CBSE Class 10 Science Question Paper 2026 Set-2 (Code: 31/5/2) is now available for download. CBSE conducted the Class 10 Science examination on Feb 25, 2026, from 10:30 AM to 1:30 PM. The question paper consists of 35 questions carrying a total of 80 marks. Part A is compulsory for all candidates. Part B has two options. Candidates have to attempt only one of the given options. Option I: Physics and Option II: Chemistry. The Science question paper 2026 was rated moderately easy by the students.
CBSE Class 10 Science Question Paper 2026 (Set 2- 31/5/2) with Answer Key
Candidates can use the link below to download the CBSE Class 10 Science 2026 Set 2 Question Paper with detailed solutions.
| CBSE Class 10 2026 Science Question Paper Set 2 with Solutions | Download PDF | Check Solution |

In human beings, the implantation of fertilised egg takes place in which part of female reproductive system?
View Solution
Concept:
Implantation is the process by which a fertilised egg (zygote) attaches itself to the wall of the female reproductive organ to begin development into an embryo.
Explanation:
Fertilisation occurs in the oviduct (fallopian tube).
The fertilised egg divides repeatedly to form a multicellular structure called a blastocyst.
This structure travels to the uterus.
It gets embedded in the thick, vascular lining of the uterus (endometrium).
Why Other Options Are Incorrect:
(A) Oviduct → Site of fertilisation, not implantation.
(B) Cervix → Lower part of uterus, not implantation site.
(D) Vagina → Passage for birth and intercourse.
Conclusion:
Implantation of the fertilised egg takes place in the uterus, where it attaches to the uterine lining and develops into an embryo. Quick Tip: Fertilisation → Oviduct. Implantation → Uterus.
Yeast multiplies by which of the following methods?
View Solution
Concept:
Yeast is a unicellular fungus that reproduces asexually by forming a small outgrowth from the parent cell. This process is called budding.
Explanation:
In budding, a small bulge or bud appears on the parent yeast cell.
The nucleus divides and one daughter nucleus moves into the bud.
The bud grows and eventually separates to form a new yeast cell.
Why Other Options Are Incorrect:
(A) Fragmentation → Seen in organisms like \textit{Spirogyra.
(B) Binary fission → Common in bacteria and amoeba.
(C) Multiple fission → Seen in organisms like \textit{Plasmodium.
Conclusion:
Yeast multiplies by budding, where a new individual develops as a small outgrowth from the parent cell. Quick Tip: Yeast reproduction = Budding. Small bud forms → Grows into new cell.
Identify the part that controls the closing and opening of the stomatal pore in leaves of plants. Select the correct option.
View Solution
Concept:
Stomata are tiny pores present on the surface of leaves that help in gas exchange and transpiration. The opening and closing of these pores are regulated by specialized cells.
Explanation:
Each stomatal pore is surrounded by two kidney-shaped cells called guard cells.
Guard cells control the opening and closing of stomata by changing their turgor pressure.
When guard cells absorb water, they swell and the pore opens.
When they lose water, they shrink and the pore closes.
Why Other Options Are Incorrect:
(A) Stomata → The pore itself, not the controlling structure.
(B) Epidermal cells → General surface cells, not specialized for control.
(D) Chloroplasts → Organelles for photosynthesis.
Conclusion:
Guard cells regulate the opening and closing of stomatal pores by controlling their turgor pressure. Quick Tip: Stomatal control = Guard cells. Turgor change → Pore opens or closes.
Choose the equation of reaction that correctly represents anaerobic respiration in muscles:
View Solution
Concept:
Anaerobic respiration is the breakdown of glucose in the absence of oxygen. In human muscle cells, this process leads to the formation of lactic acid.
Explanation:
During intense exercise, oxygen supply to muscles becomes insufficient.
Glucose is first converted into pyruvate.
In the absence of oxygen, pyruvate is converted into lactic acid with release of a small amount of energy.
Correct Reaction: \[ Glucose \rightarrow Pyruvate \xrightarrow{Lack of oxygen} Lactic acid + Energy \]
Why Other Options Are Incorrect:
(A) Ethanol formation occurs in yeast, not in human muscles.
(B) Represents aerobic respiration (presence of oxygen).
(C) Ethanol formation again applies to yeast fermentation.
Conclusion:
In human muscles, anaerobic respiration produces lactic acid and energy in the absence of oxygen. Quick Tip: Anaerobic respiration in muscles → Lactic acid formation. Yeast → Ethanol, Humans → Lactic acid.
Which of the following is a non-biodegradable pollutant?
View Solution
Concept:
Pollutants are substances that contaminate the environment. They can be biodegradable or non-biodegradable depending on whether microorganisms can decompose them.
Explanation:
Biodegradable pollutants can be broken down by microorganisms (e.g., paper, wood, vegetable peels).
Non-biodegradable pollutants cannot be decomposed easily and persist in the environment for a long time.
About DDT:
DDT (Dichloro-Diphenyl-Trichloroethane) is a synthetic pesticide.
It does not decompose naturally and accumulates in the food chain.
Causes biomagnification and environmental harm.
Conclusion:
DDT is a non-biodegradable pollutant because it does not break down naturally and persists in the environment. Quick Tip: Natural waste = Biodegradable. Chemicals like DDT = Non-biodegradable.
When a human egg is fertilized by a sperm having ‘Y’ chromosome, the zygote has the following combination of chromosomes:
View Solution
Concept:
Human body cells contain 46 chromosomes:
44 autosomes
2 sex chromosomes (XX or XY)
Explanation:
The egg always carries 22 autosomes + X chromosome.
A sperm can carry either:
22 + X → Female child (XX)
22 + Y → Male child (XY)
If fertilization occurs with a Y-bearing sperm:
\[ (22 + X) + (22 + Y) = 44 + XY \]
Conclusion:
If a sperm carrying a Y chromosome fertilizes the egg, the zygote will have 44 autosomes and XY sex chromosomes, resulting in a male child. Quick Tip: Egg always gives X. X sperm → XX (Female), Y sperm → XY (Male).
