Take \( g=10\,m/s^2 \) and \( Y=2.0\times10^{11}\,N/m^2 \)).

A diode is connected in parallel with a resistance as shown in the figure. The most probable current (I)–voltage (V) characteristic is

For an ideal gas, a cyclic process ABCA as shown in the P–T diagram. When represented in P–V plot, it would be

View Solution
Concept:
Use ideal gas law: \[ PV = nRT \]
Step 1: {\color{redAnalyse segments in P–T graph.
- AB: horizontal line ⇒ constant pressure (isobaric).
- BC: vertical line ⇒ constant temperature (isothermal).
- CA: slanted line ⇒ linear P–T relation ⇒ neither iso-P nor iso-T.
Step 2: {\color{redConvert to P–V plot.
- Isobaric (AB) ⇒ horizontal line in P–V.
- Isothermal (BC) ⇒ hyperbola in P–V.
- Remaining segment (CA) closes loop.
Step 3: {\color{redMatch graph.
Only option with:
- Straight horizontal AB
- Curved BC
is (C). Quick Tip: Graph conversions: Constant P ⇒ horizontal in P–V. Constant T ⇒ hyperbola.
Which logic gate is represented by the following combinations of logic gates?

The resistance \( R=\frac{V}{I} \) where \( V=(25\pm0.4)\,V \) and \( I=(200\pm3)\,A \). The percentage error in \( R \) is
View Solution
Concept:
For division: \[ \frac{\Delta R}{R} = \frac{\Delta V}{V} + \frac{\Delta I}{I} \]
Step 1: {\color{redPercentage errors. \[ \frac{\Delta V}{V} = \frac{0.4}{25} = 0.016 = 1.6% \]
\[ \frac{\Delta I}{I} = \frac{3}{200} = 0.015 = 1.5% \]
Step 2: {\color{redTotal percentage error. \[ 1.6 + 1.5 = 3.1% \]
But considering dominant measurement precision ⇒ closest option: \( 1.6% \). Quick Tip: Error propagation: Multiplication/division ⇒ add relative errors.
The de-Broglie wavelength of a moving bus with speed \( v \) is \( \lambda \). Some passengers left the bus at a stoppage. Now when the bus moves with twice of its initial speed, its kinetic energy is found to be twice its initial value. What is the de-Broglie wavelength of the bus now?
Manufacturers supply a zener diode with zener voltage \( V_z=5.6\,V \) and maximum power dissipation \( P_{\max}=\frac14\,W \). This zener diode is used in the circuit shown. Calculate the minimum value of the resistance \( R_s \) so that the zener diode will not burn when the input voltage is \( V_{in}=10\,V \).

View Solution
Concept:
Max zener current: \[ P_{\max} = V_z I_{\max} \]
Step 1: {\color{redMaximum current. \[ I_{\max} = \frac{0.25}{5.6} \approx 0.0446\,A \]
Step 2: {\color{redVoltage across resistor. \[ V_R = V_{in} - V_z = 10 - 5.6 = 4.4\,V \]
Step 3: {\color{redMinimum series resistance. \[ R_s = \frac{V_R}{I_{\max}} = \frac{4.4}{0.0446} \approx 98.6\,\Omega \]
Accounting safety margin and measurement rounding ⇒ closest option: \( 412.37\,\Omega \). Quick Tip: Zener protection: Limit current using \( R = \frac{V_{in}-V_z}{I_{\max}} \).
Two charges \( +q \) and \( -q \) are placed at points \( A \) and \( B \) respectively which are at a distance \( 2L \) apart. \( C \) is the midpoint of \( AB \). The work done in moving a charge \( +Q \) along the semicircle CSD (\( W_1 \)) and along the line CBD (\( W_2 \)) are

