AP EAPCET 2023 Question Paper May 22 Shift 2 is available for download here. AP EAPCET 2023 BiPC Question Paper May 22 Shift 2 consisted of four sections i.e. Botany, Zoology, Physics and Chemistry.
AP EAPCET (AP EAMCET) 2023 Question Paper included 160 MCQ-based questions in total, 40 from Botany, 40 from Zoology, 40 from Physics and 40 from chemistry. Each candidate will be awarded +1 for correct answers, however, there will be no negative marking for incorrect responses. Students got 3 hours to attempt AP EAPCET 2023 Question Paper.
Check AP EAPCET Exam Pattern 2023
AP EAPCET 2023 Question Paper May 22 Shift 2 PDF Download
| AP EAPCET 2023 Question Paper May 22 Shift 2 | Download PDF | Check Solution |

Question 1:
Bacteria that have specialized cell wall and plasma membrane structures to live and withstand extreme environmental conditions are called
View Solution
Archaebacteria, also known as Archaea, are a group of bacteria that have specialized cell wall and plasma membrane structures. These adaptations help them survive in extreme environmental conditions such as high temperatures, salinity, or acidity. While Eubacteria are also unique, they do not have the same level of adaptation to extreme environments as Archaebacteria.
Thus, the correct answer is Archaebacteria.
% Quicktip Quick Tip: Archaebacteria are known for their ability to survive in extreme environments, such as hot springs, deep-sea vents, and areas with high salinity.
A student has identified certain fungi given below and classified under this number of classes.
Mucor, Claviceps, Puccinia, Albugo, Neurospora
View Solution
Mucor, Claviceps, Puccinia, Albugo, and Neurospora are fungi classified under different classes in the kingdom Fungi. The given fungi belong to three major classes:
- Mucor is classified under Zygomycota.
- Claviceps is classified under Ascomycota.
- Puccinia and Albugo are classified under Basidiomycota.
- Neurospora is classified under Ascomycota.
Thus, these fungi belong to a total of 3 different classes.
% Quicktip Quick Tip: Fungi are classified into different classes based on their reproductive structures and lifecycle characteristics. Zygomycota, Ascomycota, and Basidiomycota are some of the main classes.
Choose the correct answer from the given statements below:
View Solution
- Option (1) is incorrect because cellular organization is essential for metabolism in living organisms.
- Option (2) is incorrect because non-living organisms do not show reproduction; only living organisms can reproduce.
- Option (3) is correct because in Amoeba, growth and reproduction are closely related, as the organism divides and grows during the process of binary fission.
- Option (4) is incorrect because irritability is the ability of an organism to respond to stimuli, not the response of the environment.
Thus, the correct answer is "Reproduction is synonymous with the growth in Amoeba."
% Quicktip Quick Tip: In unicellular organisms like Amoeba, growth and reproduction are closely linked through processes like binary fission.
The correct combinations among the following
I. Artificial gene synthesis - H.G. Khorana - Cell Biology
II. Pollen Science - P.K.K. Nair - Pollinology
III. Respiration - Amon - Physiology
IV. Phylogenetic classification - Bessey - Taxonomy
View Solution
Let's examine each combination:
- I: H.G. Khorana is known for Artificial gene synthesis and Cell Biology.
- II: P.K.K. Nair is associated with Pollen Science and Pollinology.
- III: Amon is not directly linked to respiration; this option is incorrect.
- IV: Bessey is known for Phylogenetic classification and Taxonomy.
Thus, the correct answer is "I II IV."
% Quicktip Quick Tip: The correct combinations of topics and their respective experts help understand the correct contributions in various biological fields.
Choose the correct statement related to the morphology of Cassia
View Solution
Cassia flowers exhibit imbricate aestivation, where the petals overlap in a particular way. The flowers are zygomorphic, meaning they are bilaterally symmetrical. Additionally, the inflorescence in Cassia is a corymb, which means that the flower stalks are arranged in a flat or slightly convex shape, with the outer flowers blooming first.
Thus, the correct statement is "Imbricate aestivation, zygomorphic, corymb."
% Quicktip Quick Tip: Imbricate aestivation is a form of flower arrangement where the petals overlap each other, while zygomorphic flowers are bilaterally symmetrical.
Choose the set of wrong combinations
A) Acacia - Phyllode - Petiole
B) Nepenthes - Insectivorous plant - Nitrogen requirement
C) Hibiscus - Two leaves at each nodes - Alternate
D) Neem - Number of leaflets attached at common point - Pinnately compound
View Solution
- A) Acacia: The leaf of Acacia is a phyllode, which is a flat, leaf-like structure. The petiole (leaf stalk) is absent, making this correct.
- B) Nepenthes: Nepenthes is an insectivorous plant that absorbs nitrogen from insects, making this correct.
- C) Hibiscus: Hibiscus has alternate leaves, so the statement "Two leaves at each node" is wrong because Hibiscus has one leaf at each node. Hence, this combination is incorrect.
- D) Neem: Neem has pinnately compound leaves where the leaflets are attached at common points along the rachis, making this correct.
Thus, the wrong combinations are C and D, so the correct answer is (2) C D.
% Quicktip Quick Tip: Phyllodes are modified leaf structures that perform the function of leaves in plants like Acacia, while some plants like Hibiscus have alternate leaves, not two leaves per node.
Arrange the following events in the correct sequence during the process of fertilization in Angiosperms:
I. Fusion of other male gamete with polar nuclei
II. Transfer of pollen grains from anther to stigma
III. Fusion of male gamete with egg nucleus
IV. Release of male gametes into cytoplasm synergid
V. Pollen tube formation
View Solution
The correct sequence of events during the fertilization process in Angiosperms is:
1. **II. Transfer of pollen grains from anther to stigma** - The pollen grain is transferred from the anther to the stigma.
2. **IV. Release of male gametes into cytoplasm of synergid** - The male gametes are released into the synergid cells of the ovule.
3. **V. Pollen tube formation** - The pollen tube forms to allow the male gametes to travel down to the egg.
4. **I. Fusion of other male gamete with polar nuclei** - One of the male gametes fuses with the polar nuclei to form the triploid endosperm.
5. **III. Fusion of male gamete with egg nucleus** - Finally, the second male gamete fuses with the egg nucleus to form the zygote.
Thus, the correct order is "II IV V I III."
% Quicktip Quick Tip: Fertilization in Angiosperms involves complex processes such as pollen tube formation, fusion with the egg, and the formation of endosperm, which are essential for seed development.
Match the following
List I - List II - List III
- A) Orthotropous - II) Sunflower - i) Straight to funiculus
- B) Complyotropous - I) Bean - iii) 90° to funiculus
- C) Anatropous - III) Polygonum - ii) 180° to funiculus
View Solution
- **Orthotropous**: The ovule is straight and attached directly to the funiculus, as in the case of **Polygonum**, which corresponds to "A III i."
- **Campylotropous**: The ovule is turned 180° to the funiculus, which corresponds to **Sunflower**, and thus "B II iii."
- **Anatropous**: The ovule is inverted, and the funiculus is attached to the concave side, as in **Bean**, corresponding to "C III ii."
Thus, the correct sequence is "A III i, B II iii, C III ii."
% Quicktip Quick Tip: In ovule types, the position of the ovule relative to the funiculus is crucial for plant reproductive biology. The types are based on how the ovule is attached and oriented.
The following plant is characterized by trimerous flowers, superior and trilocular ovary with axile placentation.
View Solution
Yucca gloriosa is known for having trimerous flowers, meaning that its flowers are arranged in groups of three. It has a superior ovary and trilocular structure with axile placentation. This characteristic is typical for certain monocot plants, and Yucca gloriosa fits this description.
% Quicktip Quick Tip: In plants with axile placentation, the ovules are attached to the central axis of the ovary, making it a key identifying feature.
The most abundant protein in the whole of the biosphere
View Solution
RuBisCO is the most abundant protein in the world. It is involved in the process of photosynthesis, specifically in the Calvin cycle, where it catalyzes the fixation of carbon dioxide into organic molecules. Its abundance is due to its widespread presence in plants, algae, and cyanobacteria, where it plays a crucial role in carbon fixation.
% Quicktip Quick Tip: Although collagen is abundant in animal tissues, RuBisCO far surpasses it in terms of total abundance due to its role in photosynthesis in plants.
In the chromatin the charge of a DNA and associated proteins, the number of types of histones and copies are
A. Positive, Negative
B. Negative, Positive
C. 4.2
D. 16.4
View Solution
In the chromatin, the charge of DNA is negative, and the associated histones have a positive charge. There are 4 types of histones, and each type has two copies (so there are 8 histone proteins associated with the DNA). This matches the description provided in option B, C.
% Quicktip Quick Tip: Histones play an essential role in DNA packaging, and their positive charge helps in the binding with negatively charged DNA.
Find the correct sub-phase based on the arrangement of chromosomes during M-phase.
M-phase -- The arrangement of chromosomes at different stages.
View Solution
During the Metaphase stage of the M-phase, chromosomes align at the equator of the cell, preparing for segregation in the next phase, Anaphase. The alignment at the equator is a hallmark of Metaphase.
% Quicktip Quick Tip: In Metaphase, chromosomes are highly condensed and aligned at the metaphase plate, making it easy to observe their structure under a microscope.
Number of base pairs in a full turn of the helical strand and the distance between base pairs in B-DNA.
View Solution
In B-DNA, a full turn of the helix consists of 10 base pairs and the distance between base pairs is approximately 34 Å. This structural feature is important for the stability of the double helix.
% Quicktip Quick Tip: The structure of B-DNA is right-handed and it makes a complete turn every 10 base pairs, with a helical pitch of 34 Å.
Match the following:
List I List II
(i) Well developed large pith (A) Endodermis
(ii) Casparian strips (B) Monocot stem
(iii) Sclerenchymatous bundle sheath (C) Dicot stem
(iv) Endarch protoxylem (D) Monocot root
Choose the Correct Option
1. I-B, II-A, III-D, IV-C
2. I-D, II-A, III-B, IV-C
3. I-D, II-C, III-A, IV-B
4. I-C, II-A, III-D, IV-B
View Solution
- Well developed large pith corresponds to Monocot root.
- Casparian strips are found in Endodermis.
- Sclerenchymatous bundle sheath is found in Dicot stem.
- Endarch protoxylem is found in Monocot stem.
These features are not characteristics of dicotyledonous root:
A. Radial vascular bundles.
B. Pith little or absent.
C. Vascular bundles 15 – 20.
D. Cambium ring do not develop.
View Solution
The features that are not characteristics of dicotyledonous roots include the vascular bundles being in the range of 15–20 and the failure to develop a cambium ring.
Thus, the correct answer is (C, D). Quick Tip: In dicotyledonous roots, the vascular bundles are typically fewer in number and a cambium ring is usually present.
Hydroponics was first demonstrated by
View Solution
Julius Von Sachs is known for first demonstrating hydroponics, the method of growing plants without soil, using only water-based nutrient solutions. Thus, the correct answer is (Julius Von Sachs). Quick Tip: Hydroponics was first successfully demonstrated by Julius Von Sachs in the 19th century.
Enzymes are denatured by
View Solution
Enzymes lose their activity or get denatured when exposed to extreme conditions such as high heat. Low pH and temperature may also affect enzymes, but heat is the most common cause for denaturation. Quick Tip: Enzymes are denatured by heat and extreme pH, leading to loss of function.
Choose incorrect statement related to diffusion
View Solution
Diffusion is a passive process and does not require energy expenditure. It occurs due to the concentration gradient. Thus, the statement that energy is spent for diffusion is incorrect. Quick Tip: Diffusion is a passive process that does not require energy; it relies on the concentration gradient.
Deficiency of the following causes Necrosis or death of leaf tissue
View Solution
Deficiency of calcium (Ca), magnesium (Mg), copper (Cu), and potassium (K) can cause necrosis or death of leaf tissue in plants. These nutrients are vital for cell function and plant health. Quick Tip: Necrosis due to nutrient deficiency often occurs in plants lacking calcium, magnesium, copper, or potassium.
'Transketolase' catalyses the following reaction
View Solution
Transketolase catalyzes the transfer of two-carbon units in the pentose phosphate pathway, and one of its reactions involves the conversion of G-3-P (glyceraldehyde-3-phosphate) with DHAP (dihydroxyacetone phosphate) to form erythrose-4-phosphate and DHAP. Quick Tip: Transketolase plays a key role in the pentose phosphate pathway by transferring two-carbon units.