The reasons for excessive generation of wastes are:
(i) Use and throw policy.
(ii) Increased availability of packaged food.
(iii) Increased construction wastes.
(iv) Non-sorting of dry and wet wastes.
Options:
View Solution
Concept:
Excessive waste generation is mainly caused by modern lifestyle practices, urbanization, and improper waste management.
Explanation:
(i) Use and throw policy:
Increased use of disposable items like plastic and paper products leads to more waste.
(ii) Packaged food availability:
Packaged goods generate large amounts of plastic and non-biodegradable waste.
(iii) Construction waste:
Rapid urban development produces debris such as concrete, bricks, and metals.
(iv) Non-sorting of waste:
Mixing biodegradable and non-biodegradable waste increases landfill load and prevents recycling.
Conclusion:
All the listed factors contribute to excessive waste generation, making option (C) correct. Quick Tip: Modern lifestyle + Poor waste management = More waste generation.
Assertion (A): The peristaltic movements occur all along the gut.
Reason (R): The lining of the gut has muscles that contract rhythmically to push the food in regulated manner.
View Solution
Explanation:
Peristalsis is the rhythmic contraction and relaxation of muscles that pushes food along the digestive tract.
This movement occurs throughout the alimentary canal (gut).
The gut lining contains smooth muscles that contract rhythmically to move food forward.
Conclusion:
Both the assertion and reason are true, and the reason correctly explains the assertion. Quick Tip: Remember: Peristalsis = rhythmic muscle movement of the gut. If the reason mentions smooth muscle contractions pushing food forward, it correctly explains peristalsis.
Assertion (A): Bacteria produced as a result of asexual reproduction would be similar.
Reason (R): There would be only minor differences between the bacteria generated due to small inaccuracies in DNA replication.
View Solution
Explanation:
Asexual reproduction in bacteria produces genetically similar offspring.
However, small variations may occur due to minor errors during DNA replication.
These small inaccuracies explain why offspring are similar but not always identical.
Conclusion:
Both the assertion and reason are true, and the reason correctly explains the assertion. Quick Tip: Asexual reproduction → Similar offspring. Minor DNA errors → Small variations.
Observe the given figure and identify the labelled parts P, Q, R and S.

View Solution
Concept:
The given diagram represents a reflex arc. A reflex arc is the pathway followed by nerve impulses during a reflex action. It involves receptors, sensory neurons, the spinal cord, motor neurons, and effectors.
Identification of Labelled Parts:
P – Receptor (in skin)
Q – Sensory neuron
R – Spinal cord (relay neuron/interneuron)
S – Motor neuron
Explanation:
The receptor (P) detects the stimulus (hot object).
The sensory neuron (Q) carries the impulse to the spinal cord.
The spinal cord (R) processes the information through a relay neuron.
The motor neuron (S) carries the response to the muscles (effector). Quick Tip: Reflex arc pathway: Receptor → Sensory neuron → Spinal cord → Motor neuron → Effector.
Which of the plant hormones are responsible for the following processes?
(i) Promote cell division
(ii) Inhibition of growth
(iii) Detection of light
(iv) Wilting of leaves
View Solution
Concept:
Plant hormones (phytohormones) regulate growth, development, and responses to environmental stimuli.
Answers:
(i) Promote cell division → Cytokinins
(ii) Inhibition of growth → Abscisic acid (ABA)
(iii) Detection of light → Auxins
(iv) Wilting of leaves → Abscisic acid (ABA)
Explanation:
Cytokinins stimulate cell division in plants.
Abscisic acid acts as a growth inhibitor and induces dormancy and wilting.
Auxins help plants respond to light (phototropism).
ABA causes stomatal closure leading to wilting under stress conditions. Quick Tip: Cytokinin → Cell division. Auxin → Light response. ABA → Growth inhibition & wilting.
Briefly mention the steps in double-circulation through human heart.
View Solution
Concept:
Double circulation is the process in which blood passes through the heart twice during one complete cycle — once through pulmonary circulation and once through systemic circulation.
Steps in Double Circulation:
1. Pulmonary Circulation (Heart → Lungs → Heart):
Deoxygenated blood from the body enters the right atrium through the superior and inferior vena cava.
Blood flows from the right atrium to the right ventricle.
The right ventricle pumps blood to the lungs through the pulmonary artery.
In the lungs, carbon dioxide is exchanged for oxygen.
Oxygenated blood returns to the left atrium through the pulmonary veins.
2. Systemic Circulation (Heart → Body → Heart):
Oxygenated blood moves from the left atrium to the left ventricle.
The left ventricle pumps blood to the entire body through the aorta.
Body cells use oxygen and release carbon dioxide.
Deoxygenated blood returns to the right atrium through veins.
Conclusion:
In double circulation, blood flows through the heart twice in one complete cycle — ensuring efficient oxygen supply and separation of oxygenated and deoxygenated blood. Quick Tip: Double circulation = Pulmonary + Systemic circulation. Blood passes through heart twice in one cycle.
Given below is a diagrammatic representation of cross-section of a leaf:
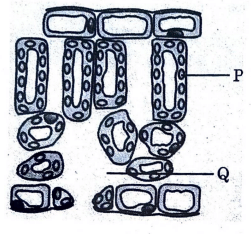
(i) Identify ‘P’ in the given diagram and write down its role in plants.
(ii) Write down a balanced equation of process of photo-synthesis.
View Solution
Concept:
A leaf cross-section shows different layers involved in photosynthesis, including epidermis, mesophyll cells, vascular bundles, and stomata.
(i) Identification of ‘P’ and its Role:
P = Palisade mesophyll
Role:
Palisade mesophyll cells contain a large number of chloroplasts.
They are the main site of photosynthesis.