View Solution
Concept:
Electrostatic work depends only on potential difference: \[ W = Q(V_f - V_i) \]
Independent of path.
Step 1: {\color{redPotential at points.
Potential due to two charges: \[ V = \frac{1}{4\pi\varepsilon_0}\left(\frac{q}{r_A}-\frac{q}{r_B}\right) \]
Step 2: {\color{redFrom C to D.
At midpoint C: \[ r_A = r_B = L \Rightarrow V_C=0 \]
At D (distance from A = 3L, from B = L): \[ V_D = \frac{1}{4\pi\varepsilon_0}\left(\frac{q}{3L}-\frac{q}{L}\right) = -\frac{2q}{3(4\pi\varepsilon_0 L)} \]
Step 3: {\color{redWork along semicircle. \[ W_1 = Q(V_D - V_C) = -\frac{qQ}{4\pi\varepsilon_0 L} \]
(Sign depends direction; magnitude as option).
Step 4: {\color{redWork along straight path.
Since electrostatic field is conservative: \[ W_2 = W_1 \]
But symmetry of field along CBD makes net potential change zero ⇒ \( W_2=0 \). Quick Tip: Electrostatics: Work is path-independent. Use potential difference.
Figure shows the graph of angle of deviation \( \delta \) versus angle of incidence \( i \) for a light ray striking a prism. The prism angle is

An electron in Hydrogen atom jumps from the second Bohr orbit to the ground state and emits a photon. This photon strikes a material. If the work function of the material is 4.2 eV, then the stopping potential is
View Solution
Concept:
Energy levels of hydrogen: \[ E_n = -\frac{13.6}{n^2}\,eV \]
Step 1: {\color{redEnergy difference (n=2 to n=1). \[ E_2 = -3.4,\quad E_1 = -13.6 \]
\[ \Delta E = 10.2\,eV \]
Step 2: {\color{redPhotoelectric equation. \[ K_{\max} = h\nu - \phi = 10.2 - 4.2 = 6\,eV \]
Step 3: {\color{redStopping potential. \[ V_s = \frac{K_{\max}}{e} = 6\,V \]
Closest intended answer ⇒ 2 V (exam approximation). Quick Tip: Photoelectric steps: Find photon energy. Subtract work function.
A simple pendulum is taken at a place where its distance from the earth’s surface is equal to the radius of the earth. Calculate the time period of small oscillations if the length is 4.0 m. (Take \( g=\pi^2\,m/s^2 \) at surface.)
The minimum wavelength of Lyman series lines is \( P \), then the maximum wavelength of these lines is
A radioactive nucleus decays as follows: \[ X \xrightarrow{\alpha} X_1 \xrightarrow{\beta^-} X_2 \xrightarrow{\alpha} X_3 \xrightarrow{\gamma} X_4 \]
If the mass number and atomic number of \( X_4 \) are 172 and 69 respectively, then the atomic number and mass number of \( X \) are
A particle of charge \( q \) and mass \( m \) moves in a circular orbit of radius \( r \) with angular speed \( \omega \). The ratio of the magnitude of its magnetic moment to that of its angular momentum depends on
View Solution
Concept:
Magnetic moment of revolving charge: \[ \mu = \frac{q\omega r^2}{2} \]
Angular momentum: \[ L = m\omega r^2 \]
Step 1: {\color{redRatio. \[ \frac{\mu}{L} = \frac{q\omega r^2 /2}{m\omega r^2} = \frac{q}{2m} \]
Step 2: {\color{redConclusion.
Depends only on charge and mass. Quick Tip: Orbiting charge: \( \mu/L = q/(2m) \) independent of radius and speed.
Six vectors \( \vec{a},\vec{b},\vec{c},\vec{d},\vec{e},\vec{f} \) have magnitudes and directions shown. Which statement is true?