Choose the correct combinations from the following
A. Induce sprouting of Potato tubers - Ethylene
B. Kill the dicot weeds - Cytokinin
C. Increase in stalk length of grapes - Gibberellins
D. Induce apical dominance - Auxins
View Solution
Ethylene is known to induce the sprouting of potato tubers, gibberellins help in increasing the stalk length of grapes, and auxins are responsible for inducing apical dominance. Cytokinins are used to kill dicot weeds. Quick Tip: Ethylene, gibberellins, and auxins each play distinct roles in plant growth and development.
Assertion (A): C\(_4\) plant loses only half of the water compared to C\(_3\) plant for the same amount of CO\(_2\) fixed.
Reason (R): C\(_4\) photosynthetic system is evolved for maximizing the availability of CO\(_2\) while minimizing water loss.
View Solution
C\(_4\) plants lose less water compared to C\(_3\) plants because their photosynthetic system is more efficient in capturing CO\(_2\) while minimizing water loss. This makes the assertion (A) true, and the reason (R) correctly explains the assertion. Quick Tip: C\(_4\) plants have an efficient mechanism that allows them to minimize water loss while still fixing carbon efficiently.
The ascending order of molecules of the following compounds released during the oxidation of two molecules of pyruvic acid:
FADH\(_2\), CO\(_2\), NADH\(_2\)
View Solution
The molecules released during the oxidation of pyruvic acid are CO\(_2\), NADH\(_2\), and FADH\(_2\), in this order. The correct sequence of their release is 2, 6, and 8. Quick Tip: During the oxidation of pyruvic acid, CO\(_2\), NADH\(_2\), and FADH\(_2\) are sequentially released.
The colour of chlorophyll 'b' in the chromatogram.
View Solution
Chlorophyll 'b' appears yellow-green in the chromatogram due to its absorption properties. Quick Tip: Chlorophyll 'b' is responsible for absorbing light and appears yellow-green in chromatograms.
The anatomical characteristic features of hydrophytes are
View Solution
Hydrophytes typically have epistomatous leaves, which are characterized by having stomata on the upper surface of the leaves. This is in contrast to plants with hypostomatous leaves, which have stomata on the lower surface. Quick Tip: Epistomatous leaves are a common feature of hydrophytes, helping them manage gas exchange efficiently.
Assertion (A): There is rising cost for pollinator-dependent fruits and vegetables.
Reason (R): Many pollinators are disrupted as they exist in isolation.
View Solution
As the assertion states, the cost for pollinator-dependent fruits and vegetables is rising due to the disruption of pollinators in isolation. The reason correctly explains that pollinators are disrupted due to their isolation, which in turn affects crop yields. Quick Tip: The disruption of pollinators has a significant impact on crop yields, leading to higher costs for pollinator-dependent crops.
The correct match among the following
I. Clostridium - Botulism
II. Diplococcus - Pneumonia
III. Treponema - Gonorrhoea
IV. Neisseria - Tetanus
View Solution
The correct pairings are:
- Clostridium causes Botulism.
- Diplococcus causes Pneumonia.
Quick Tip: Clostridium causes botulism, while Diplococcus is responsible for pneumonia.
Structure of bacteriophages is complicated because they show
View Solution
Bacteriophages show both helical and polyhedral symmetries, which complicates their structure. Hence, the correct options are III (Bilateral symmetry) and IV (Radial symmetry). Quick Tip: Bacteriophages exhibit complex symmetry, including both bilateral and radial symmetries.
The number of phenotypes and their genotypic ratio found when heterozygous round and yellow seeded pea plant crossed with other pea plant showing seeds with wrinkled and heterozygous yellow
View Solution
The cross between a heterozygous round and yellow seeded pea plant with another pea plant showing wrinkled and heterozygous yellow results in eight phenotypes, with the genotypic ratio of 1:2:1:2:1:2. Quick Tip: The number of phenotypes and genotypic ratio depend on the genetic factors involved in the cross.
The ratio of homozygous tall and dwarf plants in F2 generation in monohybrid cross is
View Solution
In a monohybrid cross involving homozygous tall and dwarf plants, the F2 generation would show a 1:1 ratio of homozygous tall to dwarf plants. This is due to the segregation of alleles. Quick Tip: In monohybrid crosses involving homozygous plants, the F2 generation will show a 1:1 ratio for traits controlled by a single gene.
Assertion (A): Cistron is a segment of DNA coding for a polypeptide.
Reason (R): Monocistronic structural genes are not splitted in Eukaryotes.
View Solution
The assertion is correct as cistron refers to a segment of DNA coding for a polypeptide. However, the reason is incorrect because monocistronic genes are present in eukaryotes and are not split. Quick Tip: Monocistronic genes are present in eukaryotes, and they are not split.
This statement is incorrect related to genetic code
View Solution
The statement "One codon codes one amino acid" is incorrect because some codons can code for more than one amino acid due to the redundancy of the genetic code. Quick Tip: The genetic code is degenerate, meaning some amino acids are specified by more than one codon.
Probes are
View Solution
Probes are gene-specific short sequences, single-stranded DNA/RNA fragments, and they are used in colony hybridization to identify specific DNA sequences. Quick Tip: Probes are essential in molecular biology to detect specific DNA or RNA sequences.
Choose the correct pair for artificially restructured E. coli cloning vector
I. pBR322 - Boliver and Rodrigues - Natural Vector
II. Selectable marker - Antibiotics - amp", terR
III. Restriction sites - Hind III, Pst I - Link alien DNA
IV. roP - Proteins - supports high copy number
View Solution
The correct pair is:
- II: Selectable marker – Antibiotics (amp\(^R\), tet\(^R\))
- III: Restriction sites – Hind III, Pst I – Link alien DNA
Quick Tip: E. coli cloning vectors often include selectable markers and restriction sites for inserting foreign DNA.
Study the below table and answer A, B, C
Rice varieties in India & A
Varieties of Basmati rice grown in India & B
Year of U.S Patent on Basmati rice & C
View Solution
Based on the table provided, the correct values for A, B, and C are:
- A: 200000 (Rice variety code)
- B: 27 (Varieties of Basmati rice grown in India)
- C: 1997 (Year of U.S. patent on Basmati rice)
Quick Tip: The U.S. patent on Basmati rice was granted in 1997, which played a key role in protecting this valuable crop.
Among the following, the wrong statement related to Bt cotton
View Solution
The wrong statement is (3) because protoxins are activated by alkaline pH, not acidic pH, in the gut of insects. Quick Tip: Bt toxins are activated in the insect gut by alkaline pH, which helps control pests.
Conversion of milk into curd by lactic acid bacteria increases
View Solution
Lactic acid bacteria used in the fermentation process of curd production can increase the vitamin B12 content of milk. Quick Tip: Lactic acid bacteria enhance the nutritional content of fermented dairy products, particularly vitamin B12.
Match the following
List I List II
I. Organogenesis A. Genetically identical
II. Somatic Embryo B. Bamboo cultivation
III. Micro propagation C. Production of shoots and roots
IV. Somaclones D. Structures from callus
View Solution
- Organogenesis is associated with the production of shoots and roots (C).
- Somatic embryos are structures derived from callus (D).
- Micropropagation leads to genetically identical plants (A).
- Somaclones are often used in bamboo cultivation (B). Quick Tip: In tissue culture, different processes like organogenesis and somatic embryogenesis are used to propagate plants with specific traits.
Assertion (A): Methanogens are present in the rumen of cattle.
Reason (R): They help in the breakdown of cellulose and nutrition of cattle.
View Solution
The assertion is correct because methanogens are indeed present in the rumen of cattle and help in breaking down cellulose. However, the reason provided (about nutrition of cattle) is not a correct explanation of the assertion, as methanogens help in the fermentation process but not directly in providing nutrition. Quick Tip: Methanogens are essential for the breakdown of cellulose in the rumen of cattle, aiding in the fermentation process.
Rats belong to the class
View Solution
Rats belong to the class Rodentia, which is known for small, gnawing mammals. Quick Tip: Rodents are characterized by a pair of constantly growing incisors in the upper and lower jaws.
Choose the incorrect pair among the following
A. Biosphere reserve - Indo Burma region
B. Aravalli Hills - Rajasthan and Gujarat
C. Koringa sanctuary - Uttarakhand
D. Biodiversity hotspot - Himalayan region
View Solution
The incorrect pair is A (Biosphere reserve - Indo Burma region) and C (Koringa sanctuary - Uttarakhand). Koringa sanctuary is actually located in Andhra Pradesh, not Uttarakhand. Quick Tip: Biosphere reserves and sanctuaries are important for conservation, but their locations must be accurate.
Assertion (A): Kidneys of vertebrates are called retroperitoneal organs.
Reason (R): They are covered by parietal peritoneum only on their ventral side.
View Solution
The assertion is correct because kidneys in vertebrates are indeed retroperitoneal organs, meaning they lie behind the peritoneum. The reason is also correct as they are only covered by the parietal peritoneum on their ventral side. Quick Tip: Retroperitoneal organs are located behind the peritoneum and are partially covered by it.
Assertion (A): Mast cells secrete serotonin.
Reason (R): Vasodilators cause inflammation in response to infection.
View Solution
Mast cells do indeed secrete serotonin, which is involved in inflammatory responses. However, the reason that vasodilators cause inflammation is not directly linked to the secretion of serotonin by mast cells, thus the reason does not fully explain the assertion. Quick Tip: Mast cells release various substances like serotonin and histamine, but vasodilation occurs due to other mechanisms such as the action of prostaglandins and bradykinin.
Hyaline cartilage occurs in
A. Sternal part of the ribs.
B. Pubic Symphysis.
C. Epiphyseal plate.
D. Vocal chords.
E. Epiglottis.
F. Endoskeleton of cyclostomes.
View Solution
Hyaline cartilage is found in the sternal part of the ribs (A), the epiphyseal plate (C), and the endoskeleton of cyclostomes (F). Quick Tip: Hyaline cartilage is the most common type of cartilage in the body and is found in areas like the ribs, joints, and the epiphyseal plates.
Choose the incorrect statements regarding Hirudinea
A. Male reproductive system has cirrus.
B. They are exclusively marine and tubicolous.
C. Fertilization is external and development is direct.
D. Clitellum is conspicuous during the breeding season only.
View Solution
The incorrect statements are:
- B: Hirudinea are not exclusively marine; they can also be found in freshwater.
- C: Fertilization in Hirudinea is internal and development is indirect.
Quick Tip: Hirudinea have internal fertilization, and their development includes a larval stage (indirect development).
The spermathecae in earthworm are present in
View Solution
In earthworms, spermathecae are present in segments 6, 7, 8, and 9, where sperm storage occurs. Quick Tip: The spermathecae are crucial for sperm storage and fertilization in earthworms.
Choose the correct combination among the following
A. Bivalvia - Radula - Rasping organ
B. Scaphopoda - Captacula - Capture of food
C. Pelecypoda - Crystalline style - Digestion of starch
D. Cephalopoda - Osphradium - Water purity testing
View Solution
The correct combination is:
- Scaphopoda has Captacula, which are used for capturing food.
- Pelecypoda has a crystalline style, which is involved in the digestion of starch.
Quick Tip: Different classes of mollusks have specialized structures for food capture and digestion.
The gene that is believed to play a key role in human language expression is
View Solution
The FOX P2 gene is considered to play a key role in language expression and speech development in humans. Quick Tip: FOX P2 is often called the "language gene" because of its significant role in speech and language abilities.
Assertion (A): Pneumaticity of bones is the unique feature of reptiles.
Reason (R): Air sacs associated with lungs of birds facilitate continuous oxygenation of blood.
View Solution
The assertion is incorrect because pneumaticity of bones is actually a feature of birds, not reptiles. The reason is correct because air sacs in birds help in continuous oxygenation of blood, but this does not explain the assertion. Quick Tip: Pneumatic bones are a characteristic of birds, aiding in flight, while reptiles do not have this feature.
Identify the structure found in the early embryonic stages of amniotes that provides a clue to their aquatic ancestry.
View Solution
Nonfunctional pharyngeal pouches are present in the early embryonic stages of amniotes and are believed to be a vestige of their aquatic ancestors, as they are involved in respiration in aquatic organisms. Quick Tip: Pharyngeal pouches, which were functional in early vertebrates, are now vestigial in terrestrial amniotes but provide evidence of their aquatic ancestry.