These cells absorb maximum sunlight for food production.
(ii) Balanced Equation of Photosynthesis: \[ 6CO_2 + 6H_2O \xrightarrow{Sunlight, Chlorophyll} C_6H_{12}O_6 + 6O_2 \]
Conclusion:
Palisade mesophyll is responsible for photosynthesis in leaves, and the balanced equation shows conversion of carbon dioxide and water into glucose and oxygen using sunlight. Quick Tip: Palisade mesophyll = Main photosynthesis site. Photosynthesis: CO\(_2\) + H\(_2\)O → Glucose + O\(_2\) (sunlight).
Mention any one harmful effect of using plastic bags on the environment. Suggest better alternatives to the usage of plastic bags.
View Solution
Harmful Effect:
Plastic bags are non-biodegradable and remain in the environment for a long time, causing pollution and harm to animals and soil fertility.
Better Alternatives:
Use cloth or cotton bags.
Use jute or paper bags.
Reusable biodegradable bags.
Conclusion:
Replacing plastic bags with eco-friendly alternatives helps reduce pollution and protect the environment. Quick Tip: Plastic bags = Non-biodegradable. Use cloth or jute bags for eco-friendly living.
Paddy fields require a large amount of water. The pesticides and chemical fertilizers used are washed down into the soil or waterbodies. How do these chemicals reach our bodies? What is this phenomenon known as?
View Solution
Explanation:
Chemical fertilizers and pesticides enter water bodies and soil through runoff.
These chemicals are absorbed by plants or aquatic organisms.
When humans consume these plants or animals, the chemicals enter our bodies through the food chain.
Their concentration increases at higher trophic levels.
Name of the Phenomenon:
Biomagnification
Conclusion:
Harmful chemicals enter our bodies through the food chain, and their increasing concentration at successive trophic levels is called biomagnification. Quick Tip: Toxins increase along food chain = Biomagnification.
Mention the gland and the hormone secreted by it in scary situation in human beings.
View Solution
Gland: Adrenal gland
Hormone: Adrenaline (Epinephrine)
Explanation:
In frightening or emergency situations, the adrenal glands release adrenaline.
This prepares the body for “fight or flight” response. Quick Tip: Fear or stress → Adrenal gland releases Adrenaline.
Write two responses that enable the human body to be ready to deal with such situation.
View Solution
Responses of the Body:
Increased heart rate and blood pressure.
Faster breathing rate to supply more oxygen to muscles.
Increased blood flow to muscles.
Pupils dilate for better vision.
(Any two responses are acceptable.)
Conclusion:
These physiological responses prepare the body to react quickly in dangerous or stressful situations. Quick Tip: Adrenaline response = Faster heart rate + Rapid breathing + Alert body.
Write the structure and function of Bowman’s capsule.
View Solution
Structure:
Bowman’s capsule is a cup-shaped, double-walled structure.
It surrounds the glomerulus in the nephron of the kidney.
It forms the initial part of the renal tubule.
Function:
It collects the filtrate formed by ultrafiltration of blood in the glomerulus.
It allows water and small solutes (like urea, salts, glucose) to pass while retaining blood cells and proteins. Quick Tip: Bowman’s capsule = Cup-shaped part of nephron that collects filtrate from glomerulus.
Write the structure and function of Bowman’s capsule.
View Solution
Structure:
Bowman’s capsule is a cup-shaped, double-walled structure.
It surrounds the glomerulus in the nephron of the kidney.
It forms the initial part of the renal tubule.
Function:
It collects the filtrate formed by ultrafiltration of blood in the glomerulus.
It allows water and small solutes (like urea, salts, glucose) to pass while retaining blood cells and proteins. Quick Tip: Bowman’s capsule = Cup-shaped part of nephron that collects filtrate from glomerulus.
What is excretion? Why is it necessary for any living organism?
View Solution
Definition:
Excretion is the process of removal of metabolic waste products and toxic substances from the body.
Necessity:
Removes harmful metabolic wastes like urea, carbon dioxide, and excess salts.
Prevents accumulation of toxic substances in the body.
Maintains internal balance (homeostasis).
Helps proper functioning of cells and organs. Quick Tip: Excretion removes metabolic wastes and maintains body balance.
State two similarities between lungs and kidneys.
View Solution
Similarities:
Both are excretory organs that remove waste products from the body.
Lungs remove carbon dioxide and water vapour, while kidneys remove urea and excess salts.
Both help maintain internal balance of the body. Quick Tip: Lungs and kidneys = Excretory organs helping maintain internal balance.
(i) Sugarcane does not produce seeds so name the process through which it will be able to reproduce.
View Solution
Process: Vegetative propagation
Explanation:
Sugarcane reproduces asexually through stem cuttings, where new plants grow from nodes of the stem. Quick Tip: Sugarcane reproduces by vegetative propagation using stem cuttings.
(ii) List any two advantages of this method.
View Solution
Advantages:
Produces genetically identical plants (true to parent).
Faster method of reproduction.
Useful for plants that do not produce viable seeds.
(Any two acceptable) Quick Tip: Vegetative propagation = Fast and produces identical plants.
(iii) Besides sugarcane, give two more examples of plants that reproduce by this method.
View Solution
Examples:
Potato
Ginger
Rose
Bryophyllum
(Any two acceptable) Quick Tip: Vegetative propagation examples: Potato, Ginger, Bryophyllum, Rose.
(iv) Why regeneration is not possible in all the types of animals?
View Solution
Explanation:
Regeneration requires specialized cells capable of dividing and differentiating.
Higher animals have complex body organization and specialized tissues.
Most cells in higher animals cannot divide and form whole organisms.
Conclusion:
Therefore, regeneration is limited to simpler organisms and not possible in all animals. Quick Tip: Complex body structure limits regeneration in higher animals.
(i) State the changes taking place in the following structures of flower post fertilization:
(a) Zygote
(b) Ovule
(c) Ovary
(d) Sepals
View Solution
Changes after Fertilization:
(a) Zygote → Develops into embryo.
