Kerala Plus Two 2025 Chemistry (SY-625) Model Question Paper with Solutions PDFs are now available for download. The Kerala Plus Two Physics Model Examination 2025 was conducted by the Directorate of Higher Secondary Education (DHSE), Kerala, to help students prepare for the final board exams. The paper is designed as per the latest syllabus and exam pattern prescribed for the academic year 2025–26.
Kerala Plus Two 2025 Chemistry (SY-625) Model Question Paper with solutions
| Kerala Plus Two 2025 Chemistry (SY-625) Model Question Paper | Check Solutions |

Which of the following is the reason for low concentration of oxygen in the blood and tissues of people living at high altitude?
View Solution
Step 1: Understanding conditions at high altitude
At high altitudes, the density of air decreases significantly. This means that the air becomes thinner as we move higher above sea level.
Step 2: Role of atmospheric pressure
Atmospheric pressure decreases with increase in altitude. Due to low atmospheric pressure, the partial pressure of oxygen in the air also decreases.
Step 3: Effect on oxygen concentration in blood
Because of the reduced partial pressure of oxygen, less oxygen diffuses from the lungs into the blood. As a result, the concentration of oxygen in the blood and body tissues becomes low.
Step 4: Conclusion
The main reason for low oxygen concentration in the blood and tissues of people living at high altitudes is the low atmospheric pressure.
Final Answer:
Low atmospheric pressure Quick Tip: At higher altitudes, lower atmospheric pressure leads to reduced oxygen availability, which can cause altitude sickness.
For the reaction \( \mathrm{CHCl_3 + Cl_2 \rightarrow CCl_4 + HCl} \), the rate law is given by \[ r = k[\mathrm{CHCl_3}][\mathrm{Cl_2}]^{\frac{1}{2}}. \]
What is the order of reaction?
View Solution
Step 1: Understanding the rate law.
The given rate law for the reaction is:
\[ r = k[\mathrm{CHCl_3}]^1[\mathrm{Cl_2}]^{\frac{1}{2}} \]
The powers of the concentration terms represent the order of reaction with respect to each reactant.
Step 2: Order with respect to each reactant.
Order with respect to \( \mathrm{CHCl_3} \) is \(1\).
Order with respect to \( \mathrm{Cl_2} \) is \( \dfrac{1}{2} \).
Step 3: Overall order of reaction.
The overall order of the reaction is the sum of the powers of all reactant concentrations in the rate law:
\[ Overall order = 1 + \frac{1}{2} = \frac{3}{2} \]
Step 4: Conclusion.
Thus, the given reaction is of fractional order.
Final Answer:
\[ \boxed{Order of reaction = \dfrac{3}{2}} \] Quick Tip: The order of a reaction is obtained by adding the exponents of reactant concentrations in the rate law.
Identify the configuration of the transition element, which shows the highest spin-only magnetic moment.
View Solution
Step 1: Understanding spin-only magnetic moment
The spin-only magnetic moment of a transition element depends on the number of unpaired electrons present in its d-orbitals. It is given by the formula: \[ \mu = \sqrt{n(n+2)} \, BM \]
where \( n \) is the number of unpaired electrons.
Step 2: Relation between configuration and unpaired electrons
According to Hund’s rule, electrons occupy degenerate orbitals singly before pairing. Hence, the configuration with the maximum number of unpaired electrons will show the highest magnetic moment.
Step 3: Analysis of the given configurations
\(3d^2\): 2 unpaired electrons
\(3d^5\): 5 unpaired electrons (maximum possible in d-subshell)
\(3d^7\): 3 unpaired electrons
\(3d^8\): 2 unpaired electrons
Step 4: Conclusion
The \(3d^5\) configuration has the maximum number of unpaired electrons and therefore shows the highest spin-only magnetic moment.
Final Answer: \(3d^5\) Quick Tip: The spin-only magnetic moment increases with the number of unpaired electrons; the \(d^5\) configuration has the maximum unpaired electrons in the d-subshell.
Analyse the following statements and choose the correct option.
Statement I: Boiling points of alcohols are high.
Statement II: Alcohols can form intermolecular hydrogen bonding.
View Solution
Step 1: Understanding Statement I
Alcohols generally have higher boiling points when compared to hydrocarbons of similar molecular mass. This indicates that stronger intermolecular forces are present between alcohol molecules.
Step 2: Understanding Statement II
Alcohols contain the \(-\mathrm{OH}\) functional group. Due to the high electronegativity of oxygen, alcohol molecules are capable of forming strong intermolecular hydrogen bonds with each other.
Step 3: Linking both statements
Intermolecular hydrogen bonding increases the force of attraction between alcohol molecules. As a result, more energy is required to separate the molecules during boiling, leading to higher boiling points.
Step 4: Conclusion
Both Statement I and Statement II are true, and Statement II correctly explains why alcohols have high boiling points.
Final Answer:
Both statements are true and Statement II is the correct explanation of Statement I Quick Tip: The presence of intermolecular hydrogen bonding significantly increases boiling points of compounds like alcohols.
How many monosaccharide units are present in the sugar present in milk?
View Solution
Step 1: Identify the Sugar Present in Milk.
The sugar present in milk is known as lactose. Lactose is commonly referred to as milk sugar.
Step 2: Nature of Lactose.
Lactose is a disaccharide. It is formed by the combination of two monosaccharide units.
Step 3: Composition of Lactose.
Lactose consists of one molecule of glucose and one molecule of galactose joined together by a glycosidic bond.
Step 4: Conclusion.
Since lactose is made up of two monosaccharide units, the number of monosaccharide units present in the sugar found in milk is two.
Quick Tip: Disaccharides are carbohydrates formed by the combination of two monosaccharides.
When raisins are kept in water, they swell in size. Name the phenomenon responsible for it and give any one application of the phenomenon.