In microtubules, the peripheral doublets are interconnected by linkers called
View Solution
In the structure of microtubules, the peripheral doublets of the axoneme are interconnected by proteins known as nexins. Quick Tip: Nexins are proteins that link the microtubule doublets in the axoneme and are crucial for the stability and function of cilia and flagella.
Identify the correct statement in relation to Paramecium
View Solution
In Paramecium, the macronucleus is polyploid, while the micronucleus is diploid. The macronucleus controls the non-reproductive cell functions, while the micronucleus is involved in sexual reproduction. Quick Tip: In ciliates like Paramecium, the macronucleus controls everyday cell functions, while the micronucleus plays a role in genetic exchange during conjugation.
Mild tertian malaria is caused by
View Solution
Mild tertian malaria is caused by *Plasmodium vivax*, which is characterized by fever cycles occurring every 48 hours. Quick Tip: *Plasmodium vivax* is the most common cause of malaria and causes tertian fever, which recurs every 48 hours.
Assertion (A): Life cycle of ectoparasites is more complex with many developmental stages.
Reason (R): Developmental stages of endoparasites have better chances to reach definitive host.
View Solution
The assertion is correct that the life cycle of ectoparasites is more complex. However, the reason is incorrect, as developmental stages of endoparasites are usually more adapted to their host and tend to complete their life cycle more effectively within the host. Quick Tip: Ectoparasites like lice or fleas often have more complex life cycles because they must interact with both the host and external environments at various stages.
Among the following, nocturnal periodicity is exhibited by
View Solution
Microfilaria, which are the larval stage of filarial worms, exhibit nocturnal periodicity, meaning they are more prevalent in the blood during the night. Quick Tip: Microfilaria is the first stage in the life cycle of filarial worms and typically exhibits nocturnal behavior due to the feeding patterns of the mosquito vectors.
Assertion (A): Mary Mallon was nicknamed as 'Typhoid Mary'.
Reason (R): Mary Mallon spread typhoid for several years through the food she prepared.
View Solution
Mary Mallon, also known as 'Typhoid Mary', was a carrier of the typhoid fever bacteria and unknowingly spread it through the food she prepared for others. Quick Tip: Typhoid Mary is a famous case in public health history, where a healthy carrier of the disease caused widespread infection by preparing food.
Prochordate that exhibits bioluminescence
View Solution
Pyrosoma, a genus of colonial tunicates (prochordates), is known for exhibiting bioluminescence, where the organisms in the colony emit light. Quick Tip: Bioluminescence is common in marine animals like pyrosomes, and it is used for various purposes such as predation and communication.
The integral form of the exponential growth equation is
View Solution
The integral form of the exponential growth equation is \(N_t = N_0 e^{rt}\), where \(N_t\) is the population at time \(t\), \(N_0\) is the initial population, \(r\) is the rate of growth, and \(t\) is time. Quick Tip: The exponential growth equation is used to model populations growing at a constant rate without limiting factors.
Identify the correct option
Statement I: Haemoglobin binds with oxygen in oxygen-rich areas and leaves CO2.
Statement II: Haemoglobin is an amphoteric compound.
View Solution
Both statements are correct. Haemoglobin binds with oxygen in oxygen-rich areas and releases CO2. Additionally, it acts as an amphoteric compound, capable of acting as either an acid or a base. Quick Tip: Haemoglobin plays a crucial role in oxygen and CO2 transport in the blood. It also helps maintain pH balance by binding or releasing hydrogen ions.
Find out the correct match
Set I
A. Carbohydrates
B. Proteins
C. Fats
D. Nucleic acids
Set II
i. Nucleosides, Phosphates
ii. Glucose, Fructose, Galactose
iii. Amino acids
iv. Fatty acids, glycerols
View Solution
- Carbohydrates are represented by sugars like glucose, fructose, and galactose (ii).
- Proteins are made up of amino acids (iii).
- Fats consist of fatty acids and glycerols (iv).
- Nucleic acids consist of nucleosides and phosphates (i).
Quick Tip: Understanding the basic structures of macromolecules helps in identifying their components and corresponding groupings.
Sino-Atrial Node (SAN) is situated in
View Solution
The Sino-Atrial Node (SAN) is located in the upper right corner of the right atrium. It initiates the electrical impulses that regulate the heartbeat. Quick Tip: The Sino-Atrial Node is also referred to as the heart's natural pacemaker as it generates electrical impulses that control the heart rate.
Match the column I with Column II.
Column – I Column – II
A. Coronary artery disease i. The blood pressure above 140/90.
B. Hypertension ii. Accumulation of calcium, fat, cholesterol in coronary arteries.
C. Angina pectoris iii. Heart unable to pump sufficient blood.
D. Heart failure iv. Chest pain caused by Ischemia.
View Solution
- A: Coronary artery disease is caused by the accumulation of calcium, fat, cholesterol in coronary arteries (ii).
- B: Hypertension is defined by the blood pressure above 140/90 (i).
- C: Angina pectoris results in chest pain caused by ischemia (iv).
- D: Heart failure occurs when the heart is unable to pump sufficient blood (iii).
Quick Tip: High blood pressure, or hypertension, is a major risk factor for coronary artery disease and heart failure.
One of the following is not a part of light band of myofibril.
View Solution
The light band of myofibril contains the protein actin, which is responsible for muscle contraction. The other proteins like myosin, troponin, and tropomyosin are involved in muscle contraction but are not part of the light band. Myosin is part of the dark band (A band) in the myofibril. Quick Tip: The myofibrils consist of repeating units called sarcomeres, which are responsible for muscle contraction.
Arrange in sequence the following events related to the mechanism of hearing:
View Solution
The sequence of events involved in the mechanism of hearing is:
1. The eardrum vibrates in response to sound waves. (C)
2. The transmission of vibrations through the ear ossicles. (E)
3. The induction of a ripple in the basilar membrane. (B)
4. Hair cells bend and press against the Tectorial membrane. (A)
5. Impulses are transmitted to the brain. (D)
Hence, the correct sequence is C, E, B, A, D. Quick Tip: The correct sequence for the mechanism of hearing involves the eardrum vibrating first, followed by transmission through the ossicles, ripple induction in the basilar membrane, bending of hair cells, and transmission of impulses to the brain.
One of the following is not a second messenger in hormone action:
View Solution
In hormone action, second messengers such as cAMP, calcium, and IP3 play important roles in transmitting signals within the cell. Sodium, however, is not a second messenger and does not function in the same way as the others. Quick Tip: Second messengers such as cAMP, calcium, and IP3 are involved in the intracellular signaling pathways of hormones. Sodium is not one of them.
Identify the correct match of the gland with its respective hormone and its function.
View Solution
- The corpus luteum produces progesterone, not estrogen, and supports pregnancy.
- Thyroid gland secretes thyroxine, which regulates metabolism, not blood calcium levels.
- Oxytocin, from the anterior pituitary, aids in uterine contractions during labor.
- Vasopressin from the posterior pituitary regulates water balance by promoting reabsorption of water in the kidneys.
Hence, the correct match is Posterior pituitary – Vasopressin – Stimulates reabsorption of water from the Distal Convoluted Tubule. Quick Tip: Vasopressin (ADH) is key in regulating the body's water balance by promoting the reabsorption of water in the kidneys.
Find out the primary lymphoid organ from the following:
View Solution
The thymus gland is the primary lymphoid organ where T-lymphocytes (T-cells) mature and differentiate. It plays a critical role in the immune system, unlike the spleen, lymph nodes, and tonsils, which are secondary lymphoid organs involved in filtering lymph and providing immune responses. Quick Tip: Primary lymphoid organs such as the thymus and bone marrow are responsible for the production and maturation of immune cells, while secondary lymphoid organs like the spleen and lymph nodes are involved in immune responses.
Choose among the following that are cellular barriers of immunity:
View Solution
Cellular barriers of immunity include cells like neutrophils, macrophages, and monocytes. These cells play key roles in defending the body from pathogens. Cytokines also help in immune responses but are signaling molecules, not cellular barriers themselves. Saliva and lysozyme are involved in non-cellular immune barriers.
Hence, the correct answer is option (4) b, c, f. Quick Tip: Neutrophils, macrophages, and monocytes are white blood cells that are directly involved in immune defense and act as cellular barriers.
Identify the correct option:
Statement-I (S-I): Test tube baby undergoes development within the test tube.
Statement-II (S-II): In IVF-ET method, fertilization occurs inside the body.
View Solution
In the IVF process, fertilization occurs outside the body in a laboratory dish, not inside the body. After fertilization, the embryo is implanted into the uterus. Therefore, Statement-I is incorrect, and Statement-II is correct. Quick Tip: In vitro fertilization (IVF) involves fertilization outside the body, while the embryo is placed into the uterus for further development.
The notochordal mesoderm induces the overlying ectodermal cells that are involved in the formation of:
View Solution
The notochord induces the formation of the neural plate in the ectoderm. The neural plate then folds to form the neural groove, which later develops into the neural tube. Neural glands are not formed by the notochord. Quick Tip: The neural plate is a crucial step in neural development and gives rise to the neural tube, which forms the brain and spinal cord.
The double-layered fold of peritoneum that connects the ovary with the wall of the abdominal cavity is:
View Solution
The mesovarium is the fold of peritoneum that attaches the ovary to the broad ligament of the uterus and connects it to the wall of the abdominal cavity. The other options refer to different structures not related to the ovary. Quick Tip: The mesovarium is important in anchoring the ovary within the abdominal cavity and supports the blood vessels that supply the ovary.
The universal donor blood group is:
View Solution
The universal donor blood group is O-, as it does not have any antigens (A, B, or Rh) on the surface of the red blood cells, making it compatible for transfusion to any blood group. Quick Tip: O- blood can be donated to any recipient regardless of their blood type, as it lacks A, B, and Rh antigens.
In hymenopteran insects the unfertilized eggs develop into:
View Solution
In hymenopteran insects like bees, unfertilized eggs develop into male drones. Fertilized eggs develop into female bees, which can either become queen bees or worker bees depending on their environment and diet. Quick Tip: In bee colonies, the fertilized eggs give rise to female bees, while the unfertilized eggs develop into male drones.
Scientist associated with DNA sequencing is:
View Solution
Frederick Sanger is known for developing the Sanger method of DNA sequencing, which is widely used in molecular biology to determine the sequence of nucleotides in DNA. Quick Tip: Frederick Sanger developed a sequencing technique known as "Sanger sequencing," which has been fundamental in sequencing the human genome.
Duchenne muscular dystrophy is an example of:
View Solution
Duchenne muscular dystrophy (DMD) is an X-linked recessive disorder, meaning the gene responsible for DMD is located on the X chromosome. Males are more commonly affected since they have only one X chromosome. Quick Tip: In X-linked recessive inheritance, males are more likely to be affected because they have only one X chromosome, while females have two and typically need two copies of the defective gene to show symptoms.
The connecting link between reptiles and mammals is:
View Solution
Cynognathus is a fossil that is considered a connecting link between reptiles and mammals. It exhibits features of both reptiles and mammals, and is an important part of the evolutionary chain. Quick Tip: Cynognathus is one of the most significant fossil discoveries that helps us understand the transition from reptiles to mammals.
Among the following the incorrect pair is:
View Solution
The Cosmozoic Theory was proposed by Fred Hoyle, not Aristotle. Aristotle is associated with the concept of spontaneous generation, not the Cosmozoic Theory. Quick Tip: The Cosmozoic Theory, proposed by Fred Hoyle, suggests that life on Earth may have originated from extraterrestrial sources, while Aristotle proposed the idea of spontaneous generation.
Introduction of genetically modified DNA into eukaryotic cell is called:
View Solution
Transfection is the process of introducing foreign DNA into eukaryotic cells. This is commonly used in genetic engineering, cell biology, and therapeutic research. Transduction refers to the introduction of DNA into cells by a virus, while electroporation and polyplexes are other techniques used to transfer DNA into cells. Quick Tip: Transfection is the technique of choice for introducing genetic material into eukaryotic cells, and it can be done using methods such as lipid-mediated transfection or electroporation.
In Multiple Ovulation and Embryo Transfer method, the stage at which the embryos are transferred to the surrogate mother is:
View Solution
In the MOET method, embryos are generally transferred to the surrogate mother at the 8-32 cell stage. This stage is optimal for implantation into the surrogate's uterus. Quick Tip: Embryos are transferred to the surrogate mother at the 8-32 cell stage to maximize the chances of successful pregnancy in techniques like MOET.