(b) Ovule → Develops into seed.
(c) Ovary → Develops into fruit.
(d) Sepals → Usually wither and fall off (may persist in some plants). Quick Tip: Post-fertilization: Zygote→Embryo, Ovule→Seed, Ovary→Fruit.
(ii) Define germination.
View Solution
Definition:
Germination is the process by which a seed develops into a new plant under suitable conditions such as water, oxygen, and proper temperature. Quick Tip: Germination = Seed grows into new plant under suitable conditions.
Which of the following set of compounds does not belong to same homologous series?
View Solution
Concept:
Compounds in the same homologous series have the same functional group and general formula and differ by a –CH\(_2\) unit.
Explanation:
Alkanes follow the general formula C\(_n\)H\(_{2n+2}\).
C\(_5\)H\(_{12}\) is an alkane (fits the formula).
C\(_4\)H\(_8\) follows C\(_n\)H\(_{2n}\), which is the formula of alkenes.
Thus, they belong to different homologous series.
Conclusion:
C\(_4\)H\(_8\) and C\(_5\)H\(_{12}\) do not belong to the same homologous series. Quick Tip: Same homologous series → Same general formula. Alkanes: C\(_n\)H\(_{2n+2}\), Alkenes: C\(_n\)H\(_{2n}\).
Which of the following is an amphoteric oxide?
View Solution
Concept:
Amphoteric oxides are oxides that react with both acids and bases to form salt and water.
Explanation:
Na\(_2\)O and K\(_2\)O are basic oxides.
CO\(_2\) is an acidic oxide.
Al\(_2\)O\(_3\) reacts with both acids and bases, showing amphoteric nature.
Conclusion:
Aluminium oxide (Al\(_2\)O\(_3\)) is an amphoteric oxide. Quick Tip: Amphoteric oxides react with both acids and bases (e.g., Al\(_2\)O\(_3\), ZnO).
In the following diagram, if acetic acid of same concentration is taken in place of dilute sulphuric acid, then
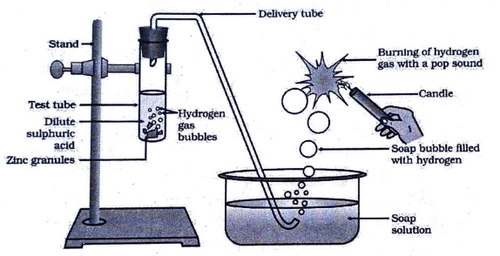
View Solution
Concept:
Metals react with acids to produce hydrogen gas. The rate and amount of hydrogen evolved depend on the strength of the acid.
Explanation:
Dilute sulphuric acid is a strong acid and ionizes completely in water.
Acetic acid is a weak acid and ionizes only partially.
Fewer H\(^+\) ions are available in acetic acid compared to sulphuric acid.
Hence, less hydrogen gas will be produced when acetic acid is used.
Conclusion:
Since acetic acid is a weak acid, the amount of hydrogen gas evolved will be less compared to dilute sulphuric acid. Quick Tip: Strong acid → More H\(_2\) gas. Weak acid (like acetic acid) → Less H\(_2\) gas.
The volume ratio of hydrogen and oxygen gases liberated during electrolysis of water is:
View Solution
Concept:
Electrolysis of water decomposes it into hydrogen and oxygen gases. The ratio of gases produced is based on the chemical equation.
Balanced Equation: \[ 2H_2O(l) \rightarrow 2H_2(g) + O_2(g) \]
Explanation:
2 molecules of hydrogen gas are produced for every 1 molecule of oxygen gas.
Hence, the volume ratio of hydrogen to oxygen is 2 : 1.
Conclusion:
During electrolysis of water, hydrogen and oxygen gases are produced in the ratio 2 : 1 by volume. Quick Tip: Electrolysis of water → H\(_2\) : O\(_2\) = 2 : 1.
Study the following table and select the correct option:
View Solution
Concept:
The nature of a salt depends on the strength of the acid and base from which it is formed.
Strong acid + Strong base → Neutral salt
Strong acid + Weak base → Acidic salt
Weak acid + Strong base → Basic salt
Analysis of Options:
(A) NaCl from HCl (strong acid) and NaOH (strong base) → Neutral, not basic ❌
(B) Na\(_2\)CO\(_3\) from weak acid (H\(_2\)CO\(_3\)) and strong base → Basic, not neutral ❌
(C) Na\(_2\)SO\(_4\) from strong acid and strong base → Neutral, not acidic ❌
(D) CH\(_3\)COONa from weak acid (CH\(_3\)COOH) and strong base (NaOH) → Basic ✔
Conclusion:
Sodium acetate (CH\(_3\)COONa) is formed from a weak acid and a strong base, making it a basic salt. Quick Tip: Weak acid + Strong base → Basic salt (e.g., CH\(_3\)COONa).
Which of the following will not undergo addition reaction?
View Solution
Concept:
Addition reactions occur in unsaturated hydrocarbons (alkenes and alkynes) that contain double or triple bonds. Saturated hydrocarbons (alkanes) do not undergo addition reactions.
Explanation:
C\(_4\)H\(_8\) → Alkene (unsaturated) ✔
C\(_2\)H\(_2\) → Alkyne (unsaturated) ✔
C\(_2\)H\(_4\) → Alkene (unsaturated) ✔
C\(_3\)H\(_8\) → Alkane (saturated) ✖
Conclusion:
C\(_3\)H\(_8\) is a saturated hydrocarbon and does not undergo addition reaction. Quick Tip: Addition reactions occur in unsaturated hydrocarbons, not alkanes.
On reaction with dilute HCl, which of the following pair of metals will evolve hydrogen gas?
View Solution
Concept:
Metals above hydrogen in the reactivity series react with dilute acids to produce hydrogen gas. Metals below hydrogen do not displace hydrogen from acids.
