
Exams Prep Master
The laboratory preparation of hydrogen gas usually involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules. Granulated zinc is ideal for the preparation of hydrogen gas in chemical laboratories because it usually contains a small amount of copper, which has the ability to act as a catalyst to the associated chemical reaction and, therefore, increase the rate of the chemical reaction without actually participating in it. An experimental procedure for the laboratory preparation of hydrogen gas is provided below.
| Table of Contents |
Key Takeaways: Hydrogen Gas, Application of Hydrogen Gas, Preparation of Hydrogen Gas, Electrolysis of Water, Properties of Hydrogen Gas
Procedure for the Laboratory Preparation of Hydrogen Gas
[Click Here for Sample Questions]
Step 1: Measure out a few grams of zinc granules into a 500 mL flask.
Step 2: Pour dilute hydrochloric acid over the zinc granules using a thistle funnel. If you don't have access to hydrochloric acid, dilute sulphuric acid can be used instead.
Step 3: Hydrogen gas will be gathered automatically using a delivery tube and water displacement downhill. The fact that hydrogen gas is lighter than water explains this.
The following diagram depicts the setup for producing hydrogen gas in the laboratory.
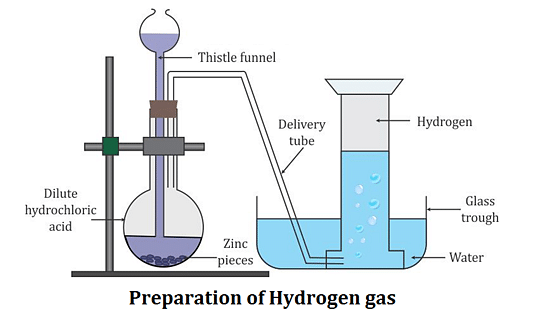
Procedure for the Laboratory Preparation of Hydrogen Gas
The chemical processes that occur during this method of obtaining hydrogen gas are given below.
- General Format: Metal + Dilute Acid → Salt of Metal & Acid + Hydrogen
- With Hydrochloric Acid: Zn + 2HCl → ZnCl2 + H2
- With Sulphuric Acid: Zn + H2SO4 → ZnSO4 + H2
Hydrogen is made in the laboratory by reacting dil. H2SO4 with powdered zinc.
Zn + H2SO4 ——–> ZnSO4 + H2
Zinc granules are inserted in a Woulfe's bottle, which is then filled with water. A thistle funnel and a delivery tube are included in the bottle,conc H2SO4 is gently poured into the thistle funnel. The acid is diluted as it falls into Woulfe's bottle, where it combines with zinc to produce dihydrogen gas. Water is displaced downwards to collect it.The generated dihydrogen bubbles can sometimes adhere to the zinc metal's surface, preventing the acid from reacting further. A scenario like this can be prevented by adding a few copper sulphate crystals to the reaction mixture.
Commercially Produced Hydrogen Gas by Electrolysis of Water
[Click Here for Sample Questions]
Water is electrolyzed in a cell after a tiny amount of acid or alkali is added to make it a good conductor. The cathode in this cell is iron sheet, whereas the anode is nickel plated iron sheet.A diaphragm made of asbestos separates the two electrodes, preventing hydrogen and oxygen from combining. Hydrogen gas is collected at the cathode and dioxygen is collected at the anode when an electric current is passed.
When a 20 percent NaOH ( sodium hydroxide ) solution is used for electrolysis, the water decomposes in the following way:
At cathode
H+ + e‾ ———–> H•
H• + H• ———> H2
At anode
4OH‾ ———> 4OH + e‾
4 OH‾ ——–> 2H2O + O2
Preparation of Pure Hydrogen Gas
[Click Here for Sample Questions]
(1) By the action of pure sulphuric acid on magnesium ribbon
Mg + H2SO4 (dil) ——-> MgSO4 + H2
(2) By the electrolysis of a warm solution of barium hydroxide using platinum or Nickel electrodes.
(3) By the action of water on sodium hydride
NaH + H2O ——–> NaOH + H2
(4) By the action of KOH on scrap aluminium
2Al + 2KOH + 2H2O———-> 2KAlO2 + 3H2
Precautions to Follow When Preparing Hydrogen Gas in Laboratory
[Click Here for Sample Questions]
Before using the device to collect hydrogen gas, steps must be taken to guarantee that all of the air inside the apparatus has been displaced. This is due to the fact that hydrogen gas interacts violently with air.