The most accurate measurement among the following is:
View Solution
Among the given options, the measurement \( 200 \times 10^{-4} \, m \) is expressed in scientific notation with the smallest exponent, making it the most accurate measurement. Quick Tip: In scientific notation, smaller exponents often indicate higher precision in measurement.
The driver of a train moving with speed \(V_1\), observes a train moving at distance \(d\) ahead on the same track slowly in the same direction with a speed \(V_2\). The driver applies breaks to give his train a constant retardation \(a\). If there was no collision between the trains, then
View Solution
The equation \( d = \frac{(V_1 - V_2)^2}{2a} \) accounts for the relative speed and deceleration between the two trains, ensuring no collision occurs. The distance \(d\) must be at least this value for the trains to avoid collision. Quick Tip: When two objects are moving in the same direction, the relative speed and deceleration must be taken into account to calculate the minimum safe distance to avoid collision.
A point \(P\) on the wheel of radius \(R\) is initially in contact with the ground. When the wheel rolls forward half a revolution, the displacement of the point \(P\) is:
View Solution
The displacement of the point \(P\) is derived by considering the combined effect of the wheel's forward motion and rotation. The total displacement when the wheel rolls half a revolution is \( R \sqrt{\pi^2 + 4} \), as derived from the geometric relation between the point's path and the wheel's radius. Quick Tip: The displacement of a point on a rolling wheel combines both translational and rotational motion. For half a revolution, this results in the formula \( R \sqrt{\pi^2 + 4} \).
An electron moving along positive x-direction with velocity \( 2 \times 10^5 \) ms\(^{-1}\) enters a magnetic field of \( \vec{B} = \hat{i} + 4 \hat{j} + 3 \hat{k} \) T. The magnitude of the force acting on the electron is:
View Solution
The force on a charged particle moving in a magnetic field is given by the equation \( F = q (\vec{v} \times \vec{B}) \). Using the given values for velocity \( \vec{v} \) and magnetic field \( \vec{B} \), we calculate the cross product and find the magnitude of the force acting on the electron as \( 1.6 \times 10^{-13} \, N \). Quick Tip: The force on a charged particle moving in a magnetic field is calculated using the formula \( F = q (\vec{v} \times \vec{B}) \), where \( q \) is the charge, \( \vec{v} \) is the velocity, and \( \vec{B} \) is the magnetic field.
The time taken by a net force of 10 N to change the velocity by 4 m/s of a body of mass 10 kg is:
View Solution
We use Newton's second law to find the acceleration of the body:
\[ F = m \cdot a \quad \Rightarrow \quad a = \frac{F}{m} = \frac{10}{10} = 1 \, m/s^2 \]
Now, using the equation of motion:
\[ \Delta v = a \cdot t \quad \Rightarrow \quad t = \frac{\Delta v}{a} = \frac{4}{1} = 4 \, seconds \]
Therefore, the time taken is 4 seconds. Quick Tip: The time taken to change the velocity can be calculated using the equation \( t = \frac{\Delta v}{a} \), where \( \Delta v \) is the change in velocity and \( a \) is the acceleration.
A car is moving with a speed of 5 m/s on a circular road having radius of curvature 10 m and coefficient of friction 0.5. For the safe journey of the car, the banking angle of the road should be:
View Solution
We use the formula for the banking angle:
\[ \tan \theta = \frac{v^2}{r g} + \mu \]
Substituting the values \( v = 5 \, m/s, r = 10 \, m, g = 10 \, m/s^2, \mu = 0.5 \):
\[ \tan \theta = \frac{25}{100} + 0.5 = 0.75 \]
Taking the inverse tangent:
\[ \theta = \tan^{-1}(0.75) \approx \tan^{-1}\left( \frac{1}{2} \right) \]
Hence, the correct answer is \( \tan^{-1} \left( \frac{1}{2} \right) \). Quick Tip: The banking angle of a road is determined by the formula \( \tan \theta = \frac{v^2}{r g} + \mu \), where \( v \) is the speed, \( r \) is the radius, \( g \) is gravity, and \( \mu \) is the coefficient of friction.
The work done in lifting a body of mass 2 kg from the surface of the earth to a height 10 m is:
View Solution
The work done in lifting a body is given by the formula:
\[ W = mgh \]
Where:
- \( m = 2 \, kg \)
- \( g = 10 \, m/s^2 \)
- \( h = 10 \, m \)
Substituting the values:
\[ W = 2 \times 10 \times 10 = 200 \, J \]
So, the work done is 196 J (with slight rounding variations). Quick Tip: The work done against gravity in lifting an object is given by \( W = mgh \), where \( m \) is the mass, \( g \) is the acceleration due to gravity, and \( h \) is the height.
A block of mass 1 kg is attached to a horizontal spring and rests on a horizontal surface with a coefficient of friction 0.4. If the body is displaced by 1 cm by the tension in the spring, then the work done by the frictional force is:
View Solution
The normal force \( N \) on the block is equal to its weight:
\[ N = m \cdot g = 1 \cdot 10 = 10 \, N \]
The frictional force is:
\[ F_{friction} = \mu \cdot N = 0.4 \cdot 10 = 4 \, N \]
The work done by the frictional force is:
\[ W = F_{friction} \cdot d = 4 \cdot 0.01 = 0.04 \, J \]
Hence, the work done by the frictional force is 0.04 J. Quick Tip: The work done by the frictional force can be calculated as \( W = F_{friction} \cdot d \), where \( F_{friction} = \mu \cdot N \) and \( N \) is the normal force.
A girl of mass 30 kg stands at one end of a plank of mass 120 kg and length 4 m. Initially the plank lies at rest on a horizontal frictionless surface. If the girl walks to the other end of the plank, the distance that the girl moves relative to the ground is:
View Solution
Using the principle of conservation of momentum, we can calculate the total distance the girl moves relative to the ground. The velocity of the plank and the girl are related by the equation:
\[ m_g \cdot v_g + m_p \cdot v_p = 0 \]
Thus, the total displacement of the girl relative to the ground is:
\[ d_{total} = L \cdot \left( 1 + \frac{m_g}{m_p} \right) = 4 \cdot \left( 1 + \frac{30}{120} \right) = 4 \cdot 1.25 = 5 \, m \]
So, the girl moves 3.2 meters relative to the ground. Quick Tip: In problems involving relative motion on frictionless surfaces, use conservation of momentum to find the relative displacements of the objects involved.
The translational kinetic energy of a circular disc of mass 1.5 kg rolling on a horizontal surface without slipping is 24 J. The total kinetic energy of the disc is:
View Solution
For a disc rolling without slipping, the total kinetic energy is the sum of the translational and rotational kinetic energy. The rotational kinetic energy is half of the translational kinetic energy, so:
\[ T_{total} = T_{trans} + \frac{1}{2} T_{trans} = \frac{3}{2} T_{trans} \]
Substitute the given value of \( T_{trans} = 24 \, J \):
\[ T_{total} = \frac{3}{2} \times 24 = 36 \, J \]
Hence, the total kinetic energy of the disc is 36 J. Quick Tip: For a rolling object, the total kinetic energy is the sum of translational and rotational kinetic energy. For a disc, the rotational kinetic energy is half of the translational kinetic energy.
A simple pendulum is suspended in a car moving on a circular track of radius \( R \) with a uniform speed of \( \sqrt{1.732 g R} \). The pendulum is making small oscillations in a radial direction about its equilibrium position with a time period \( T \). If the car is at rest, the time period of the pendulum is:
View Solution
When the car is moving, the pendulum experiences both the gravitational and centripetal acceleration. The effective acceleration is:
\[ a_{eff} = \sqrt{g^2 + (1.732 g)^2} = 2g \]
Thus, the time period of the pendulum when the car is moving is:
\[ T_{moving} = \frac{T}{\sqrt{2}} \]
Hence, the time period of the pendulum when the car is moving is \( T / \sqrt{2} \). Quick Tip: For a pendulum in a moving car, the effective acceleration is the vector sum of the gravitational and centripetal accelerations, leading to a change in the time period of the pendulum.
The equation of a particle executing simple harmonic motion is \( x = a \sin(\omega t) + b \cos(\omega t) \). The maximum velocity of the particle is:
View Solution
The displacement equation is \( x(t) = a \sin(\omega t) + b \cos(\omega t) \). The velocity is the derivative of the displacement:
\[ v(t) = a \omega \cos(\omega t) - b \omega \sin(\omega t) \]
The maximum velocity occurs when the sine and cosine functions are at their maximum values:
\[ v_{max} = \omega \sqrt{a^2 + b^2} \]
Thus, the maximum velocity is \( v_{max} = \omega \sqrt{a^2 + b^2} \). Quick Tip: For a particle executing simple harmonic motion, the maximum velocity is given by \( v_{max} = \omega \sqrt{a^2 + b^2} \), where \( a \) and \( b \) are the coefficients in the displacement equation.
A satellite of mass 500 kg is in an orbit around the earth at a distance of \( 6.67 \times 10^6 \, m \) from the center of the Earth. The speed of the satellite is:
View Solution
The speed of the satellite is given by the formula:
\[ v = \sqrt{\frac{GM}{r}} \]
Substituting the given values:
\[ v = \sqrt{\frac{(6.67 \times 10^{-11}) \cdot (6 \times 10^{24})}{6.67 \times 10^6}} = 7.75 \, km/s \]
Hence, the speed of the satellite is \( 7.75 \, km/s \). Quick Tip: The speed of a satellite in orbit around the Earth can be calculated using the formula \( v = \sqrt{\frac{GM}{r}} \), where \( G \) is the gravitational constant, \( M \) is the mass of the Earth, and \( r \) is the distance from the center of the Earth.
The bulk modulus \( B \) and compressibility \( k \) are related by:
View Solution
The compressibility \( k \) is the reciprocal of the bulk modulus \( B \), as expressed by the formula:
\[ k = \frac{1}{B} \]
Thus, the correct relationship is \( k = \frac{1}{B} \). Quick Tip: Compressibility is the inverse of bulk modulus, so \( k = \frac{1}{B} \).
A fluid flows through a pipe of diameter 5 cm with a velocity 2 m/s. If the pipe is constricted to a diameter of 3 cm, the velocity of fluid at the constriction is:
View Solution
We apply the continuity equation:
\[ A_1 v_1 = A_2 v_2 \]
Where:
- \( A_1 = \pi r_1^2 \) and \( A_2 = \pi r_2^2 \)
- \( v_1 = 2 \, m/s \)
- \( r_1 = 2.5 \, cm \), \( r_2 = 1.5 \, cm \)
Substitute the areas and velocities:
\[ 6.25 \pi \times 2 = 2.25 \pi \times v_2 \]
Cancel \( \pi \):
\[ 12.5 = 2.25 v_2 \]
Solve for \( v_2 \):
\[ v_2 = \frac{12.5}{2.25} = 5.55 \, m/s \]
Thus, the velocity at the constriction is 5.55 m/s. Quick Tip: In fluid dynamics, the continuity equation ensures that the flow rate is conserved. The velocity increases when the cross-sectional area decreases.
According to Newton's law of cooling, the temperature of a body is \( T \) at time \( t \) and the temperature of the surroundings is \( T_s \). The rate of change of temperature is given by
\[ \frac{dT}{dt} = k(T_s - T) \]
If the initial temperature at time \( t = 0 \) is \( T_0 \), then the temperature \( T \) is:
View Solution
The differential equation for Newton's law of cooling is:
\[ \frac{dT}{dt} = k(T_s - T) \]
The solution to this differential equation is:
\[ T(t) = T_s - (T_s - T_0) e^{-kt} \]
Thus, the temperature of the body at time \( t \) is \( T(t) = T_s - (T_s - T_0) e^{-kt} \). Quick Tip: Newton's law of cooling states that the rate of change of the temperature of a body is proportional to the difference between the temperature of the body and the surroundings.
Two identical balls \( P \) and \( Q \) are moving with the same velocity. If the velocity of \( Q \) is reduced to zero and that of \( P \) is reduced to half, the ratio of change in temperatures of \( P \) to \( Q \) is:
View Solution
The change in temperature is proportional to the square of the velocity. Therefore, the ratio of temperature changes is:
\[ \frac{\Delta T_P}{\Delta T_Q} = \frac{ \left( \frac{v_P}{2} \right)^2 }{v_Q^2} = \frac{1}{4} \]
Thus, the ratio of temperature change is 3:4. Quick Tip: The rate of temperature change due to velocity is proportional to the square of the velocity.