Explanation:
Copper and silver are below hydrogen → Do not evolve H\(_2\).
Zinc, iron, magnesium, and aluminium are above hydrogen → Evolve H\(_2\).
Only the pair where both metals react is magnesium and aluminium.
Conclusion:
Magnesium and aluminium react with dilute HCl and evolve hydrogen gas. Quick Tip: Metals above hydrogen in reactivity series evolve H\(_2\) with acids.
Assertion (A): Reaction of quick lime with water is an exothermic reaction.
Reason (R): A large amount of heat is evolved on the reaction of quick lime and water.
View Solution
Concept:
An exothermic reaction is one in which heat is released during the reaction.
Explanation:
Quicklime (CaO) reacts with water to form slaked lime (Ca(OH)\(_2\)).
\[ CaO + H_2O \rightarrow Ca(OH)_2 + Heat \]
A large amount of heat is released in this reaction.
Therefore, it is an exothermic reaction.
Conclusion:
Both the assertion and reason are true, and the reason correctly explains why the reaction is exothermic. Quick Tip: Quicklime + Water → Slaked lime + Heat (Exothermic reaction).
How is a universal indicator obtained? How is the wide range of pH of solution tested by it?
View Solution
Concept:
A universal indicator is a mixture of several indicators that shows different colours at different pH values. It is used to determine the pH of a solution over a wide range.
How Universal Indicator is Obtained:
It is prepared by mixing a number of indicators such as methyl orange, phenolphthalein, bromothymol blue, etc.
Each indicator works over a different pH range.
The combined mixture gives a gradual colour change across the full pH scale.
Testing Wide Range of pH using Universal Indicator:
A few drops of universal indicator are added to the solution.
The solution shows a specific colour depending on its pH.
The observed colour is compared with a standard pH colour chart.
This helps determine whether the solution is acidic, neutral, or basic and gives an approximate pH value (0–14).
Conclusion:
A universal indicator is made by mixing several indicators and is used to test a wide pH range by comparing the colour change with a standard pH chart. Quick Tip: Universal indicator = Mixture of indicators. Colour change + pH chart → Determine pH (0–14).
What happens when
(i) Calcium carbonate is heated?
(ii) Silver bromide is exposed to sunlight?
(iii) Lead is added to copper (II) chloride solution?
Write balanced chemical equations of the reactions involved in support of your answer.
View Solution
(i) Heating of Calcium Carbonate:
Observation:
Calcium carbonate decomposes into calcium oxide and carbon dioxide on heating.
Type of Reaction: Thermal decomposition reaction
\[ CaCO_3 (s) \xrightarrow{\Delta} CaO (s) + CO_2 (g) \]
(ii) Silver Bromide in Sunlight:
Observation:
Silver bromide decomposes into silver and bromine in presence of sunlight, turning grey.
Type of Reaction: Photochemical decomposition reaction
\[ 2AgBr (s) \xrightarrow{Sunlight} 2Ag (s) + Br_2 (g) \]
(iii) Lead added to Copper (II) Chloride Solution:
Observation:
Lead displaces copper from copper chloride solution, forming lead chloride and copper metal.
Type of Reaction: Displacement reaction
\[ Pb (s) + CuCl_2 (aq) \rightarrow PbCl_2 (s) + Cu (s) \]
Conclusion:
These reactions illustrate decomposition (thermal and photochemical) and displacement reactions with balanced chemical equations. Quick Tip: Heating → Thermal decomposition. Sunlight → Photochemical reaction. Reactive metal → Displacement reaction.
(a) Give the chemical name and formula of Plaster of Paris.
View Solution
Chemical Name: Calcium sulphate hemihydrate
Formula: CaSO\(_4 \cdot \frac{1}{2}\)H\(_2\)O Quick Tip: Plaster of Paris = Calcium sulphate hemihydrate.
(b) Write the chemical equation of its preparation.
View Solution
Plaster of Paris is prepared by heating gypsum.
\[ CaSO_4 \cdot 2H_2O \xrightarrow{100-120^\circ C} CaSO_4 \cdot \frac{1}{2}H_2O + \frac{3}{2}H_2O \] Quick Tip: Heating gypsum produces Plaster of Paris.
(c) Give any two uses of it.
View Solution
Uses of Plaster of Paris:
Making casts to support fractured bones.
Making statues, toys, and decorative materials.
Used in false ceilings and moulds.
(Any two acceptable) Quick Tip: POP uses: Medical casts and decorative items.
(a) Name the acid present in ant’s sting.
View Solution
Acid: Formic acid (Methanoic acid) Quick Tip: Ant sting contains formic acid.
(i) While diluting an acid, it is recommended that the acid should be added to water.
View Solution
Dilution of acid is highly exothermic. Adding acid slowly to water allows heat to dissipate safely and prevents splashing or explosion. Quick Tip: Always add acid to water to avoid splashing due to heat.
(ii) Baking soda is used as an antacid.
View Solution
Baking soda (NaHCO\(_3\)) is basic in nature and neutralizes excess stomach acid, providing relief from acidity and indigestion. Quick Tip: Baking soda neutralizes excess acid, so it works as an antacid.
Write structural formula and name of 4\(^{th}\) member of alcohol homologous series.
View Solution
Homologous Series: Alcohols follow the general formula C\(_n\)H\(_{2n+1}\)OH.
First four members:
1\(^{st}\) → Methanol (CH\(_3\)OH)
2\(^{nd}\) → Ethanol (C\(_2\)H\(_5\)OH)
3\(^{rd}\) → Propanol (C\(_3\)H\(_7\)OH)
4\(^{th}\) → Butanol (C\(_4\)H\(_9\)OH)
Name: Butanol
Structural Formula:
CH\(_3\)–CH\(_2\)–CH\(_2\)–CH\(_2\)OH Quick Tip: 4\(^{th}\) alcohol = Butanol (C\(_4\)H\(_9\)OH).