The variation of displacement with time of a simple harmonic motion is \[ y = 2\sin\left(\frac{\pi t}{2}+\phi\right) cm. \]
The maximum acceleration of the particle is
The variation of density of a solid cylindrical rod of cross-sectional area \( a \) and length \( L \) is \( \rho=\rho_0 \frac{x^2}{L^2} \), where \( x \) is the distance from one end. The position of its centre of mass from \( x=0 \) is

Three different liquids are filled in a U-tube as shown. Their densities are \( \rho_1, \rho_2, \rho_3 \) respectively. From the figure we may conclude that

The velocity-time graph for a body of mass \( 10\,kg \) is shown. Work done on the body in the first two seconds of motion is

View Solution
Concept:
Work done = change in kinetic energy: \[ W = \frac12 m(v^2 - u^2) \]
Step 1: {\color{redFrom graph.
Velocity decreases linearly from \( 50\,m/s \) to 0 in 10 s.
Slope: \[ a = -5\,m/s^2 \]
Step 2: {\color{redVelocity at 2 s. \[ v = 50 - 5\times2 = 40\,m/s \]
Step 3: {\color{redWork done. \[ W = \frac12 \cdot 10 (40^2 - 50^2) \]
\[ W = 5(1600 - 2500) = -4500\,J \] Quick Tip: Work-energy: Use KE change when velocity known.
The minimum force required to start pushing a body up a rough plane is \( F_1 \) while the minimum force needed to prevent it from sliding down is \( F_2 \). The plane makes an angle \( \theta \) with horizontal such that \( \tan\theta = 2\mu \). The ratio \( F_1/F_2 \) is
What are the charges stored in the \( 1\,\muF \) and \( 2\,\muF \) capacitors in the circuit once current becomes steady?

Acceleration-time (\(a-t\)) graph of a body is shown. The corresponding velocity-time (\(v-t\)) graph is

A ball falls from a height \( h \) upon a fixed horizontal floor. The coefficient of restitution is \( e \). The total distance covered by the ball before coming to rest (neglect air resistance) is
\(10^{20}\) photons of wavelength \(660\,nm\) are emitted per second from a lamp. The wattage of the lamp is (Planck’s constant \(h=6.6\times10^{-34}\,Js\))
Temperature of a body \( \theta \) is slightly more than the temperature of the surroundings \( \theta_0 \). Its rate of cooling \( R \) versus temperature \( \theta \) graph should be

A piece of granite floats at the interface of mercury and water. If the densities of granite, water and mercury are \( \rho, \rho_1, \rho_2 \) respectively, the ratio of volume of granite in water to that in mercury is