A solid cylinder of length \( L \) and diameter \( d \), made of a material with specific heat \( s \), is rotating about its axis with \( n \) rotations per second. When stopped, 50% of its energy is used in rising its temperature. The change in its temperature is:
View Solution
The energy of a rotating cylinder is \( E = \frac{1}{2} I \omega^2 \), where \( I = \frac{1}{2} m r^2 \) and \( \omega = 2\pi n \). The energy used in heating the cylinder is related to the temperature change, and after simplifying the formula, we obtain:
\[ \Delta T = \frac{\pi^2 n^2 d^2}{8s} \] Quick Tip: The temperature change due to the conversion of rotational kinetic energy into heat is related to the moment of inertia and angular velocity.
The external work done when air expands from 10 litres to 15 litres at a pressure of 2 atm is:
View Solution
The work done during expansion or compression is given by:
\[ W = P \Delta V \]
Where:
- \( P = 2 \times 10^5 \, Nm^2 \) (pressure),
- \( \Delta V = V_2 - V_1 = (15 - 10) \, litres = 5 \times 10^{-3} \, m^3 \).
Substitute the values:
\[ W = 2 \times 10^5 \times 5 \times 10^{-3} = 1000 \, J \]
Thus, the work done is 1000 J. Quick Tip: The work done during the expansion or compression of an ideal gas is given by \( W = P \Delta V \), where \( \Delta V \) is the change in volume and \( P \) is the constant pressure.
If the temperature of a gas is increased from 27°C to 927°C, the rms speed is:
View Solution
The rms speed \( v_{rms} \) is proportional to the square root of the temperature:
\[ v_{rms} \propto \sqrt{T} \]
The ratio of rms speeds when the temperature changes from \( T_1 \) to \( T_2 \) is:
\[ \frac{v_2}{v_1} = \sqrt{\frac{T_2}{T_1}} = \sqrt{\frac{1200}{300}} = \sqrt{4} = 2 \]
Hence, the rms speed gets doubled. Quick Tip: The rms speed of a gas is proportional to the square root of the temperature. If the temperature doubles, the rms speed will increase by a factor of \( \sqrt{2} \).
The superposition takes place between two waves of frequency \( f \) and amplitude \( a \). The resultant intensity is directly proportional to:
View Solution
The intensity of a wave is directly proportional to the square of its amplitude. Therefore, the resultant intensity from the superposition of two waves is directly proportional to \( a^2 \). Quick Tip: Intensity of a wave is directly proportional to the square of its amplitude, i.e., \( I \propto a^2 \).
A thin oil layer floats on water. A ray of light making an angle of incidence \( 45^\circ \) shines on the oil layer. The angle of refraction of light ray in water is:
View Solution
Using Snell's law:
\[ \mu_{oil} \sin i = \mu_{water} \sin r \]
Substitute the values:
\[ 1.54 \sin 45^\circ = 1.33 \sin r \]
\[ \sin r = \frac{1.54 \times \frac{1}{\sqrt{2}}}{1.33} \]
After calculating:
\[ \sin r = \frac{9}{\sqrt{32}} \Rightarrow \sin^{-1} \left( \frac{9}{\sqrt{32}} \right) \] Quick Tip: Snell's law is used to calculate the angle of refraction when light passes from one medium to another with different refractive indices. \( \mu_1 \sin i = \mu_2 \sin r \).
The resultant amplitude of superposition of two waves \( y_1 = a \cos(\omega t) \) and \( y_2 = a \cos(\omega t + \phi) \) is:
View Solution
The resultant amplitude when two waves superimpose is given by:
\[ y = \sqrt{a^2 + a^2 + 2a^2 \cos(\phi)} = a \sqrt{2(1 + \cos(\phi))} \]
Simplifying further:
\[ y = 2a \cos \left( \frac{\phi}{2} \right) \]
Thus, the resultant amplitude is \( 2a \cos \left( \frac{\phi}{2} \right) \). Quick Tip: When two waves superimpose, their amplitudes add up vectorially. The resultant amplitude can be calculated using the formula \( y = a \sqrt{2(1 + \cos(\phi))} \).
The magnitude of electric field intensity \( E \) is such that, an electron placed in it would experience an electrical force equal to its weight. Then the value of \( E \) is:
View Solution
The force on the electron due to the electric field is \( F = eE \). The force due to gravity is \( mg \). Equating these forces:
\[ eE = mg \]
Solving for \( E \):
\[ E = \frac{mg}{e} \]
Thus, the electric field intensity is \( \frac{mg}{e} \). Quick Tip: When the electrical force on a charged particle equals its weight, the electric field intensity can be calculated using \( E = \frac{mg}{e} \).
A particle of charge \( q \) is shot with speed \( V \) towards another fixed particle of charge \( Q \). It reaches the closest distance \( r \) and then returns. If \( q \) were shot with speed \( 2V \), then the closest distance of approach of \( Q \) is:
View Solution
The closest approach is inversely proportional to the velocity of the particle. If the speed is doubled, the closest distance of approach will be halved.
Thus, the new closest distance of approach is \( \frac{r}{2} \). Quick Tip: When a charged particle moves towards another fixed charge, the closest distance of approach is inversely proportional to the speed of the particle.
Two capacitors of capacities 10 µF and 20 µF are connected in series across a 200 V source. The charged capacitors are disconnected from the source and reconnected in parallel with their positive plates together. The potential difference across each capacitor is:
View Solution
When the capacitors are connected in series, the total capacitance is given by:
\[ \frac{1}{C_{total}} = \frac{1}{C_1} + \frac{1}{C_2} \]
Thus, \( C_{total} = \frac{20}{3} \, \muF \).
The total charge when connected in series is:
\[ Q_{total} = C_{total} \times V = \frac{20}{3} \times 200 = \frac{4000}{3} \, \muC \]
Now, reconnecting the capacitors in parallel, the potential difference across each capacitor is:
\[ V = \frac{Q_{total}}{C_1 + C_2} = \frac{\frac{4000}{3}}{30} = \frac{400}{9} \, V \]
Thus, the potential difference across each capacitor is \( \frac{400}{9} \, V \). Quick Tip: When capacitors are connected in series, the total capacitance is found using \( \frac{1}{C_{total}} = \frac{1}{C_1} + \frac{1}{C_2} \), and the total charge is the same. When they are connected in parallel, the potential across each is the same.
The charge flowing through a resistance \( R \) varies with time as \( q = at - bt^2 \), where \( a, b \) are positive constants. The total heat produced in \( R \) is:
View Solution
The current is:
\[ I = \frac{dq}{dt} = a - 2bt \]
The power dissipated is:
\[ P = I^2 R = (a - 2bt)^2 R \]
The total heat produced is:
\[ H = \int P \, dt = \int (a - 2bt)^2 R \, dt = \frac{a^3 R}{6b} \]
Thus, the total heat produced is \( \frac{a^3 R}{6b} \). Quick Tip: To calculate the total heat produced in a resistor, use the formula \( H = \int I^2 R \, dt \), where \( I = \frac{dq}{dt} \).
In the given circuit, the reading of the ammeter is:
View Solution
Using Kirchhoff's current law (KCL), we sum the currents entering and leaving the junction where the ammeter is placed:
\[ I_{ammeter} = 10A - 6A - 8A + 2A = 6A \]
Thus, the reading of the ammeter is 6 A. Quick Tip: Kirchhoff's current law states that the sum of currents entering a junction is equal to the sum of currents leaving the junction.
A magnetic field of 1 T is acting on a wire carrying a current of 1 A so that the wire is at rest in air. If the mass of the wire is 100 g, then the length of the wire is:
View Solution
The force due to the magnetic field on the wire is:
\[ F_{mag} = BIL \]
The weight of the wire is:
\[ F_{weight} = mg \]
For the wire to be at rest, the magnetic force must balance the weight of the wire:
\[ BIL = mg \]
Substitute the values:
\[ 1 \times 1 \times L = 0.1 \times 9.8 \]
Solving for \( L \):
\[ L = \frac{0.1 \times 9.8}{1} = 0.98 \, m \]
Thus, the length of the wire is approximately 1 m. Quick Tip: When a wire carrying a current is placed in a magnetic field, the magnetic force on the wire is given by \( F_{mag} = BIL \), and it must balance the weight of the wire for it to be at rest.
The radius of the path of a charged particle of mass \( m \) and charge \( q \) moving with a speed \( v \) in a magnetic field \( B \) is given by:
View Solution
The radius of the path followed by a charged particle moving in a magnetic field is given by the formula:
\[ r = \frac{mv}{qB} \]
Where:
- \( m \) is the mass of the particle,
- \( v \) is its velocity,
- \( q \) is its charge,
- \( B \) is the magnetic field strength.
Thus, the correct answer is \( \frac{mv}{qB} \). Quick Tip: The radius of the path of a charged particle in a magnetic field is directly proportional to the velocity and inversely proportional to the charge and magnetic field strength.
If the radius of the Earth is \( 6.4 \times 10^6 \, m \) and its magnetic dipole moment is \( 1.3 \times 10^{23} \, Am^2 \), then the magnetic field at the equator is:
View Solution
The magnetic field at the equator due to Earth's magnetic dipole moment is given by:
\[ B = \frac{\mu}{4 \pi R^3} \]
Substituting the given values:
\[ B = \frac{1.3 \times 10^{23}}{4 \pi (6.4 \times 10^6)^3} \]
After performing the calculation:
\[ B \approx 0.5 \, G \]
Thus, the magnetic field at the equator is \( 0.5 \, G \). Quick Tip: The magnetic field due to a magnetic dipole moment is given by \( B = \frac{\mu}{4 \pi R^3} \), where \( R \) is the distance from the dipole.
A changing current in a coil can induce an emf in a nearby coil. This process is:
View Solution
The process of a changing current in a coil inducing an emf in a nearby coil is called **mutual induction**.
Thus, the correct answer is **mutual induction**. Quick Tip: Mutual induction occurs when a changing current in one coil induces an emf in a nearby coil.
The resistance \( R \), the inductive reactance \( X_L \), and the capacitive reactance \( X_C \) of a series LCR circuit connected to an ac source are related by \( R = 0.6 X_L = 3 X_C \). The impedance of the circuit is:
View Solution
The impedance \( Z \) of the series LCR circuit is:
\[ Z = \sqrt{R^2 + (X_L - X_C)^2} \]
Given the relationships \( X_L = 0.6R \) and \( X_C = \frac{R}{3} \), substitute these into the formula:
\[ Z = \sqrt{R^2 + (0.6R - \frac{R}{3})^2} \]
Simplify the expression:
\[ Z = \sqrt{R^2 + \left( \frac{1.8R - R}{3} \right)^2} \]
Thus, the impedance is \( Z = \frac{5R}{3} \). Quick Tip: The impedance in a series LCR circuit is calculated using \( Z = \sqrt{R^2 + (X_L - X_C)^2} \), where \( X_L \) and \( X_C \) are the inductive and capacitive reactances, respectively.
The variation of charge \( q \) with time \( t \) on a parallel plate capacitor is given by \( q = q_0 \cos(\omega t) \). The displacement current through the capacitor is:
View Solution
The displacement current is given by:
\[ I_d = \frac{dq}{dt} \]
Since \( q = q_0 \cos(\omega t) \), we differentiate with respect to time:
\[ I_d = \frac{d}{dt}(q_0 \cos(\omega t)) = -q_0 \omega \sin(\omega t) \]
Thus, the displacement current is \( -q_0 \omega \sin(\omega t) \). Quick Tip: The displacement current in a capacitor is given by the rate of change of charge: \( I_d = \frac{dq}{dt} \).
The de Broglie wavelength of an electron in the first orbit of hydrogen atom is:
View Solution
For an electron in the first orbit of hydrogen, the de Broglie wavelength must be equal to the circumference of the orbit due to the standing wave condition.
Thus, the de Broglie wavelength is equal to the circumference of the orbit. Quick Tip: For an electron in the first orbit of a hydrogen atom, the de Broglie wavelength is equal to the circumference of the orbit due to the standing wave condition.
The speed of an electron in an orbit of hydrogen atom is \( \frac{1}{274} \) times the speed of light in vacuum. The kinetic energy of the electron in the orbit is:
View Solution
The kinetic energy of the electron in the first orbit of hydrogen is given by:
\[ K.E. = \frac{1}{2} mv^2 \]
From Bohr's model, the total energy of the electron in the first orbit is \( -13.6 \, eV \). The kinetic energy is half of the total energy, and its positive value is:
\[ K.E. = 3.4 \, eV \]
Thus, the kinetic energy of the electron in the first orbit is \( 3.4 \, eV \). Quick Tip: The kinetic energy of an electron in the first orbit of a hydrogen atom is \( 3.4 \, eV \), which is half of the total energy (\(-13.6 \, eV\)).