View Solution
When raisins are kept in water, they swell in size due to the phenomenon called osmosis.
More specifically, this process is known as endosmosis.
Osmosis is the process by which water (or solvent) molecules move from a region of higher concentration of water (dilute solution) to a region of lower concentration of water (concentrated solution) through a semi-permeable membrane.
The outer covering of raisins acts as a semi-permeable membrane.
When raisins are placed in pure water, the concentration of water outside the raisin is higher than that inside the raisin.
As a result, water molecules move into the raisin through the semi-permeable membrane.
This inward movement of water causes the raisins to absorb water and swell in size.
This type of osmosis, where water enters the cell or object, is called endosmosis.
Application of Osmosis:
Osmosis plays an important role in the absorption of water by plant roots.
Root hair cells absorb water from the soil by osmosis, which is essential for the growth, nutrition, and survival of plants.
Quick Tip: Swelling of raisins in water is a classic example of endosmosis, a type of osmosis.
A galvanic cell has a cell potential of 1.1 V. If an opposing potential of 1.1 V is applied to this cell, what will happen to the cell reaction and the current flowing through the cell?
View Solution
A galvanic cell converts chemical energy into electrical energy due to a spontaneous redox reaction occurring inside the cell. The flow of current depends on the potential difference between the cell and any external opposing potential applied to it.
Step 1: Understanding Cell Potential.
The cell potential of a galvanic cell represents the maximum electrical potential difference that can be obtained from the spontaneous chemical reaction. In this case, the cell potential is given as \( 1.1 \, V \).
Step 2: Effect of Applying an Opposing Potential.
When an external potential is applied in a direction opposite to the cell potential, it tends to oppose the spontaneous flow of electrons. The effective potential driving the current is the difference between the cell potential and the applied opposing potential.
Step 3: Condition When Opposing Potential Equals Cell Potential.
Here, the applied opposing potential is exactly equal to the cell potential, i.e., \( 1.1 \, V \). Therefore, the net potential difference across the cell becomes zero.
\[ Net potential = 1.1 \, V - 1.1 \, V = 0 \]
Step 4: Effect on Cell Reaction.
When the net potential is zero, the cell reaction neither proceeds forward nor backward. The chemical reaction reaches equilibrium. As a result, there is no further conversion of chemical energy into electrical energy.
Step 5: Effect on Current Flow.
Since there is no net driving force for the movement of electrons, no current flows through the external circuit. The current becomes zero under this condition.
Step 6: Final Conclusion.
When an opposing potential equal to the cell potential is applied, the galvanic cell stops functioning. The cell reaction comes to equilibrium, and the current flowing through the cell becomes zero.
Quick Tip: When the applied opposing potential equals the cell potential, the net emf becomes zero and no current flows.
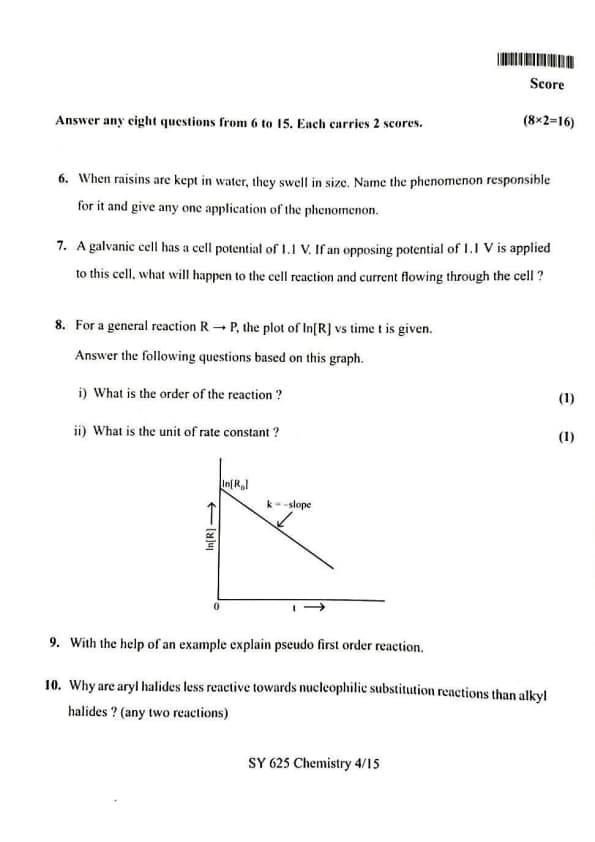
For a general reaction \( R \rightarrow P \), the plot of \(\ln[R]\) versus time \(t\) is given. Answer the following questions based on this graph:
(i) What is the order of the reaction?
(ii) What is the unit of rate constant?

View Solution
(i) Order of the Reaction
Step 1: Interpretation of the given graph.
The graph shows a straight line when \(\ln[R]\) is plotted against time \(t\). The slope of the straight line is negative and is equal to \(-k\), where \(k\) is the rate constant.
Step 2: Relation with integrated rate law.
For a first order reaction, the integrated rate law is given by:
\[ \ln[R] = \ln[R_0] - kt \]
This equation represents a straight line with slope \(-k\) and intercept \(\ln[R_0]\) when \(\ln[R]\) is plotted against time.
Step 3: Conclusion.
Since the given graph is a straight line for \(\ln[R]\) versus \(t\), the reaction follows first order kinetics.
Answer (i):
\[ \boxed{The reaction is of first order} \]
(ii) Unit of Rate Constant
Step 4: Unit of rate constant for first order reaction.
For a first order reaction, the unit of rate constant \(k\) is inverse of time.
Step 5: SI unit.
The SI unit of time is second. Therefore, the SI unit of rate constant is:
\[ s^{-1} \]
Answer (ii):
\[ \boxed{Unit of rate constant = s^{-1}} \] Quick Tip: A straight line graph of \(\ln[R]\) versus time always indicates a first order reaction.