(b) What happens when ethanol is heated with alkaline KMnO\(_4\)? Write chemical equation involved.
View Solution
Observation:
Ethanol is oxidized to ethanoic acid by alkaline potassium permanganate.
Reaction: \[ CH_3CH_2OH + [O] \xrightarrow{KMnO_4} CH_3COOH + H_2O \]
Explanation:
KMnO\(_4\) acts as an oxidizing agent and converts alcohol into a carboxylic acid. Quick Tip: KMnO\(_4\) oxidizes ethanol → Ethanoic acid.
(c) Write the chemical equation of reaction of ethanol with ethanoic acid in the presence of concentrated H\(_2\)SO\(_4\). Write the name of this reaction.
View Solution
Reaction: \[ CH_3COOH + C_2H_5OH \xrightarrow{conc. H_2SO_4} CH_3COOC_2H_5 + H_2O \]
Name of Reaction: Esterification reaction
Explanation:
Ethanol reacts with ethanoic acid to form an ester (ethyl ethanoate) with a fruity smell. Quick Tip: Alcohol + Acid → Ester (Esterification).
(c) What happens when ethanol is heated with excess concentrated sulphuric acid at 443 K? Write chemical equation involved. What is the role of concentrated sulphuric acid in this reaction?
View Solution
Observation:
Ethanol gets dehydrated to form ethene.
Reaction: \[ C_2H_5OH \xrightarrow{443\,K,\, conc.\, H_2SO_4} C_2H_4 + H_2O \]
Role of Concentrated H\(_2\)SO\(_4\):
Acts as a dehydrating agent.
Removes water from ethanol to form ethene. Quick Tip: Conc. H\(_2\)SO\(_4\) at 443 K → Dehydration of ethanol to ethene.
(i) Give reasons for the following:
(I) Ionic compounds have generally high melting points and boiling points.
View Solution
Ionic compounds have strong electrostatic forces of attraction between oppositely charged ions. A large amount of energy is required to break these forces, resulting in high melting and boiling points. Quick Tip: Strong ionic bonds → High melting and boiling points.
(i) Give reasons for the following:
(I) Ionic compounds have generally high melting points and boiling points.
View Solution
Ionic compounds have strong electrostatic forces of attraction between oppositely charged ions. A large amount of energy is required to break these forces, resulting in high melting and boiling points. Quick Tip: Strong ionic bonds → High melting and boiling points.
(II) Solder, an alloy of lead and tin, is used for welding electrical wires.
View Solution
Solder has a low melting point and good electrical conductivity. It melts easily and solidifies quickly, forming a strong and conductive joint between wires. Quick Tip: Solder has low melting point and good conductivity, ideal for joining wires.
(III) Carbon cannot reduce the oxides of Na or Mg.
View Solution
Sodium and magnesium are highly reactive metals and lie above carbon in the reactivity series. Hence, carbon cannot reduce their oxides. Quick Tip: Carbon reduces oxides of less reactive metals only.
(III) Carbon cannot reduce the oxides of Na or Mg.
View Solution
Sodium and magnesium are highly reactive metals and lie above carbon in the reactivity series. Hence, carbon cannot reduce their oxides. Quick Tip: Carbon reduces oxides of less reactive metals only.
(ii) Give reasons for the following:
(I) The wires carrying current in homes have a coating of PVC.
View Solution
PVC is an electrical insulator. It prevents electric shocks and protects wires from moisture and damage.
(II) To make hot water tanks, copper is used and not steel.
Copper is less reactive and a better conductor of heat. It does not corrode easily, making it suitable for hot water tanks. Quick Tip: PVC = Insulator. Copper = Good conductor and corrosion resistant.
(iii) Show the formation of ionic compound CaO with electron dot structure.
View Solution
Atomic numbers: Ca = 20, O = 8
Electronic configuration:
Ca = 2, 8, 8, 2 (loses 2 electrons)
O = 2, 6 (gains 2 electrons)
Formation:
Calcium transfers two electrons to oxygen forming Ca\(^{2+}\) and O\(^{2-}\).
\[ Ca \rightarrow Ca^{2+} + 2e^- \] \[ O + 2e^- \rightarrow O^{2-} \]
Result: Ca\(^{2+}\) and O\(^{2-}\) combine to form CaO. Quick Tip: Ca loses 2e\(^-\), O gains 2e\(^-\) → Ionic bond forming CaO.
To restore clear vision in persons whose size of the eyeball has reduced, he/she is suggested to use suitable
View Solution
Concept:
A reduced eyeball size causes hypermetropia (long-sightedness), where distant objects are clear but nearby objects are blurred.
Explanation:
In hypermetropia, the image forms behind the retina.
A converging (convex) lens converges light rays before they enter the eye.
This helps form the image on the retina, restoring clear vision.
Conclusion:
A converging (convex) lens is used to correct vision when the eyeball size is reduced. Quick Tip: Hypermetropia → Use convex (converging) lens.
Rays from the sun converge at a point 25 cm behind a convex lens. The distance at which an object be placed in front of the lens to get a virtual image, is:
View Solution
Concept:
Parallel rays from the sun focus at the focal point of a convex lens.
So focal length, \(f = 25\) cm.
Key Rule:
A convex lens forms a virtual image only when the object is placed within the focal length (i.e., object distance \(<\) focal length).
Analysis of Options:
20 cm \(<\) 25 cm → Virtual image possible ✔
40 cm, 50 cm, more than 50 cm → Object beyond focal length → Real image ✖
Conclusion:
The object must be placed closer than the focal length, so 20 cm is correct. Quick Tip: Convex lens gives virtual image only when object is within focal length.
Assertion (A): The needle of a magnetic compass kept in strong external magnetic field, always aligns itself in north-south direction on the earth.
Reason (R): Behaviour of the needle of a compass is same as that of a freely suspended bar magnet.