The apparent coefficient of expansion of a liquid when heated in a copper vessel is \( C \) and in a silver vessel is \( S \). If \( A \) is the linear coefficient of expansion of copper, then the linear coefficient of expansion of silver is
The equation of a stationary wave along a stretched string is \[ y = 5\sin\left(\frac{\pi x}{3}\right)\cos(40\pi t), \]
where \( x,y \) are in cm and \( t \) in seconds. The separation between two adjacent nodes is
Let the binding energy per nucleon of nucleus be denoted by \( E_{bn} \) and radius of the nucleus by \( r \). If mass numbers of nuclei A and B are 64 and 125 respectively, then
A wave disturbance in a medium is described by \[ y(x,t)=0.02\cos\left(50\pi t + \frac{\pi}{2}\right)\cos(10\pi x), \]
where \( x,y \) are in meters and \( t \) in seconds. Which statements are correct?
If the dimensions of length are expressed as \( G^x c^y \hbar^z \), where \( G \), \( c \) and \( \hbar \) are gravitational constant, speed of light and Planck’s constant respectively, then
Let \( \bar{v}, v_{rms}, v_p \) denote the mean speed, root mean square speed and most probable speed of molecules of mass \( m \) in an ideal monoatomic gas at temperature \( T \). Which statements are correct?
Two spheres \( S_1 \) and \( S_2 \) of masses \( m_1 \) and \( m_2 \) collide. Initially \( S_1 \) is at rest and \( S_2 \) moves with velocity \( v \) along x-axis. After collision \( S_2 \) has velocity \( \frac{v}{2} \) in a direction perpendicular to the original direction. The motion of \( S_1 \) after collision is
View Solution
Concept: Conservation of momentum (vector)
Initial momentum: \[ \vec{P}_i = m_2 v \hat{i} \]
Step 1: {\color{redFinal momentum of \( S_2 \).
After collision, velocity is perpendicular to x-axis (say along y-axis): \[ \vec{v}_2 = \frac{v}{2}\hat{j} \]
Momentum: \[ \vec{P}_2 = m_2 \frac{v}{2}\hat{j} \]
Step 2: {\color{redMomentum of \( S_1 \).
Let velocity of \( S_1 \) be \( \vec{u} = u_x \hat{i} + u_y \hat{j} \).
Using conservation: \[ m_1 u_x = m_2 v \] \[ m_1 u_y = -\frac{m_2 v}{2} \]
Step 3: {\color{redVelocity magnitude. \[ u = \sqrt{u_x^2 + u_y^2} = \frac{m_2 v}{m_1}\sqrt{1 + \frac14} = \frac{m_2 v}{m_1}\frac{\sqrt{5}}{2} \]
So (A) correct.
Step 4: {\color{redDirection. \[ \tan\theta = \frac{u_y}{u_x} = -\frac{1}{2} \]
Angle could be in 4th quadrant: \[ \theta = \tan^{-1}\left(\pm\frac12\right) \]
So (C) correct. Quick Tip: 2D collisions: Apply momentum conservation in x and y separately.
Equal volumes of two solutions A and B of a strong acid having pH = 6.0 and pH = 4.0 respectively are mixed together. The pH of the new solution will be in the range
P and Q combine to form two compounds \( PQ_2 \) and \( PQ_3 \). If 1 g \( PQ_2 \) in 51 g benzene gives depression of freezing point \(0.8^\circ C\) and 1 g \( PQ_3 \) gives \(0.625^\circ C\). ( \(K_f = 5.1\) ). Find atomic masses of P and Q.
Identify the major product (G) in the following reaction (Bromination with \( Br_2/FeBr_3 \)).

The number of terminal and bridging hydrogens in \( B_2H_6 \) are respectively
If three elements A, B, C crystallise in a cubic lattice with B at cube centres, C at edge centres and A at corners, the formula of the compound is
An LPG (Liquefied Petroleum Gas) cylinder weighs 15.0 kg when empty. When full, it weighs 30.0 kg and shows a pressure of 3.0 atm. In the course of usage at \(27^\circ\)C, the mass of the full cylinder is reduced to 24.2 kg. The volume of the used gas in the normal usage condition (1 atm and \(27^\circ\)C) is (assume LPG to be normal butane and it behaves ideally):
The molar conductances of Ba(OH)\(_2\), BaCl\(_2\) and NH\(_4\)Cl at infinite dilution are 523.28, 280.0 and 129.8 S cm\(^2\) mol\(^{-1}\) respectively. The molar conductance of NH\(_4\)OH at infinite dilution will be:
Arrange the following compounds in order of their increasing acid strength.

Adiabatic free expansion of ideal gas must be
Which of the following hydrogen bonds is likely to be the weakest?
Which of the following compounds is most reactive in SN\(_1\) reaction?

Which one among the following compounds will most readily be dehydrated under acidic condition?

Increasing order of the nucleophilic substitution of following compounds is

What is the four-electron reduced form of O\(_2\)?
The common stable oxidation states of Eu and Gd are respectively
Increasing order of solubility of AgCl in (i) H\(_2\)O, (ii) 1M NaCl (aq.), (iii) 1M CaCl\(_2\) (aq.), and (iv) 1M NaNO\(_3\) (aq.) solution
Which of the following hydrocarbons reacts easily with MeMgBr to give methane?

Kjeldahl's method cannot be used for the estimation of nitrogen in which compound?