With the help of an example, explain pseudo first order reaction.
View Solution
Step 1: Meaning of Pseudo First Order Reaction.
A pseudo first order reaction is a reaction which is actually of higher order but appears to follow first order kinetics under certain conditions. This happens when one of the reactants is present in very large excess compared to the other reactant. Due to its large excess, the concentration of that reactant remains almost constant throughout the reaction.
Step 2: Reason for First Order Behavior.
Since the concentration of the excess reactant does not change appreciably, it is treated as a constant. As a result, the rate of reaction depends only on the concentration of the limiting reactant, making the reaction appear as a first order reaction.
Step 3: Example – Hydrolysis of Ethyl Acetate.
A common example of a pseudo first order reaction is the acid-catalyzed hydrolysis of ethyl acetate:
\[ CH_3COOC_2H_5 + H_2O \rightarrow CH_3COOH + C_2H_5OH \]
In this reaction, water is taken in large excess compared to ethyl acetate.
Step 4: Rate Law Explanation.
The actual rate law for the reaction is:
\[ Rate = k[CH_3COOC_2H_5][H_2O] \]
Since water is present in large excess, its concentration remains constant. Therefore, the rate law becomes:
\[ Rate = k'[CH_3COOC_2H_5] \]
where \( k' = k[H_2O] \). This shows first order dependence on ethyl acetate.
Step 5: Conclusion.
Although the reaction is actually of second order, it behaves like a first order reaction due to the large excess of one reactant. Hence, it is called a pseudo first order reaction.
Quick Tip: Pseudo first order reactions appear first order because one reactant is present in large excess.
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides? (Any two reasons)
View Solution
Aryl halides are much less reactive towards nucleophilic substitution reactions compared to alkyl halides due to their structural and electronic characteristics.
Step 1: Partial Double Bond Character of C–X Bond.
In aryl halides, the halogen atom is directly attached to an aromatic ring. Due to resonance, the lone pair of electrons on the halogen overlaps with the π-electron system of the benzene ring. This gives the carbon–halogen bond a partial double bond character.
As a result, the C–X bond becomes shorter, stronger, and more stable, making it difficult for nucleophiles to break the bond.
Step 2: Instability of Aryl Carbocation.
Nucleophilic substitution reactions such as \( SN1 \) involve the formation of a carbocation intermediate. In aryl halides, formation of an aryl carbocation is highly unstable because it disrupts the aromaticity of the benzene ring. Therefore, \( SN1 \) reactions do not occur easily.
Step 3: Difficulty of Backside Attack.
For \( SN2 \) reactions, a backside attack by the nucleophile is required. In aryl halides, the planar structure of the benzene ring and the presence of delocalized π-electrons create steric and electronic hindrance, preventing effective backside attack by nucleophiles.
Step 4: Conclusion.
Due to strong C–X bond, resonance stabilization, and instability of aryl carbocations, aryl halides are much less reactive towards nucleophilic substitution reactions compared to alkyl halides.
Quick Tip: Aryl halides resist nucleophilic substitution because of resonance stabilization and strong C–X bonding.
Explain the manufacture of ethanol from molasses.
View Solution
Step 1: Meaning of Molasses.
Molasses is a dark brown, viscous liquid obtained as a by-product during the manufacture of sugar from sugarcane. It contains a high percentage of fermentable sugars such as sucrose, glucose, and fructose, which are suitable for ethanol production.
Step 2: Dilution of Molasses.
Molasses is first diluted with water to reduce the sugar concentration to about 8–10%. This dilution is necessary to provide suitable conditions for fermentation.
Step 3: Fermentation Process.
The diluted molasses is transferred to fermentation tanks and yeast (Saccharomyces cerevisiae) is added. The fermentation is carried out at a temperature of about 30–35\(^\circ\)C in the absence of air.
During fermentation, sugars present in molasses are converted into ethanol and carbon dioxide as shown below:
\[ \mathrm{C_6H_{12}O_6 \xrightarrow{yeast} 2C_2H_5OH + 2CO_2} \]
Step 4: Distillation.
After fermentation, the fermented liquid (wash) contains about 8–10% ethanol. This mixture is subjected to fractional distillation to separate ethanol from water and other impurities.
Step 5: Rectification.
The distillate obtained is further purified to obtain rectified spirit containing about 95% ethanol. Absolute alcohol can be prepared by removing the remaining water using suitable drying agents.
Step 6: Conclusion.
Thus, ethanol is manufactured from molasses by dilution, fermentation using yeast, followed by distillation and rectification.
Quick Tip: Molasses is the most economical raw material for the large-scale production of ethanol.
Match the following:

View Solution
Step 1: Understanding each reaction or process.
(i) Conversion of phenol to salicylic acid
This conversion is carried out by Kolbe’s reaction, in which sodium phenoxide reacts with carbon dioxide followed by acidification to give salicylic acid.
(ii) Reaction of alkyl halide with sodium alkoxide
This reaction is known as Williamson’s synthesis and is used for the preparation of ethers.
(iii) Manufacture of phenol
Phenol is manufactured on an industrial scale by the Cumene process.
(iv) Conversion of ketone to 2\(^\circ\) alcohol
This conversion is achieved using Grignard reagents, which add to the carbonyl group of ketones to form secondary alcohols after hydrolysis.
Step 2: Correct Matching.
\[ \begin{array}{c c} Column I & Column II
\hline (i) & (b) Kolbe’s reaction
(ii) & (c) Williamson’s synthesis
(iii) & (d) Cumene
(iv) & (a) Grignard reagents
\end{array} \]
Step 3: Conclusion.
Thus, each reaction or process in Column I is correctly matched with its corresponding name or reagent in Column II.
Quick Tip: Remember: Kolbe for salicylic acid, Williamson for ethers, Cumene for phenol, and Grignard for alcohol formation.