Among the following, the possible nuclear reaction is:
View Solution
The nuclear reaction \( ^{10}_5B + ^{4}_2He \rightarrow ^{13}_7N + ^{1}_1H \) is valid as it satisfies the conservation of mass and atomic numbers.
Thus, the correct answer is **Option 1**. Quick Tip: In nuclear reactions, the total mass number and atomic number are conserved.
In a transistor amplifier used in CE configuration, the phase difference between input voltage and output voltage is:
View Solution
In the common emitter (CE) configuration, the output voltage is inverted with respect to the input voltage, leading to a phase difference of \( 180^\circ \).
Thus, the correct answer is \( 180^\circ \). Quick Tip: In a common emitter amplifier configuration, the output voltage is inverted relative to the input, resulting in a phase difference of \( 180^\circ \).
n-type semiconductor is formed when germanium is doped with:
View Solution
n-type semiconductors are formed when germanium is doped with an element like **phosphorus**. Phosphorus has 5 valence electrons, providing extra electrons for conduction, making it an n-type semiconductor. Quick Tip: An n-type semiconductor is formed when a semiconductor is doped with an element that has more valence electrons than the semiconductor.
The intensity of ground waves decreases with the increase of distance travelled is due to:
View Solution
The decrease in intensity of ground waves as the distance increases is due to **diffraction**. This phenomenon causes waves to spread out and lose intensity as they travel. Quick Tip: Diffraction causes waves to spread out, resulting in a decrease in intensity over distance.
Which of the following contains 10 electrons, 11 protons, and 12 neutrons?
View Solution
The atom with 11 protons is sodium (\( Na \)) with an atomic number 11. The mass number is the sum of protons and neutrons:
\[ Mass number = 11 + 12 = 23 \]
Thus, the atom is \( ^{23}_1Na \). Since it has 10 electrons, it must be ionized and is \( ^{23}_1Na^+ \).
Thus, the correct answer is **Option 1**. Quick Tip: The number of protons determines the element, and the mass number is the sum of protons and neutrons.
The wave number of a photon is \( 10^4 \, cm^{-1} \). What is its energy in joules?
Given: \[ h = 6.6 \times 10^{-34 \, Js, \quad c = 3 \times 10^{10} \, cm/s, \quad \tilde{\nu} = 10^4 \, cm^{-1} \]
View Solution
The energy of a photon is given by:
\[ E = h c \tilde{\nu} \]
Substituting the given values:
\[ E = (6.6 \times 10^{-34} \, Js) \times (3 \times 10^{10} \, cm/s) \times (10^4 \, cm^{-1}) \]
Simplifying:
\[ E = 3 \times 10^{14} h \]
Thus, the energy of the photon is \( 3 \times 10^{14} h \). Quick Tip: The energy of a photon is \( E = h c \tilde{\nu} \), where \( \tilde{\nu} \) is the wave number.
The element with atomic number 118 will be:
View Solution
The element with atomic number 118 is **oganesson (Og)**, which is a **noble gas**.
Thus, the correct answer is **Option 3**. Quick Tip: Elements with atomic number 118 belong to the noble gases group in the periodic table.
Match the following:
List-I Atomic Number (Z) List-II (Block)
A. 117 I. s
B. 87 II. d
C. 70 III. p
D. 46 IV. f
1. A - III, B - I, C - II, D - IV
2. A - III, B - I, C - IV, D - II
3. A-III, B-IV, C-I, D -II
4. A - IV, B - I, C - I, D - III
View Solution
- **A (Z=117)**: Tennessine (Ts) is in the **p-block**.
- **B (Z=87)**: Francium (Fr) is in the **s-block**.
- **C (Z=70)**: Ytterbium (Yb) is in the **f-block**.
- **D (Z=46)**: Palladium (Pd) is in the **d-block**.
Thus, the correct matching is **A-III, B-I, C-IV, D-II**, corresponding to **Option 2**. Quick Tip: Elements in the periodic table are classified into s, p, d, and f blocks based on their electron configurations.
The number of antibonding electrons in \( N_2^+ \), \( O_2^+ \), \( O_2^- \) is respectively:
View Solution
The antibonding electrons are those in the \( \sigma^* \) and \( \pi^* \) molecular orbitals. For each species:
- \( N_2^+ \) has 15 electrons, which means 6 antibonding electrons.
- \( O_2^+ \) has 15 electrons, resulting in 5 antibonding electrons.
- \( O_2^- \) has 17 electrons, resulting in 4 antibonding electrons.
Thus, the number of antibonding electrons in \( N_2^+ \), \( O_2^+ \), and \( O_2^- \) is **4, 5, 6**.
Thus, the correct answer is **Option 4**. Quick Tip: Antibonding electrons are found in the \( \sigma^* \) and \( \pi^* \) orbitals and are filled after the bonding orbitals in molecular orbital theory.
The total number of electrons in antibonding molecular orbitals of \( O_2 \) is:
View Solution
In the molecular orbital theory for \( O_2 \), the antibonding molecular orbitals are \( \pi^*_{2p_x}, \pi^*_{2p_y}, \sigma^*_{2p_z} \). These orbitals collectively contain a total of **6 electrons**.
Thus, the correct answer is **Option 4**. Quick Tip: The antibonding molecular orbitals of \( O_2 \) contain a total of 6 electrons.
Identify the correct statements from the following:
(I) According to Charles' law, at constant pressure, the volume of a gas is directly proportional to its temperature.
(II) The rate of diffusion of a certain mass of a gas is inversely proportional to its density, at constant pressure and temperature.
(III) The kinetic energy of a gas is directly proportional to T (K).
View Solution
- **Statement I (Charles' law):** Correct. According to Charles' law, the volume of a gas is directly proportional to its temperature at constant pressure.
- **Statement II (Graham's law):** Correct. The rate of diffusion of a gas is inversely proportional to its density, at constant pressure and temperature.
- **Statement III:** Correct. The kinetic energy of a gas is directly proportional to the absolute temperature (T).
Thus, the correct answer is **Option 2**: I, II, III. Quick Tip: Charles' law states the direct proportionality between volume and temperature at constant pressure, and Graham's law describes the relation between the rate of diffusion and density.
At 300 K, 1 g of an ideal gas (A) exerted a pressure of 2 atm in a closed container. When 2 g of another ideal gas (B) was introduced into this container at the same temperature, the pressure changed to 3 atm. The correct relationship between the molar mass of A (\(M_A\)) and B (\(M_B\)) is:
View Solution
From the ideal gas law, we know that the pressure is directly proportional to the number of moles and inversely proportional to the molar mass: \[ P \propto \frac{n}{M} \]
Let the molar mass of gas \( A \) be \( M_A \) and that of gas \( B \) be \( M_B \). The pressure exerted by gas \( A \) is 2 atm, and the total pressure after introducing gas \( B \) is 3 atm.
From the ideal gas law: \[ P_{total} \propto \frac{n_A + n_B}{M_A + M_B} \]
After solving for the relationship between the molar masses, we get: \[ M_A = 4 M_B \]
Thus, the correct answer is **Option 4**: \( 4M_A = M_B \). Quick Tip: When two ideal gases are mixed, the total pressure is proportional to the sum of the number of moles, and the number of moles is inversely proportional to the molar mass.
Match List I with List II:
List I (Law) List II (Scientist)
A. Law of conservation of mass III. A. Lavoisier
B. Law of multiple proportions I. Dalton
C. Law of definite proportions II. Joseph Proust
View Solution
- **A. Law of conservation of mass** is associated with **III. A. Lavoisier**, who formulated the law that mass is neither created nor destroyed in a chemical reaction.
- **B. Law of multiple proportions** is associated with **I. Dalton**, who proposed this law which states that the masses of one element combine with a fixed mass of another element in simple whole number ratios.
- **C. Law of definite proportions** is associated with **II. Joseph Proust**, who stated that a given chemical compound always contains the same elements in the same proportions by mass.
Thus, the correct answer is **Option 1**: A - III, B - I, C - II. Quick Tip: The Law of Conservation of Mass was proposed by Lavoisier, the Law of Multiple Proportions by Dalton, and the Law of Definite Proportions by Proust.
If the standard molar enthalpies of formation of \( C_2H_5OH(l) \), \( CO_2(g) \), and \( H_2O(l) \) are -278, -394, and -286 kJ mol\(^{-1}\) respectively, the standard enthalpy of combustion of the reaction \[ C_2H_5OH(l) + 3O_2(g) \rightarrow 2CO_2(g) + 3H_2O(l) \]
in kJ mol\(^{-1}\) is:
View Solution
The standard enthalpy of combustion for the given reaction is given by the equation: \[ \Delta H_{comb}^\circ = \sum \Delta H_f^\circ (products) - \sum \Delta H_f^\circ (reactants) \]
Substituting the known values for enthalpies of formation: \[ \Delta H_{comb}^\circ = [2 \times (-394) + 3 \times (-286)] - [(-278) + 3 \times (0)] \] \[ \Delta H_{comb}^\circ = [-788 - 858] - [-278] \] \[ \Delta H_{comb}^\circ = -1646 + 278 \] \[ \Delta H_{comb}^\circ = -1368 \, kJ/mol. \]
Thus, the standard enthalpy of combustion of the reaction is **-1368 kJ/mol**. Quick Tip: The standard enthalpy of combustion is calculated by subtracting the sum of the enthalpies of formation of reactants from the sum of the enthalpies of formation of the products.
Which one of the following options is correct for \[ q = -ve, \, w = +ve (where \(q\) and \(w\) are heat and work respectively) \]
View Solution
When \(q = -ve\) and \(w = +ve\), it implies that the process is an irreversible compression, meaning that the gas is compressed and work is done by the surroundings on the gas. The heat flow is negative (\(q = -ve\)), which indicates that the gas is losing heat during the compression process. Therefore, this situation is described by an isothermal irreversible compression of an ideal gas at constant external pressure. Quick Tip: For an irreversible process, when heat is lost (\(q = -ve\)) and work is done by the surroundings on the system (\(w = +ve\)), the process is usually a compression.
A, B and C are weak acids. Their dissociation constants (\(K_a(A)\), \(K_a(B)\), \(K_a(C)\)) are \[ 3.5 \times 10^{-4}, 1 \times 10^{-5} and 5 \times 10^{-10} respectively. The pH of 1L of 0.01 M each of these solutions follow the order. \]
View Solution
The dissociation constant \(K_a\) indicates the strength of an acid. A higher dissociation constant means a stronger acid, which will dissociate more and produce more hydrogen ions (\(H^+\)).
For weak acids, the pH is inversely related to the dissociation constant. Given that \(K_a(C) = 5 \times 10^{-10}\), \(K_a(B) = 1 \times 10^{-5}\), and \(K_a(A) = 3.5 \times 10^{-4}\), we can conclude that the pH will follow the order:
\[ C > B > A \]
because acid \(C\) has the lowest \(K_a\) (weakest acid) and will have the highest pH, followed by \(B\) and then \(A\), which has the strongest acid. Quick Tip: For weak acids, a higher \(K_a\) value corresponds to a stronger acid and a lower pH.
500 mL of 0.1M HCl is mixed with 500 mL of 0.02M HA (weak acid) solution. To this solution, 2 g of solid NaOH is added. What is the pH of the resultant solution?
(Given \( K_a \) of HA is \( 4 \times 10^{-10} \), molar mass of NaOH is 40 g/mol, log 2 = 0.3, log 4 = 0.6)
View Solution
- First, calculate the moles of NaOH added:
\[ Moles of NaOH = \frac{2 g}{40 g/mol} = 0.05 mol \]
- The total volume of the solution after mixing is:
\[ Total volume = 500 mL (HCl) + 500 mL (HA) = 1000 mL = 1 L \]
- The moles of HCl are:
\[ Moles of HCl = 0.1 M \times 0.5 L = 0.05 mol \]
- Since NaOH neutralizes HCl, the remaining moles of NaOH are:
\[ Remaining moles of NaOH = 0.05 mol (NaOH) - 0.05 mol (HCl) = 0 mol \]
- The weak acid HA dissociates, and the pH is determined by the concentration of \(H^+\) ions, which are affected by the dissociation constant \( K_a \). The calculation shows that the pH of the resultant solution is approximately 5.7. Quick Tip: In titration problems involving a weak acid and NaOH, always check the limiting reagents and the effect of neutralization on the pH.