View Solution
Explanation:
A compass needle aligns along the direction of the strongest magnetic field acting on it.
If a strong external magnetic field is present, the needle will align with that field, not necessarily in the Earth's north-south direction.
Hence, the assertion is false.
Reason Analysis:
A compass needle behaves like a small freely suspended bar magnet.
Therefore, the reason is true.
Conclusion:
Assertion is false but reason is true. Hence, option (D) is correct. Quick Tip: Compass aligns with strongest magnetic field present.
(a) Define absolute refractive index of an optical medium.
View Solution
The absolute refractive index of a medium is defined as the ratio of the speed of light in vacuum to the speed of light in that medium.
\[ n = \frac{c}{v} \]
Where, \(c\) = speed of light in vacuum \(v\) = speed of light in the medium Quick Tip: Absolute refractive index = Speed in vacuum ÷ Speed in medium.
(b) Arrange the given material mediums in increasing order of speed of light through them.
Given Refractive Indices:
A = 1.50
B = 1.46
C = 1.31
D = 1.77
View Solution
Concept:
Speed of light in a medium is inversely proportional to refractive index.
Higher refractive index → Lower speed of light.
Arrangement:
Highest refractive index to lowest (for increasing speed):
D (1.77) \(<\) A (1.50) \(<\) B (1.46) \(<\) C (1.31)
Answer:
D \(\rightarrow\) A \(\rightarrow\) B \(\rightarrow\) C Quick Tip: Higher refractive index → Slower light speed.
(i) How does the change in curvature of the eye lens help us in the process of seeing the nearby objects clearly?
View Solution
Explanation:
To see nearby objects, the ciliary muscles contract.
This increases the curvature (thickness) of the eye lens.
The focal length of the lens decreases.
As a result, the image of the nearby object is formed clearly on the retina. Quick Tip: Nearby object → Lens becomes thicker → Focal length decreases.
(ii) State the range of the power of accommodation of a normal human eye.
View Solution
The power of accommodation of a normal human eye is about 4 dioptres.
It enables the eye to adjust focus from infinity (distant vision) to about 25 cm (near point). Quick Tip: Normal eye power of accommodation ≈ 4 D.
Draw a ray diagram to show the correction of eye defect of an old man who cannot see an object placed closer than 1 m from his eye clearly.
View Solution
Defect Identified:
This condition is hypermetropia (long-sightedness), common in old age (presbyopia).
Correction:
A convex (converging) lens is used.
The convex lens converges light rays from a nearby object.
It helps form the image on the retina.
Ray Diagram Description:
Rays from a nearby object pass through a convex lens.
The convex lens bends rays to converge before entering the eye.
The eye lens then focuses them on the retina. Quick Tip: Cannot see near objects → Use convex lens (hypermetropia correction).
Why does an electric bulb become dim when an electric heater in parallel circuit is switched ON?
View Solution
Explanation:
When a heater is switched ON in parallel, the total current drawn from the source increases.
Due to internal resistance of the power supply or wiring, the voltage across each appliance may drop slightly.
As a result, less voltage is available across the bulb.
Reduced voltage decreases the brightness of the bulb, making it appear dim. Quick Tip: More appliances ON → More current → Voltage drop → Bulb dims.
(b) How to connect three resistors each of resistance 8 \(\Omega\), so that the equivalent resistance of the combination is 12 \(\Omega\)? Draw diagram of the combination and justify your answer.
View Solution
Required: Equivalent resistance = 12 \(\Omega\) using three 8 \(\Omega\) resistors.
Combination:
Connect two 8 \(\Omega\) resistors in parallel.
Then connect this parallel combination in series with the third 8 \(\Omega\) resistor.
Calculation:
Step 1: Parallel combination of two resistors \[ \frac{1}{R_p} = \frac{1}{8} + \frac{1}{8} = \frac{2}{8} = \frac{1}{4} \] \[ R_p = 4 \Omega \]
Step 2: Series with third resistor \[ R_{eq} = 4 + 8 = 12 \Omega \]
Diagram Description:
Two resistors connected side by side (parallel).
Their ends connected to a third resistor in series.
Conclusion:
Two resistors in parallel and one in series give the required equivalent resistance of 12 \(\Omega\). Quick Tip: Two equal resistors in parallel → Half resistance, then add series resistor.
An object is placed at a distance of 30 cm in front of a convex lens of focal length 15 cm. Use lens formula to determine the position of the image. What will be the size of the image in this case?
View Solution
Given:
Object distance, \(u = -30\) cm (in front of lens)
Focal length, \(f = +15\) cm (convex lens)
Lens Formula: \[ \frac{1}{f} = \frac{1}{v} - \frac{1}{u} \]
Substitute values: \[ \frac{1}{15} = \frac{1}{v} - \left(-\frac{1}{30}\right) \] \[ \frac{1}{15} = \frac{1}{v} + \frac{1}{30} \]
\[ \frac{1}{v} = \frac{1}{15} - \frac{1}{30} \]
\[ \frac{1}{v} = \frac{2 - 1}{30} = \frac{1}{30} \]
\[ v = +30 cm \]
Position of Image:
Image is formed 30 cm on the other side of the lens.
Size of Image:
Magnification, \[ m = \frac{v}{u} = \frac{30}{-30} = -1 \]
Magnitude of magnification = 1 → Image size = Object size
Negative sign → Image is inverted
Conclusion:
Image is formed at 30 cm on the other side of the lens.
The image is real, inverted, and equal in size to the object. Quick Tip: Object at 2f of convex lens → Image at 2f, same size, inverted.
(a) Draw the ray diagram for refraction of light through a glass prism and mark angle of refraction and angle of deviation.
View Solution
Ray Diagram Description:
A ray of light is incident on one face of the glass prism.
It bends towards the normal on entering the prism (refraction).
The refracted ray travels inside the prism and strikes the second face.
On emerging into air, it bends away from the normal.