Identify the product obtained in the following reaction:

View Solution
Step 1: Nature of the Reactant.
Phenol is an activated aromatic compound due to the presence of the hydroxyl (\(-OH\)) group, which is an electron-donating group. This group strongly activates the benzene ring towards electrophilic substitution reactions and directs incoming substituents to the ortho and para positions.
Step 2: Nature of the Reagent.
Concentrated nitric acid is a strong nitrating agent. In the presence of such a strong nitrating condition, multiple nitration occurs instead of mono-nitration.
Step 3: Reaction Outcome.
When phenol reacts with concentrated nitric acid, nitration occurs at all the activated positions of the benzene ring, leading to the formation of trinitro phenol.
Step 4: Identification of the Product.
The product formed is 2,4,6-trinitrophenol, commonly known as picric acid.
Final Answer.
Thus, the product obtained in the reaction is:
\[ \boxed{Picric acid (2,4,6-trinitrophenol)} \]
Quick Tip: Strong activating groups like \(-OH\) cause multiple substitution when reacted with strong electrophiles.
Explain Rosenmund reduction for the preparation of an aldehyde.
View Solution
Step 1: Definition of Rosenmund Reduction.
Rosenmund reduction is a method used for the preparation of aldehydes from acid chlorides. In this reaction, an acid chloride is reduced to the corresponding aldehyde without further reduction to alcohol.
Step 2: Reagents Used.
The reduction is carried out using hydrogen gas in the presence of a poisoned palladium catalyst. The catalyst used is palladium deposited on barium sulphate (\( Pd/BaSO_4 \)), often poisoned with sulphur or quinoline to control the reaction.
Step 3: Reason for Using a Poisoned Catalyst.
The poisoned catalyst reduces the activity of palladium, preventing the aldehyde from being further reduced to the corresponding alcohol.
Step 4: General Reaction.
\[ RCOCl + H_2 \xrightarrow{Pd/BaSO_4} RCHO + HCl \]
Step 5: Importance of the Reaction.
Rosenmund reduction is an important laboratory method for selectively preparing aldehydes from acid chlorides.
Final Answer.
Thus, Rosenmund reduction converts an acid chloride into an aldehyde using hydrogen gas and a poisoned palladium catalyst.
Quick Tip: Rosenmund reduction stops at the aldehyde stage due to the use of a poisoned catalyst.
\( \mathrm{CH_3CH_2NH_2} \) is more basic than \( \mathrm{C_6H_5NH_2} \). Give reason.
View Solution
The basic nature of amines depends on the availability of the lone pair of electrons on the nitrogen atom.
A base is a substance that can donate a lone pair of electrons to accept a proton.
In \( \mathrm{CH_3CH_2NH_2} \) (ethylamine), the ethyl group (\( \mathrm{CH_3CH_2{-}} \)) shows a strong +I (electron-donating inductive) effect.
This +I effect pushes electron density towards the nitrogen atom, increasing the availability of its lone pair of electrons.
As a result, ethylamine can accept a proton more easily and hence shows higher basicity.
On the other hand, in \( \mathrm{C_6H_5NH_2} \) (aniline), the lone pair of electrons on the nitrogen atom is involved in resonance with the benzene ring.
Due to resonance, the lone pair gets delocalized over the aromatic ring and becomes less available for protonation.
This decreases the basic strength of aniline.
Therefore, due to the +I effect in ethylamine and resonance effect in aniline, \( \mathrm{CH_3CH_2NH_2} \) is more basic than \( \mathrm{C_6H_5NH_2} \).
Quick Tip: Greater availability of the nitrogen lone pair increases the basic strength of amines.
When 1.00 g of a nonelectrolyte solute is dissolved in 50 g of benzene, the freezing point of benzene is lowered by 0.40 K. Give reason.
View Solution
Step 1: Concept of Freezing Point Depression.
Freezing point depression is a colligative property of solutions. It depends only on the number of solute particles present in the solution and not on the nature of the solute.
Step 2: Effect of Nonelectrolyte Solute.
A nonelectrolyte solute does not dissociate into ions when dissolved in a solvent. However, its particles still interfere with the regular arrangement of solvent molecules required for solidification.
Step 3: Reason for Lowering of Freezing Point.
When a nonelectrolyte solute is dissolved in benzene, the vapour pressure of the solution becomes lower than that of pure benzene. As a result, benzene now freezes at a lower temperature than its normal freezing point.
Step 4: Conclusion.
Thus, the freezing point of benzene is lowered by 0.40 K due to the presence of solute particles which hinder the freezing process, showing the phenomenon of freezing point depression.
Quick Tip: Freezing point depression is a colligative property and depends only on the number of solute particles.
Find the molar mass of the above solute, if the freezing point depression constant of benzene is \(5.12 \, K kg mol^{-1}\).
View Solution
Step 1: Given data.
Mass of solute, \( w_2 = 1.00 \, g \)
Mass of solvent (benzene), \( w_1 = 50 \, g = 0.050 \, kg \)
Freezing point depression, \( \Delta T_f = 0.40 \, K \)
Freezing point depression constant, \( K_f = 5.12 \, K kg mol^{-1} \)
Step 2: Formula for freezing point depression.
\[ \Delta T_f = K_f \times m \]
where \( m \) is the molality of the solution.
Step 3: Expression for molality.
\[ m = \frac{w_2}{M} \times \frac{1000}{w_1} \]
where \( M \) is the molar mass of the solute.
Step 4: Substitution into the formula.
\[ 0.40 = 5.12 \times \frac{1.00}{M} \times \frac{1000}{50} \]
Step 5: Simplification.
\[ 0.40 = 5.12 \times \frac{20}{M} \] \[ 0.40 = \frac{102.4}{M} \]
Step 6: Calculation of molar mass.
\[ M = \frac{102.4}{0.40} \] \[ M = 256 \, g mol^{-1} \]
Step 7: Final Answer.