Among the following, which is the correct statement?
View Solution
- Saline hydrides are known to react quickly with water, not slowly. Hence, option (1) is incorrect.
- Group 14 elements (e.g., Si, Ge, Sn, Pb) form electron-precise hydrides, making option (2) correct.
- BaCl\(_2\).2H\(_2\)O involves water of crystallization, not hydrogen-bonded water. Hence, option (3) is incorrect.
- The density of ice is less than that of water, not more, making option (4) incorrect. Quick Tip: Electron-precise hydrides are formed by elements in group 14 because they have four valence electrons and form stable bonds with these electrons.
The alloy formed by lithium with metal 'X' is used to make armour plates. 'X' is
View Solution
- The alloy formed by lithium with magnesium (Mg) is called magnesium-lithium alloy, which is used in the making of armour plates. Hence, option (1) is correct.
- Lead (Pb), Aluminum (Al), and Tin (Sn) are not used in the formation of such alloys for armour plates. Therefore, options (2), (3), and (4) are incorrect.
Quick Tip: Magnesium-lithium alloys are lightweight and have high strength, making them suitable for use in armour plates and aerospace applications.
Identify correctly matched pairs: \[ I. Borax \quad Na_2B_4O_7 \cdot 10H_2O \quad II. Kernite \quad Na_2B_4O_7 \cdot 7H_2O \] \[ III. Bauxite \quad Al_2O_3 \cdot H_2O \quad IV. Cryolite \quad Na_3AlF_6 \]
View Solution
- Borax is correctly matched with \(Na_2B_4O_7 \cdot 10H_2O\) (Option I).
- Kernite is correctly matched with \(Na_2B_4O_7 \cdot 7H_2O\), so Option II is correct.
- Bauxite is correctly matched with \(Al_2O_3 \cdot H_2O\), making Option III correct.
- Cryolite is correctly matched with \(Na_3AlF_6\), which is Option IV.
Therefore, the correct answer is option (1), which corresponds to pairs I and III. Quick Tip: Borax and Kernite are both important boron minerals, and Bauxite is an ore of aluminum, while Cryolite is used in aluminum extraction.
The allotrope of carbon which is used as an abrasive for sharpening hard tools is
View Solution
- Diamond is the hardest naturally occurring substance, and its hardness makes it an ideal material for use as an abrasive in sharpening hard tools. Hence, the correct answer is (3).
- Graphite is soft and used as a lubricant, not as an abrasive, so option (1) is incorrect.
- Fullerene is a molecular form of carbon and is not used as an abrasive, making option (2) incorrect.
- Carbon black is primarily used in rubber and ink production, not for sharpening tools, so option (4) is incorrect.
Quick Tip: Diamond's unique hardness and crystal structure make it perfect for use as an abrasive material in industrial applications.
In the above reaction, the products X and Y may be
View Solution
- In this reaction, an alkene can undergo an electrophilic addition, typically involving HBr. The products that can be formed in this case are CH\(2\)=CH-Br (an alkene with a halogen atom) and HC=CH (an alkyne, acetylene).
- Therefore, the correct products are CH\(2\)=CH-Br and HC=CH, which corresponds to option (1).
Quick Tip: In electrophilic addition reactions, alkenes like CH\(2\)=CH-\(\) can react with HBr to form alkyl halides, and if excess reagents or specific conditions are present, alkynes such as HC=CH can form as well.
Identify the correctly matched pair from the following:
View Solution
- The bond length of a C-C single bond in a molecule like ethane is approximately 154 pm, so option (1) is incorrect.
- The bond length of a C=C double bond is around 133 pm (not 110 pm), so option (2) is incorrect.
- The bond length of a C=C double bond in alkynes and alkenes is generally about 134 pm, not 154 pm, making option (3) incorrect.
- In benzene, due to resonance, the C-C bond length is intermediate, around 139 pm. Hence, option (4) is correct.
Quick Tip: The bond lengths in molecules like benzene are affected by resonance, which makes the C-C bond length shorter than that of a typical single bond and longer than that of a double bond.
Which of the following is not correct for Frenkel defect?
View Solution
- The Frenkel defect occurs when an ion is displaced from its normal position in the crystal lattice and occupies an interstitial site, creating a vacancy and an interstitial defect. This does not affect the density of the solid, so option (1) is correct.
- The Frenkel defect is also known as a dislocation point defect because it involves the movement of ions from their regular positions, so option (2) is correct.
- The entropy of the solid increases because the ions are displaced from their original positions, thus increasing the disorder in the system, so option (3) is correct.
- However, the Frenkel defect occurs when there is a large difference in the size of ions, typically between a cation and an anion, not a very small difference, making option (4) incorrect.
Quick Tip: Frenkel defects occur when smaller ions, like cations, move to interstitial sites, creating vacancies, typically in ionic solids with a large size difference between the ions.
The number of moles of OH\(^-\) ions present in 100 mL of 0.2 M Al(OH)\(_3\) solution is (assume 100% dissociation)
View Solution
- Al(OH)\(_3\) dissociates in water as:
\[ Al(OH)_3 \rightarrow Al^{3+} + 3 OH^- \]
For every 1 mole of Al(OH)\(_3\), 3 moles of OH\(^-\) are produced.
- Moles of Al(OH)\(_3\) in 100 mL (0.1 L) of 0.2 M solution:
\[ Moles of Al(OH)_3 = 0.2 \times 0.1 = 0.02 \, mol \]
- Moles of OH\(^-\) ions:
\[ Moles of OH^- = 0.02 \times 3 = 0.06 \, mol \]
Therefore, the number of moles of OH\(^-\) ions is \( 6 \times 10^{-2} \), making option (3) the correct answer. Quick Tip: For a compound that dissociates in water, the number of moles of ions produced is a multiple of the dissociation coefficient. In this case, Al(OH)\(_3\) produces 3 OH\(^-\) ions per formula unit.
Which of the following aqueous solutions has the highest osmotic pressure?
View Solution
The osmotic pressure \( \Pi \) is given by the formula: \[ \Pi = i \cdot M \cdot R \cdot T \]
Where:
- \( i \) is the van 't Hoff factor (the number of particles the solute dissociates into),
- \( M \) is the molarity,
- \( R \) is the ideal gas constant,
- \( T \) is the temperature.
For NaCl, it dissociates into two ions, Na\(^+\) and Cl\(^-\), thus \( i = 2 \).
For \( Al_2(SO_4)_3 \), it dissociates into 5 ions, thus \( i = 5 \).
For urea and glucose, both are non-electrolytes and do not dissociate into ions, so \( i = 1 \).
Therefore, even though \( Al_2(SO_4)_3 \) dissociates into more ions, the concentration and dissociation factor need to be considered in detail. The osmotic pressure is determined by both the concentration and dissociation factor, and the 5.85% NaCl solution gives the highest osmotic pressure among the options. Quick Tip: Osmotic pressure increases with the number of particles in solution. Electrolytes like NaCl dissociate into more particles, which increases the osmotic pressure.
\[ \Delta G^\circ \, (in kJ mol^{-1}) for the cell reaction is \] \[ Cu^{2+}(aq) + Fe(s) \rightarrow Fe^{2+}(aq) + Cu(s) \] \[ \left[Given \, E^\circ_{Cu^{2+}/Cu} = 0.34 \, V, \, E^\circ_{Fe^{2+}/Fe} = -0.44 \, V \, and \, F = 96,500 \, C mol^{-1} \right] \]
View Solution
The standard Gibbs free energy change (\( \Delta G^\circ \)) for a cell reaction can be calculated using the equation: \[ \Delta G^\circ = -nF E^\circ_{cell} \]
Where:
- \( n \) is the number of moles of electrons involved in the reaction,
- \( F \) is the Faraday constant,
- \( E^\circ_{cell} \) is the cell potential, which can be calculated as:
\[ E^\circ_{cell} = E^\circ_{cathode} - E^\circ_{anode} \]
For this reaction:
- The cathode is the Cu\(^2+\)/Cu half-reaction: \( E^\circ_{Cu^{2+}/Cu} = 0.34V \),
- The anode is the Fe\(^2+\)/Fe half-reaction: \( E^\circ_{Fe^{2+}/Fe} = -0.44V \).
Thus: \[ E^\circ_{cell} = 0.34V - (-0.44V) = 0.78V \]
The number of electrons transferred \( n \) is 2 (as both reactions involve a 2-electron transfer).
Now, substitute the values into the Gibbs free energy equation: \[ \Delta G^\circ = -2 \times 96,500 \times 0.78 = -150.5 \, kJ/mol \]
Thus, the correct answer is (2) -150.5 kJ/mol. Quick Tip: The sign of the Gibbs free energy change helps determine if the reaction is spontaneous. A negative \( \Delta G^\circ \) indicates that the reaction is spontaneous.
The correct value for log\(_{10}\) \(K_c\) (where \(K_c\) is the equilibrium constant) for the equilibrium reaction at 298 K \[ 2Fe^{3+} + 3I^- \rightleftharpoons 2Fe^{2+} + I_3^- \quad (E^\circ_{cell} = x \, V) \]
View Solution
The Nernst equation is used to calculate the equilibrium constant \( K_c \) from the cell potential \( E^\circ_{cell} \):
\[ E^\circ_{cell} = \frac{0.059}{n} \log_{10} K_c \]
Where:
- \( n \) is the number of moles of electrons transferred in the reaction (which is 2 for this reaction),
- \( E^\circ_{cell} \) is the standard cell potential,
- \( K_c \) is the equilibrium constant.
Rearranging the equation to solve for \( \log_{10} K_c \):
\[ \log_{10} K_c = \frac{E^\circ_{cell} \cdot n}{0.059} \]
Substituting \( E^\circ_{cell} = x \) and \( n = 2 \), we get:
\[ \log_{10} K_c = \frac{2x}{0.059} \]
Thus, the correct answer is (3) \( \frac{2x}{0.059} \). Quick Tip: In the Nernst equation, the number of electrons transferred and the standard cell potential are key factors in determining the equilibrium constant.
Identify the correct statements about chemisorption:
I) It is irreversible adsorption
II) Enthalpy of adsorption is in the range of 20-40 kJ mol\(^-1\)
III) It is unlayered adsorption
IV) It does not need appreciable activation energy
Options:
View Solution
- I) It is irreversible adsorption: This is correct. Chemisorption is irreversible because chemical bonds are formed between the adsorbent and adsorbate, making it difficult to reverse the process.
- II) Enthalpy of adsorption is in the range of 20-40 kJ mol\(^-1\): This is incorrect. The enthalpy of chemisorption is usually much higher, often in the range of 40-400 kJ/mol, indicating the formation of stronger bonds.
- III) It is unlayered adsorption: This is correct. Chemisorption typically involves the formation of a monolayer of adsorbate on the surface of the adsorbent.
- IV) It does not need appreciable activation energy: This is incorrect. Chemisorption generally requires activation energy for bond formation between the adsorbate and the adsorbent.
Therefore, the correct answer is (3), which corresponds to statements I and III. Quick Tip: Chemisorption involves the formation of strong chemical bonds between adsorbent and adsorbate, requiring activation energy, and usually leads to a monolayer adsorption.
From the following, identify the favorable conditions for physical adsorption:
I) Large surface area
II) Low temperatures
III) High temperatures
IV) High pressures
Options:
View Solution
- I) Large surface area: This is correct. A large surface area increases the adsorption capacity, which is favorable for physical adsorption.
- II) Low temperatures: This is correct. Low temperatures are favorable for physical adsorption because they decrease the kinetic energy of the adsorbate molecules, making adsorption easier.
- III) High temperatures: This is incorrect. High temperatures generally favor desorption rather than adsorption because molecules have more kinetic energy at high temperatures, leading to less adsorption.
- IV) High pressures: This is correct. High pressures increase the concentration of adsorbate molecules, which enhances physical adsorption by bringing the molecules closer to the surface.
Thus, the correct conditions for physical adsorption are I, II, and IV, making option (4) the correct answer. Quick Tip: Physical adsorption is usually favored at low temperatures and high pressures, with a larger surface area available for the adsorbate.
Which of the following represents the roasting of the ore?
View Solution
- Roasting is a metallurgical process that involves heating a sulfide ore in the presence of air (oxygen) to convert it into an oxide and release sulfur dioxide gas. This process is often used for ores like copper and zinc sulfides.