Also Read: Catalysis
Properties of Hydrogen Gas
[Click Here for Sample Questions]
- It is a colourless, odourless, and tasteless gas.
- It is the lightest material that has ever been discovered.
- It is mildly soluble in water due to its non-polar molecules.
- It has the ability to liquefy at low temperatures and high pressures.
Application of Hydrogen Gas
[Click Here for Sample Questions]
- Many balloons and airships are filled with this gas.
- It's also employed in the hardening of animal and vegetable oils.
- In oxy-hydrogen blow pipes, hydrogen gas is commonly utilised as a fuel.
- In the manufacturing of ammonia, which is used to make a variety of fertilizers.
- In the production of bulk chemicals like methanol, CO(g) + 2H2 (g) ——-> CH3OH (l)
- in the hydrogen chloride manufacturing process, H2 (g ) + Cl2 (g) ——–> 2HCl (g)
- used in the production of metal hydrides
- To convert heavy metal oxides to metal in metallurgy
- for cutting and welding with atomic hydrogen and oxy-hydrogen torches
- In space exploration, liquid hydrogen is employed as a rocket fuel.
- It's also utilised to generate electricity in fuel cells.It produces more energy per unit mass and does not pollute the environment.
Things to Remember
- The laboratory preparation of hydrogen gas generally involves the action of dilute sulphuric acid or dilute hydrochloric acid on zinc granules.
- Before using the device for producing the hydrogen gas, steps must be taken to guarantee that all of the air inside the apparatus has been displaced.
- This is due to the fact that hydrogen gas interacts violently with air.
- Hydrogen gas is a colourless gas that does not have any distinct odour.
- This gas is sparingly soluble in water and the solubility of this gas in water is not affected too much by any changes in temperature.
- This gas is usually used to fill many balloons and airships.
- It is also used for the hardening of animal oils and certain vegetable oils.
Sample Questions
Ques. How many hydrogen-bonded water molecule(s) are there in CuSO4 .5H2O? (2 marks)
Ans. Only one water molecule, which exists outside the brackets i,e.coordination sphere, which is hydrogen-bonded. The other four molecules of water are coordinated.
Ques. Comment on the reactions of dihydrogen with (i) chlorine, (ii) sodium, and (iii) copper(II) oxide (3 marks)
Ans. (i) Dihydrogen reduces chlorine into chloride (Cl– ) ion and itself gets oxidised to H+ ion by chlorine to form hydrogen chloride. An electron pair is shared between H and Cl leading to the formation of a covalent molecule.
(ii) Dihydrogen is reduced by sodium to form NaH. An electron is transferred from Na to H leading to the formation of an ionic compound, Na+ H – .
(iii) Dihydrogen reduces copper(II) oxide to copper in zero oxidation state and itself gets oxidised to H2O, which is a covalent molecule.
Ques. Would you expect the hydrides of N, O and F to have lower boiling points than the hydrides of their subsequent group members ? Give reasons. (3 marks)
Ans. On the basis of molecular masses of NH3 , H2O and HF, their boiling points are expected to be lower than those of the subsequent group member hydrides. However, due to higher electronegativity of N, O and F, the magnitude of hydrogen bonding in their hydrides will be quite appreciable. Hence, the boiling points NH3 , H2O and HF will be higher than the hydrides of their subsequent group members.
Ques. Can phosphorus with outer electronic configuration 3s2 3p3 form PH5 ? (2 marks)
Ans. Although phosphorus exhibits +3 and +5 oxidation states, it cannot form PH5 . Besides some other considerations, high value of dihydrogen and value of hydrogen do not favour the highest oxidation state of P, and consequently the formation of PH5 .
Ques. Calculate the strength of 10 volume solution of hydrogen peroxide. (3 marks)
Ans. 10 volume solution of H2O2 means that 1L of this H2O2 solution will give 10 L of oxygen at STP
2H2O2 → O2 (g) + (1)
2 x 34g 22.7 L at STP
On the basis of above equation 22.7 L of O2 is produced from 68 g H2O2 at STP 10 L of O2 at STP is produced from
68 x 10 /22.7 g = 29.9 g = 30 g H2O2
Therefore, strength of H2O2 in 10 volume H2O2 solution = 30 g/L = 3% of H2O2 solution
Check-Out:







Comments