Thus, the molar mass of the solute is:
\[ \boxed{256 \, g mol^{-1}} \]
Quick Tip: Use freezing point depression to determine molar mass of nonelectrolyte solutes.
Classify the following as primary and secondary batteries:
Lead storage battery, Mercury cell, Nickel-Cadmium cell, Dry cell
View Solution
Step 1: Meaning of Primary and Secondary Batteries.
Primary batteries are those batteries in which the chemical reaction is irreversible. Once the chemicals are exhausted, these batteries cannot be recharged.
Secondary batteries are those batteries in which the chemical reaction is reversible. These batteries can be recharged and used again.
Step 2: Classification of Given Batteries.
Primary Batteries:
(a) Dry cell: The chemical reaction is irreversible, so it cannot be recharged.
(b) Mercury cell: It is a primary cell used in devices like watches and hearing aids and cannot be recharged.
Secondary Batteries:
(a) Lead storage battery: It can be recharged and is commonly used in automobiles.
(b) Nickel-Cadmium cell: It is a rechargeable battery widely used in electronic devices.
Step 3: Final Classification.
Primary batteries: Dry cell, Mercury cell
Secondary batteries: Lead storage battery, Nickel-Cadmium cell
Quick Tip: Rechargeable batteries are always secondary batteries.
What advantage do the fuel cells have over primary and secondary batteries?
View Solution
Step 1: Understanding Fuel Cells.
Fuel cells are electrochemical devices that convert the chemical energy of a fuel directly into electrical energy. As long as fuel and oxidant are supplied continuously, fuel cells can produce electricity.
Step 2: Advantage over Primary Batteries.
Unlike primary batteries, fuel cells do not get exhausted because fresh fuel is continuously supplied. Therefore, they do not need replacement after use.
Step 3: Advantage over Secondary Batteries.
Unlike secondary batteries, fuel cells do not require recharging. They can operate continuously without interruption as long as fuel is available.
Step 4: Additional Advantages.
Fuel cells have higher efficiency, produce less pollution, and provide a constant voltage output. They are environmentally friendly as the by-products are usually water and heat.
Step 5: Conclusion.
Thus, the main advantage of fuel cells over primary and secondary batteries is their continuous operation without the need for replacement or recharging, along with higher efficiency and lower environmental impact.
Quick Tip: Fuel cells generate electricity as long as fuel is supplied.
What is the effect of a catalyst on the activation energy of a reaction?
View Solution
A catalyst is a substance which increases the rate of a chemical reaction without itself undergoing any permanent chemical change.
The main effect of a catalyst is on the activation energy of the reaction.
A catalyst provides an alternative reaction pathway having lower activation energy compared to the uncatalysed reaction.
Due to this decrease in activation energy, a larger number of reacting molecules possess sufficient energy to cross the energy barrier and form products.
As a result, the rate of reaction increases.
It is important to note that a catalyst does not change the overall energy of reactants and products; it only lowers the energy of the transition state.
Thus, a catalyst speeds up a reaction by lowering its activation energy.
Quick Tip: A catalyst increases reaction rate by providing a pathway with lower activation energy.
The rate constants of a reaction at 500 K and 700 K are 0.02 s\(^{-1}\) and 0.07 s\(^{-1}\) respectively. Calculate the value of activation energy.
View Solution
Given:
Rate constant at temperature \( T_1 = 500 \, K \), \( k_1 = 0.02 \, s^{-1} \)
Rate constant at temperature \( T_2 = 700 \, K \), \( k_2 = 0.07 \, s^{-1} \)
Gas constant, \( R = 8.314 \, J mol^{-1}K^{-1} \)
The Arrhenius equation relating rate constants and temperature is:
\[ \ln \left( \frac{k_2}{k_1} \right) = \frac{E_a}{R} \left( \frac{1}{T_1} - \frac{1}{T_2} \right) \]
Substituting the given values:
\[ \ln \left( \frac{0.07}{0.02} \right) = \frac{E_a}{8.314} \left( \frac{1}{500} - \frac{1}{700} \right) \]
\[ \ln (3.5) = \frac{E_a}{8.314} \left( \frac{700 - 500}{500 \times 700} \right) \]
\[ 1.2528 = \frac{E_a}{8.314} \times \frac{200}{350000} \]
\[ 1.2528 = \frac{E_a}{8.314} \times 0.0005714 \]
\[ E_a = \frac{1.2528 \times 8.314}{0.0005714} \]
\[ E_a \approx 18237 \, J mol^{-1} \]
\[ E_a \approx 18.24 \, kJ mol^{-1} \]
Thus, the activation energy of the reaction is approximately \( 18.24 \, kJ mol^{-1} \).
Quick Tip: Arrhenius equation helps determine activation energy using rate constants at two different temperatures.
When a chromite ore is fused with sodium carbonate in excess of air and the product is dissolved in water, a yellow solution of compound (A) is obtained. After treatment of this yellow solution with sulphuric acid, compound (B) can be crystallised from the solution. When compound (B) is treated with KCl, orange crystals of compound (C) crystallise out. Identify A to C and write their formulae.
View Solution
This question is based on the metallurgy and chemistry of chromium compounds. The reactions involve oxidation, acidification, and salt formation, leading to characteristic color changes that help identify the compounds.
Step 1: Formation of Compound (A).
Chromite ore has the formula \( FeCr_2O_4 \). When chromite ore is fused with sodium carbonate (\( Na_2CO_3 \)) in the presence of excess air, chromium(III) is oxidized to chromium(VI), forming sodium chromate.
The reaction can be represented as:
\[ 4FeCr_2O_4 + 8Na_2CO_3 + 7O_2 \rightarrow 8Na_2CrO_4 + 2Fe_2O_3 + 8CO_2 \]
Sodium chromate dissolves in water to give a yellow solution.