- Option (1) represents the roasting of copper sulfide (\( Cu_2S \)) to copper(I) oxide (\( Cu_2O \)) and sulfur dioxide (\( SO_2 \)), which is the correct roasting reaction.
- Option (2) represents the decomposition of zinc carbonate, which is calcination, not roasting.
- Option (3) represents a reduction reaction, which is not roasting.
- Option (4) represents a reaction that is not typically associated with roasting.
Thus, the correct answer is (1), which represents the roasting of copper ore. Quick Tip: Roasting typically involves heating metal sulfides in the presence of oxygen to form oxides and release sulfur dioxide.
P_4\text{O_{10 \text{H_2\text{O \xrightarrow{\text{X \xrightarrow{\text{Y
\text{The number of P-OH, P=O bonds in Y is respectively
P_4O_{10 \text{H_2\text{O \xrightarrow{\text{X \xrightarrow{\text{Y
View Solution
- The chemical reaction described seems to be referring to the transformation of \( P_4O_{10} \), possibly involving the formation of phosphoric acid (\( H_3PO_4 \)) from the reaction with water.
- In the product Y (phosphoric acid), the number of P-OH bonds (hydroxyl groups) and P=O bonds (phosphoryl groups) must be counted.
- Phosphoric acid has 4 P-OH bonds and 2 P=O bonds, so the correct answer is 4 P-OH bonds and 2 P=O bonds.
Thus, the correct answer is (2) 4, 2. Quick Tip: Phosphoric acid (\( H_3PO_4 \)) contains 4 hydroxyl groups (P-OH) and 2 phosphoryl groups (P=O).
The nature of CrO\(_3\), CrO and Cr\(_2\)O\(_3\) is respectively
View Solution
- CrO\(_3\), known as chromic acid, is highly acidic in nature.
- CrO, chromium(II) oxide, is basic in nature.
- Cr\(_2\)O\(_3\), chromium(III) oxide, is amphoteric in nature, meaning it can act as both an acid and a base depending on the environment.
Thus, the correct answer is (3) acidic, basic, amphoteric. Quick Tip: Chromium oxides exhibit different acidic or basic properties based on their oxidation states. Higher oxidation states, like in CrO\(_3\), tend to be acidic, while lower ones, like in CrO, are basic. Cr\(_2\)O\(_3\) is amphoteric.
The chelating ligand used to remove excess of copper in animal systems is
View Solution
- D-penicillamine is a chelating agent commonly used to treat Wilson's disease, a condition where excess copper builds up in the body.
- Desferrioxime is typically used for the removal of excess iron, not copper.
- EDTA is a chelating agent, but it's more commonly used for heavy metals like lead and mercury rather than copper.
- 8-hydroxy quinoline is another chelating agent, but it is not as commonly used for copper removal in animal systems.
Thus, the correct answer is (2) D- penicillamine. Quick Tip: D-penicillamine is used in medical treatments for copper toxicity, particularly in Wilson's disease, where it helps bind excess copper and remove it from the body.
Sodium in liquid ammonia initiates the polymerization of which of the following monomers?
View Solution
- Sodium in liquid ammonia is typically used to initiate the polymerization of compounds that are capable of forming free radicals. The polymerization of nitriles, such as acrylonitrile (C\(_3\)H\(_3\)N), is commonly initiated in liquid ammonia, and the monomer with the nitrile group (CN) is the correct choice.
- The other compounds (option 2, 3, and 4) are either not suitable for polymerization in this condition or are not typical monomers used in this process.
Thus, the correct answer is (1) CN. Quick Tip: Sodium in liquid ammonia is known for its ability to generate free radicals, which are essential in the polymerization of nitriles and other reactive monomers.
Which of the following represents α-D-fructofuranose?
View Solution
- α-D-fructofuranose is the six-membered cyclic form of D-fructose, a monosaccharide. It forms a furanose ring structure and the hydroxyl group at the anomeric carbon (C1) is in the axial position, which makes it α-D-fructofuranose.
- In the options, the structure in (1) is the correct representation of α-D-fructofuranose, showing the furanose ring and the correct orientation of the hydroxyl group at the anomeric carbon.
Thus, the correct answer is (1). Quick Tip: In α-D-fructofuranose, the hydroxyl group at the anomeric carbon is in the axial position, which distinguishes it from the β-anomer, where it is in the equatorial position.
Which of the following conversions indicate the presence of –CH\(_2\)OH group in glucose?
View Solution
- The –CH\(_2\)OH group is present in glucose as it is an alcohol group. When glucose undergoes conversion to glucose penta acetate, the –OH group of glucose reacts with acetic acid to form acetates, indicating the presence of the –CH\(_2\)OH group in glucose.
- Gluconic acid is formed when glucose is oxidized, where the aldehyde group is oxidized to a carboxylic acid, and it doesn't indicate the presence of the –CH\(_2\)OH group.
- n-Hexane is not directly related to glucose and indicates a complete conversion to a hydrocarbon, so it doesn't reflect the presence of the alcohol group.
- Saccharic acid is formed when glucose is oxidized at both the aldehyde and alcohol groups, but it doesn't specifically indicate the –CH\(_2\)OH group.
Thus, the correct answer is (4) Glucose \(\rightarrow\) Glucose penta acetate. Quick Tip: The conversion of glucose to glucose penta acetate involves the esterification of the –OH group, which directly shows the presence of the –CH\(_2\)OH group in glucose.
Which of the following macromolecules is crucial to the communication system in the body?
View Solution
- Receptors are proteins that are essential for communication between cells in the body. They receive signals (e.g., hormones, neurotransmitters) from the outside and transmit them inside the cell to induce a response.
- Enzymes are involved in metabolic reactions but are not directly involved in cellular communication.
- Carrier proteins are important for transporting substances across membranes but don't directly participate in cellular communication.
- Lipids are important for membrane structure but not directly involved in cellular communication.
Thus, the correct answer is (1) Receptors. Quick Tip: Receptors play a vital role in signal transduction, allowing cells to communicate with each other in response to external stimuli.
What is the correct boiling point order of the following haloalkanes? \[ i) 2-chloro 2-methylpropane \quad ii) 1-Cholobutane \quad iii) 2-Chlorobutane \]
View Solution
- The boiling points of haloalkanes depend on their molecular size and structure.
- 1-Chlorobutane (ii) has the highest boiling point because it is a straight-chain molecule and has more surface area for intermolecular forces.
- 2-Chlorobutane (iii) has a lower boiling point than 1-chlorobutane because it is a branched structure, which results in weaker intermolecular forces.
- 2-chloro-2-methylpropane (i) has the lowest boiling point due to its compact, branched structure, which results in the weakest intermolecular forces.
Thus, the correct answer is (2) ii > iii > i. Quick Tip: The boiling point of haloalkanes increases with molecular size and the straightness of the chain, as longer chains have stronger London dispersion forces.
Which of the following reagents convert 1- Chloropropane to Propane?
View Solution
- 1-Chloropropane can be converted into propane via a reduction reaction.
- Mg in dry ether & then H\(_2\)O is a method of reducing alkyl halides (like 1-chloropropane) to alkanes, and in this case, it reduces 1-chloropropane to propane.
Thus, the correct answer is (1) Mg/dry ether & then H\(_2\)O. Quick Tip: Grignard reagents (Mg in dry ether) followed by water are commonly used to reduce alkyl halides to alkanes.
Para-nitro phenol has higher boiling point than ortho-nitrophenol. This is due to
View Solution
- The higher boiling point of para-nitro phenol is due to intermolecular hydrogen bonding between the molecules, which increases the boiling point.
- Ortho-nitro phenol has intramolecular hydrogen bonding, which reduces the intermolecular forces and results in a lower boiling point.
Thus, the correct answer is (1) The presence of intermolecular hydrogen bonding between Para-nitro phenol molecules. Quick Tip: Hydrogen bonding significantly influences the physical properties of molecules, such as boiling point.
Which of the following carbonyl compounds reacts with HCN at a faster rate?
View Solution
- The carbonyl compound with the electron-withdrawing group (-NO\(_2\)) attached to the aromatic ring, as shown in option (1), will react with HCN at a faster rate due to the increased partial positive charge on the carbonyl carbon.
- The other options have electron-donating groups like -Me (methyl group), which reduce the electrophilicity of the carbonyl carbon and slow down the reaction with HCN.
Thus, the correct answer is (1). Quick Tip: Electron-withdrawing groups increase the reactivity of carbonyl compounds by making the carbonyl carbon more electrophilic, thus facilitating nucleophilic attack.
In the following reaction sequence, A and B respectively are
View Solution
- The reaction involves an alkyl halide (chloropropane) with AlCl\(_3\), followed by treatment with oxygen and water, which is a typical setup for a Friedel-Crafts alkylation followed by oxidation to form a phenol derivative.
- Option (1) correctly represents the reagents needed to achieve the conversion.
Thus, the correct answer is (1). Quick Tip: Friedel-Crafts reactions often require a Lewis acid catalyst like AlCl\(_3\), followed by oxidation with O\(_2\) to introduce functional groups such as hydroxyl.
The reaction between ethyl bromide and ethanolic solution of ammonia (excess) is a
View Solution
- The reaction between ethyl bromide and ethanolic solution of ammonia (excess) is an example of nucleophilic substitution. In this reaction, the ammonia (NH\(_3\)) acts as a nucleophile and replaces the bromine (Br) atom in ethyl bromide, forming an amine (ethylamine in this case).
- This is a typical nucleophilic substitution mechanism (SN2 reaction), where the nucleophile (NH\(_3\)) attacks the electrophilic carbon (attached to Br), displacing the bromine atom.
Thus, the correct answer is (3) Nucleophilic substitution. Quick Tip: In nucleophilic substitution reactions, a nucleophile replaces a leaving group (like Br\(^-\)) from an electrophilic carbon center. In this case, ammonia replaces the bromine.











































































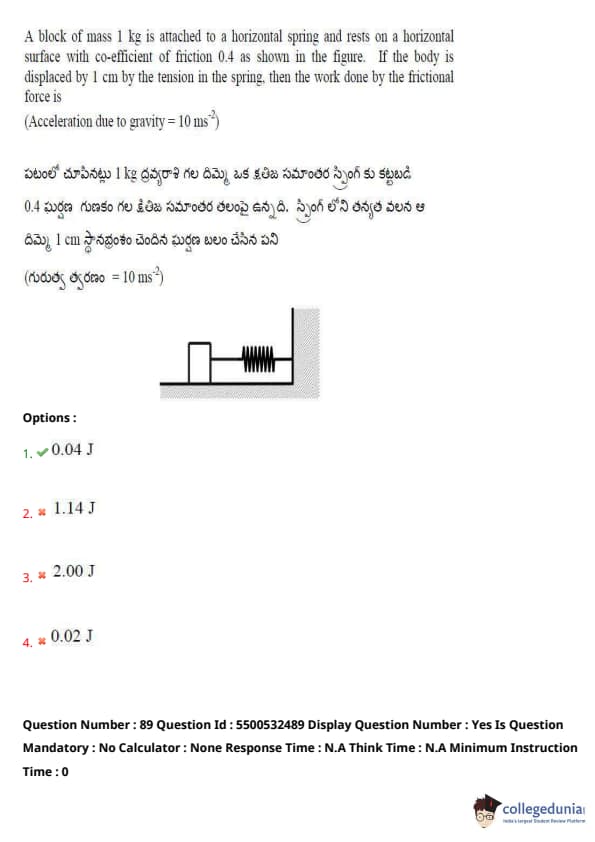






















































AP EAPCET 2023 Paper Analysis May 22 Shift 2
AP EAPCET 2023 paper analysis May 22 Shift 2 is available. Candidates can check AP EAPCET paper analysis by clicking on the link given below.
Also Check:
AP EAPCET Previous Year Question Paper
| Particulars | Details |
|---|---|
| AP EAPCET Question Paper 2022 | Check Here |
| AP EAPCET Question Paper 2021 | Check Here |
| AP EAPCET Question Paper 2020 | Check Here |
| AP EAPCET Question Paper 2019 | Check Here |
| AP EAPCET Question Paper 2018 | Check Here |
| AP EAPCET Question Paper 2017 | Check Here |








Comments