Markings:
Angle of refraction (r): Angle between refracted ray and normal inside the prism.
Angle of deviation (\(\delta\)): Angle between the direction of incident ray and emergent ray. Quick Tip: Angle of deviation = Angle between incident and emergent rays.
(b) When the path of a light ray refracted through a glass prism is reversed, how will the angle of deviation change? Explain.
View Solution
Explanation:
Refraction of light through a prism obeys the principle of reversibility of light.
If the path of the light ray is reversed, it retraces the same path in the opposite direction.
Therefore, the angle of deviation remains unchanged.
Conclusion:
When the path of light is reversed through a prism, the angle of deviation remains the same. Quick Tip: Reversibility of light → Same path → Same angle of deviation.
Passage:
Three students Shweta, Ayesha and Samridhi were performing i an
experiment to understand the factors on which the resistance of a conductor depends. Each one of them completed electric circuit with the help of a cell, an ammeter, a plug key and wire.
Shweta put nichrome wire of length 7’ in the circuit and after plugging the key, noted current in the ammeter.
Ayesha put nichrome wire of same thickness but twice the length i.e. ‘21’ in the circuit and after plugging the key, noted current in the ammeter.
Samridhi took copper wire of length ‘I’ and same thickness in the circuit and after plugging the key, noted current in the ammeter.
(a) If the ammeter reading is X ampere with nichrome wire of length \(l\), then what will be the ammeter reading if the length of nichrome wire is doubled with same area of cross-section?
View Solution
Concept:
Resistance of a wire is directly proportional to its length. \[ R \propto l \]
If length is doubled → Resistance doubles.
From Ohm’s law, \[ I = \frac{V}{R} \]
So current is inversely proportional to resistance.
Result:
If resistance doubles, current becomes half.
\[ New current = \frac{X}{2} \] Quick Tip: Length ↑ → Resistance ↑ → Current ↓.
(b) What happens to the ammeter reading if the area of cross-section of nichrome wire is doubled, keeping the length of wire \(l\) the same?
View Solution
Concept:
Resistance is inversely proportional to area of cross-section. \[ R \propto \frac{1}{A} \]
If area is doubled → Resistance becomes half.
From Ohm’s law, \[ I = \frac{V}{R} \]
So current increases when resistance decreases.
Result:
If resistance becomes half, current doubles.
\[ New current = 2X \] Quick Tip: Area ↑ → Resistance ↓ → Current ↑.
(c) Define resistivity. Write its SI unit. Compare the resistivity of an alloy with its constituent metals.
View Solution
Definition:
Resistivity is a property of a material that measures how strongly it opposes the flow of electric current.
\[ \rho = \frac{RA}{l} \]
SI Unit: Ohm metre (\(\Omega\)m)
Comparison:
Alloys generally have higher resistivity than their constituent pure metals.
This is because impurities and irregular structure increase resistance to electron flow. Quick Tip: Alloys have higher resistivity than pure metals.
(i) Tungsten is used almost exclusively for making the filament of electric lamps.
View Solution
Tungsten has a very high melting point and can withstand high temperatures hookup without melting. It also has high resistivity and emits light when heated, making it ideal for lamp filaments.
(ii) Conductors of bread-toasters are made of an alloy rather than a pure metal.
Alloys have higher resistivity and do not oxidize easily at high temperatures. They can produce more heat and last longer than pure metals. Quick Tip: Tungsten → High melting point. Alloys → High resistivity and durability.
(i) Tungsten is used almost exclusively for making the filament of electric lamps.
View Solution
Tungsten has a very high melting point and can withstand high temperatures hookup without melting. It also has high resistivity and emits light when heated, making it ideal for lamp filaments.
(ii) Conductors of bread-toasters are made of an alloy rather than a pure metal.
Alloys have higher resistivity and do not oxidize easily at high temperatures. They can produce more heat and last longer than pure metals. Quick Tip: Tungsten → High melting point. Alloys → High resistivity and durability.
(i) List any two features of the electric current used in the houses of India.
View Solution
Features:
It is alternating current (AC).
Frequency is 50 Hz.
Voltage is about 220–230 V.
(Any two acceptable) Quick Tip: Domestic supply in India = AC, 220–230 V, 50 Hz.
(ii) Write any two differences between direct current and alternating current.
View Solution
\begin{tabular{|c|c|c|
\hline
Feature & Direct Current (DC) & Alternating Current (AC)
\hline
Direction & Flows in one direction & Changes direction periodically
\hline
Source & Cells, batteries & Power stations
\hline
Magnitude & Constant & Varies with time
\hline
\end{tabular
(Any two differences acceptable) Quick Tip: DC → One direction. AC → Changes direction periodically.
(iii) How will you identify live wire and neutral wire in a domestic electric circuit?
View Solution
Method: Using a tester (neon tester).
Connect the tester to the wire.
If the tester bulb glows → It is the live wire.
If the tester does not glow → It is the neutral wire. Quick Tip: Tester glows → Live wire. No glow → Neutral wire.
(ii) Distinguish between overloading and short-circuiting of a circuit.
View Solution
\begin{tabular{|c|c|c|
\hline
Feature & Overloading & Short-Circuiting
\hline
Cause & Too many appliances connected & Live and neutral wires come in contact
\hline
Current & Excess current drawn & Sudden very large current
\hline
Damage & Heating of wires & Sparks and fire risk
\hline
\end{tabular Quick Tip: Overloading → Too many devices. Short circuit → Live and neutral touch.
(iii) Giving reason explain what type of materials are used in fuse wires.
View Solution
Fuse wires are made of materials with:
Low melting point
High resistivity
Reason:
Such materials melt quickly when excess current flows, breaking the circuit and protecting appliances.
Example: Alloys of tin and lead. Quick Tip: Fuse wire → Low melting point alloy.






































Comments