Therefore, Compound (A) is sodium chromate.
\[ \textbf{A = Sodium chromate, } Na_2CrO_4 \]
Step 2: Formation of Compound (B).
When the yellow solution of sodium chromate is treated with sulphuric acid, sodium dichromate is formed. This occurs due to acidification, which converts chromate ions into dichromate ions.
The reaction is:
\[ 2Na_2CrO_4 + H_2SO_4 \rightarrow Na_2Cr_2O_7 + Na_2SO_4 + H_2O \]
Sodium dichromate crystallises out from the solution.
Therefore, Compound (B) is sodium dichromate.
\[ \textbf{B = Sodium dichromate, } Na_2Cr_2O_7 \]
Step 3: Formation of Compound (C).
When sodium dichromate is treated with potassium chloride, a double displacement reaction takes place. Due to the lower solubility of potassium dichromate, it crystallises out as orange crystals.
The reaction is:
\[ Na_2Cr_2O_7 + 2KCl \rightarrow K_2Cr_2O_7 + 2NaCl \]
Potassium dichromate is known for its characteristic orange colour.
Therefore, Compound (C) is potassium dichromate.
\[ \textbf{C = Potassium dichromate, } K_2Cr_2O_7 \]
Step 4: Summary of Identified Compounds.
\[ \textbf{A: Sodium chromate } (Na_2CrO_4)
\textbf{B: Sodium dichromate } (Na_2Cr_2O_7)
\textbf{C: Potassium dichromate } (K_2Cr_2O_7) \] Quick Tip: Chromate ions give a yellow colour, while dichromate ions give an orange colour—this color change is a key identification test.
Give reason for the following:
(i) The enthalpies of atomisation of the transition metals are high.
(ii) The transition metals generally form coloured compounds.
(iii) Although Zr belongs to the 4d series and Hf belongs to the 5d transition series, it is quite difficult to separate them.
View Solution
(i) The enthalpies of atomisation of the transition metals are high.
Step 1: Meaning of enthalpy of atomisation.
The enthalpy of atomisation is the energy required to convert one mole of a metal in its solid state into gaseous atoms.
Step 2: Nature of bonding in transition metals.
Transition metals have a large number of unpaired electrons in their \(d\)-orbitals along with \(s\)-electrons. These electrons participate effectively in metallic bonding.
Step 3: Reason.
Due to strong metallic bonding arising from the involvement of both \(s\) and \(d\) electrons, a large amount of energy is required to separate the atoms of transition metals.
Step 4: Conclusion.
Hence, transition metals have high enthalpies of atomisation.
(ii) The transition metals generally form coloured compounds.
Step 1: Presence of partially filled \(d\)-orbitals.
Most transition metal ions possess partially filled \(d\)-orbitals.
Step 2: Electronic transitions.
When visible light falls on a transition metal compound, electrons from lower energy \(d\)-orbitals are excited to higher energy \(d\)-orbitals. This process is known as \(d\)-\(d\) transition.
Step 3: Absorption of visible light.
The energy required for these electronic transitions corresponds to the energy of visible light. As a result, certain wavelengths are absorbed and the complementary colour is observed.
Step 4: Conclusion.
Therefore, transition metals generally form coloured compounds due to \(d\)-\(d\) electronic transitions.
(iii) Although Zr belongs to the 4d series and Hf belongs to the 5d transition series, it is quite difficult to separate them.
Step 1: Atomic radii comparison.
Zirconium (Zr) and Hafnium (Hf) have nearly the same atomic and ionic radii.
Step 2: Reason for similarity – Lanthanide contraction.
The poor shielding effect of \(4f\)-electrons in the lanthanide series causes a gradual decrease in atomic size, known as lanthanide contraction. As a result, the size of Hf becomes almost identical to that of Zr.
Step 3: Similar chemical properties.
Due to their nearly identical sizes, Zr and Hf exhibit very similar chemical properties and form compounds of similar nature.
Step 4: Conclusion.
Hence, separation of Zr and Hf becomes difficult because of their similar atomic sizes and chemical behaviour caused by lanthanide contraction.
Quick Tip: Stronger metallic bonding leads to higher enthalpy of atomisation.
Write the structural formula and IUPAC name of the compound.
View Solution
Step 1: Interpretation of the given reactions.
The compound reacts with \( \mathrm{AgNO_3} \) to give a white precipitate, which confirms the presence of free chloride ions outside the coordination sphere, since white \( \mathrm{AgCl} \) is formed.
The compound does not react with \( \mathrm{BaCl_2} \), indicating that sulphate ions are not present as free ions in solution and hence are coordinated to the central metal ion.
Step 2: Writing the coordination formula.
Since one chloride ion is present outside the coordination sphere and the sulphate ion is coordinated to cobalt along with five ammonia molecules, the structural formula of the compound is:
\[ \boxed{[\mathrm{Co(NH_3)_5SO_4}]Cl} \]
Step 3: IUPAC name of the compound.
The correct IUPAC name of the compound is:
\[ \boxed{Pentaammine(sulfato)cobalt(III) chloride} \]
Quick Tip: A white precipitate with \( \mathrm{AgNO_3} \) confirms the presence of free chloride ions.
What is the coordination number of the central metal ion in the compound?
View Solution
Step 1: Definition of coordination number.
The coordination number of a central metal ion is defined as the total number of donor atoms directly bonded to it.
Step 2: Identification of ligands.
In the complex \( [\mathrm{Co(NH_3)_5SO_4}]^+ \):
Five ammonia molecules act as monodentate ligands.
The sulphate ion coordinates through one oxygen atom.
Step 3: Calculation of coordination number.
Total number of donor atoms attached to cobalt:
\[ 5 (from NH_3) + 1 (from SO_4^{2-}) = 6 \]
Final Answer.