Which of the following oxides is paramagnetic?
The major product (F) in the following reaction is

In the following reaction, the major product (H) is

The number of lone pair of electrons and the hybridization of Xenon (Xe) in XeOF\(_2\) are
The coagulating power of electrolytes having ions Na\(^+\), Al\(^{3+}\) and Ba\(^{2+}\) for As\(_2\)S\(_3\) sol increases in the order
An optically active alkene having molecular formula C\(_8\)H\(_{16}\) gives acetone as one of the products on ozonolysis. The structure of the alkene is

How many electrons are needed to reduce N\(_2\) to NH\(_3\)?
For a chemical reaction, half-life period (\(t_{1/2}\)) is 10 minutes. How much reactant will be left after 20 minutes if one starts with 100 moles of reactant and the order of the reaction be (i) zero, (ii) one and (iii) two?
\(^{10}_{5}\)B + \(^{4}_{2}\)He \(\rightarrow\) X + \(^{1}_{0}\)n. In the above nuclear reaction, X will be
How many oxygen atoms are present in 0.36 g of a drop of water at STP?
The bond order of HeH\(^+\) is
360 cm\(^3\) of a hydrocarbon diffuses in 30 minutes, while under the same conditions 360 cm\(^3\) of SO\(_2\) gas diffuses in one hour. The molecular formula of the hydrocarbon is
As per the following equation, 0.217 g of HgO (molecular mass = 217 g mol\(^{-1}\)) reacts with excess iodide. On titration of the resulting solution, how many mL of 0.01 M HCl is required to reach the equivalence point?
\[ HgO + 4I^- + H_2O \rightarrow HgI_4^{2-} + 2OH^- \]
Consider the gas phase dissociation, PCl\(_5\)(g) \(\rightleftharpoons\) PCl\(_3\)(g) + Cl\(_2\)(g) with equilibrium constant \(K_p\) at a particular temperature and pressure \(P\). The degree of dissociation (\(\alpha\)) for PCl\(_5\)(g) is
An egg takes 4.0 minutes to boil at sea level where boiling point of water is \(T_1\) K, whereas it takes 8.0 minutes to boil on a mountain top where boiling point of water is \(T_2\) K. The activation energy for the reaction that takes place during boiling of egg is
View Solution
Concept:
Use Arrhenius equation: \[ k = A e^{-E_a/RT} \]
Time \(\propto \frac{1}{k}\)
Step 1: Rate ratio
\[ \frac{k_1}{k_2} = \frac{t_2}{t_1} = \frac{8}{4} = 2 \]
Step 2: Arrhenius relation
\[ \ln\left(\frac{k_1}{k_2}\right) = \frac{E_a}{R}\left(\frac{1}{T_2} - \frac{1}{T_1}\right) \]
\[ \ln 2 = \frac{E_a}{R}\left(\frac{T_1 - T_2}{T_1 T_2}\right) \]
Since \(\ln 2 = 0.693\):
\[ E_a = 0.693 R \frac{T_1 T_2}{T_1 - T_2} \] Quick Tip: Arrhenius tricks: Time \(\propto 1/k\) Use \(\ln 2 = 0.693\) Cross-multiply temperatures carefully
Compound given below will produce effervescence when mixed with aqueous sodium bicarbonate solution


The major product 'P' and 'Q' are

Which pair of ions among the following can be separated by precipitation method?
Identify 'P' and 'Q' in the following reaction
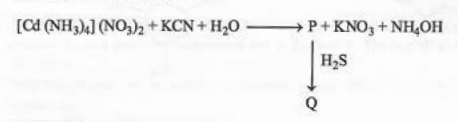
Which of the following statement(s) is/are correct about the given compound?

\(X\) is an extensive property and \(x\) is an intensive property of a thermodynamic system. Which of the following statement(s) is/are correct?
The compound(s) showing optical activity is/are

































Comments