The coordination number of the central metal ion is:
\[ \boxed{6} \]
Quick Tip: Cobalt(III) complexes commonly show coordination number six.
Using valence bond theory, predict the following in relation to the complex \([Co(NH_3)_6]^{3+}\):
(i) Type of hybridisation
(ii) Whether inner or outer orbital complex
(iii) Magnetic behaviour
View Solution
The given complex is \([Co(NH_3)_6]^{3+}\).
Here, cobalt is the central metal ion and ammonia (\(NH_3\)) acts as a neutral ligand.
Step 1: Oxidation state and electronic configuration of cobalt
The oxidation state of cobalt in the complex is \(+3\).
Atomic number of cobalt \(= 27\).
Electronic configuration of Co: \([Ar]\,3d^7\,4s^2\).
For \(Co^{3+}\), three electrons are removed:
\[ Co^{3+} : [Ar]\,3d^6 \]
Step 2: Nature of ligand and pairing of electrons
Ammonia (\(NH_3\)) is a strong field ligand.
Strong field ligands cause pairing of electrons in the \(3d\) orbitals.
Thus, all six electrons in the \(3d\) orbitals of \(Co^{3+}\) get paired.
Step 3: Type of hybridisation
After pairing, two \(3d\), one \(4s\), and three \(4p\) orbitals hybridise.
Hence, the hybridisation is:
\[ d^2sp^3 \]
This hybridisation corresponds to an octahedral geometry.
Step 4: Inner or outer orbital complex
Since inner \(3d\) orbitals are used for hybridisation, the complex is an inner orbital complex.
Step 5: Magnetic behaviour
As all electrons are paired in the \(3d\) orbitals, there are no unpaired electrons.
Therefore, the complex is diamagnetic in nature.
% Final Results
Final Results:
(i) Type of hybridisation: \( d^2sp^3 \)
(ii) Nature of complex: Inner orbital complex
(iii) Magnetic behaviour: Diamagnetic
Quick Tip: Strong field ligands like \(NH_3\) cause pairing of electrons, leading to inner orbital and diamagnetic complexes.
Write the structure of the major organic product in each of the following reactions:
(i) \(CH_3CH_2CH_2Cl + NaI \xrightarrow[heat]{acetone} \)
View Solution
Step 1: Nature of the Reaction.
This reaction is an example of the Finkelstein reaction, which involves halogen exchange. Alkyl chlorides react with sodium iodide in acetone to form alkyl iodides.
Step 2: Reason for Reaction Direction.
Sodium chloride formed in the reaction is insoluble in acetone and precipitates out. This drives the reaction forward according to Le Chatelier’s principle.
Step 3: Major Product.
Chlorine atom in propyl chloride is replaced by iodine.
\[ CH_3CH_2CH_2Cl + NaI \rightarrow CH_3CH_2CH_2I + NaCl \]
Major product:
\[ \boxed{CH_3CH_2CH_2I} \]
Write the structure of the major organic product in each of the following reactions:
\(CH_3CH_2CH_2OH + SOCl_2 \rightarrow\)
View Solution
Step 1: Identification of Reagents.
Thionyl chloride (\(SOCl_2\)) is used to convert alcohols into alkyl chlorides. The reaction proceeds smoothly with the evolution of gaseous by-products.
Step 2: Reaction Mechanism.
The hydroxyl group of the alcohol is replaced by a chlorine atom. Sulphur dioxide (\(SO_2\)) and hydrogen chloride (\(HCl\)) gases are released, making the reaction irreversible.
Step 3: Major Product.
Propanol is converted into propyl chloride.
\[ CH_3CH_2CH_2OH + SOCl_2 \rightarrow CH_3CH_2CH_2Cl + SO_2 + HCl \]
Major product:
\[ \boxed{CH_3CH_2CH_2Cl} \]
Write the structure of the major organic product in each of the following reactions:
\(CH_3CH_2CH=CH_2 + HBr \xrightarrow{peroxide}\)
View Solution
Step 1: Identification of Special Condition.
The presence of peroxide indicates that the reaction follows the free radical mechanism instead of the usual ionic mechanism. This is known as the peroxide effect or Kharasch effect.
Step 2: Regioselectivity of Addition.
Under peroxide conditions, HBr adds to an unsymmetrical alkene in an anti-Markovnikov manner. The bromine atom attaches to the carbon with more hydrogen atoms.
Step 3: Major Product.
In 1-butene, bromine gets attached to the terminal carbon atom.
\[ CH_3CH_2CH=CH_2 + HBr \rightarrow CH_3CH_2CH_2CH_2Br \]
Major product:
\[ \boxed{CH_3CH_2CH_2CH_2Br} \] Quick Tip: HBr in the presence of peroxide always follows anti-Markovnikov addition.
Choose the correct reagents from the box given below for the following conversion reactions:
Reagents:
![]()
View Solution
Each conversion requires a specific reagent based on the nature of the functional group transformation involved.
(i) A carboxylic acid to \(\alpha\)-halocarboxylic acid.
This conversion is carried out by the Hell–Volhard–Zelinsky (HVZ) reaction. In this reaction, a carboxylic acid is treated with bromine in the presence of red phosphorus, leading to halogenation at the \(\alpha\)-carbon atom.
Correct reagent:
\[ \boxed{Br_2/red P} \]
(ii) An unsaturated ester to aldehyde.
DIBAL-H (Diisobutylaluminium hydride) is a selective reducing agent that reduces esters to aldehydes when used at low temperature followed by hydrolysis. It does not further reduce the aldehyde to alcohol under controlled conditions.
Correct reagent:
\[ \boxed{DIBAL-H/H_2O} \]
(iii) A ketone to hydrocarbon.
This conversion involves the reduction of the carbonyl group of a ketone to a methylene group. The Clemmensen reduction achieves this using zinc amalgam and concentrated hydrochloric acid.
Correct reagent:
\[ \boxed{Zn-Hg/HCl} \] Quick Tip: Remember key name reactions:
HVZ reaction for \(\alpha\)-halogenation,
DIBAL-H for selective ester reduction,
Clemmensen reduction for converting carbonyls to hydrocarbons.
Write the name and one use of azo compound obtained when benzene diazonium chloride reacts with phenol.
Write the name of the above reaction.
View Solution
(i) Step 1: Reaction involved.
When benzene diazonium chloride reacts with phenol in alkaline medium, an azo coupling reaction takes place forming an azo compound.
Step 2: Name of the azo compound.
The azo compound formed is p-hydroxyazobenzene.
Step 3: Use of the azo compound.
p-Hydroxyazobenzene is used as a dye in colouring textiles, leather, paper, and inks.
Final Answer:
Name: p-hydroxyazobenzene
Use: Used as a dye in textile and colouring industries
(ii) Step 1: Identification of the reaction.
The reaction between benzene diazonium chloride and phenol leading to the formation of an azo compound is known as an azo coupling reaction.
Step 2: Conclusion.
Thus, the name of the reaction is Azo coupling reaction.
Quick Tip: Azo compounds are brightly coloured and widely used as dyes.
Write any three differences between RNA and DNA.
View Solution
Step 1: Difference in Sugar.
RNA contains ribose sugar, whereas DNA contains deoxyribose sugar.
Step 2: Difference in Nitrogenous Bases.
RNA contains uracil instead of thymine, while DNA contains thymine instead of uracil.
Step 3: Structural Difference.
RNA is usually single-stranded, whereas DNA is double-stranded and forms a double helix structure.
Step 4: Functional Difference.
RNA plays a role in protein synthesis, whereas DNA is responsible for the storage and transmission of genetic information.
Quick Tip: DNA stores genetic information, while RNA helps in its expression.
What general name is given to binary mixtures which show deviation from Raoult’s law? How many types of such mixtures are there?
View Solution
Step 1: General name of such mixtures.
Binary mixtures which do not obey Raoult’s law over the entire range of composition are known as non-ideal solutions. These solutions show either positive or negative deviation from Raoult’s law.
Step 2: Types of deviations.
There are two types of deviations shown by non-ideal solutions:
(a) Positive deviation from Raoult’s law
(b) Negative deviation from Raoult’s law
Step 3: Conclusion.
Thus, binary mixtures that deviate from Raoult’s law are called non-ideal solutions, and they are of two types based on the nature of deviation.
Quick Tip: Ideal solutions strictly obey Raoult’s law, while non-ideal solutions deviate from it.
What type of deviation is shown by a mixture of chloroform and acetone? Explain the reason for the deviation and draw a graph representing it.
View Solution
Step 1: Type of deviation.
A mixture of chloroform and acetone shows negative deviation from Raoult’s law.
Step 2: Reason for the deviation.
In pure chloroform and pure acetone, the intermolecular attractions are relatively weaker. When chloroform and acetone are mixed, strong intermolecular interactions are formed between the hydrogen atom of chloroform and the oxygen atom of acetone due to hydrogen bonding.
These stronger interactions reduce the escaping tendency of molecules into the vapour phase, thereby lowering the vapour pressure below the value predicted by Raoult’s law.
Step 3: Effect on Vapour Pressure.
Because of strong attractive forces, the total vapour pressure of the solution is less than that of an ideal solution, resulting in negative deviation.
Step 4: Graphical Representation.
In the graph of vapour pressure versus mole fraction, the curve for the solution lies below the straight line predicted by Raoult’s law, showing a minimum in vapour pressure.
Step 5: Conclusion.
Thus, chloroform–acetone mixture shows negative deviation from Raoult’s law due to strong intermolecular hydrogen bonding between unlike molecules.
Quick Tip: Negative deviation occurs when attraction between unlike molecules is stronger than like molecules.
What is meant by limiting molar conductivity of an electrolyte?
View Solution
Step 1: Meaning of molar conductivity.
Molar conductivity of an electrolyte is defined as the conductance of all the ions produced by one mole of an electrolyte dissolved in a solution. It depends on the concentration of the electrolyte.
Step 2: Definition of limiting molar conductivity.
The limiting molar conductivity of an electrolyte is the molar conductivity when the concentration of the electrolyte approaches zero, i.e., at infinite dilution.
Step 3: Explanation.
At infinite dilution, the ions are so far apart that interionic attractions become negligible. Hence, each ion moves independently and contributes fully to the conductivity.
Final Definition.
Limiting molar conductivity \( (\Lambda_m^0) \) is the maximum value of molar conductivity attained by an electrolyte at infinite dilution.
Quick Tip: Limiting molar conductivity is independent of concentration.
Write the name and statement of the law used to determine the limiting molar conductivity of an electrolyte.
View Solution
Step 1: Name of the law.
The law used to determine the limiting molar conductivity of an electrolyte is Kohlrausch’s Law of Independent Migration of Ions.
Step 2: Statement of Kohlrausch’s Law.
Kohlrausch’s law states that at infinite dilution, each ion migrates independently of the other ions, and the limiting molar conductivity of an electrolyte is equal to the sum of the limiting ionic conductivities of its constituent ions.
Step 3: Mathematical Expression.
For an electrolyte \( AB \):
\[ \Lambda_m^0 = \lambda_A^0 + \lambda_B^0 \]
Quick Tip: Kohlrausch’s law helps calculate limiting molar conductivity of weak electrolytes.
Calculate the limiting molar conductivity for \( \mathrm{CaCl_2} \). Given \( \lambda^0 \) values for \( \mathrm{Ca^{2+}} \) and \( \mathrm{Cl^-} \) are 119.0 S cm\(^2\) mol\(^{-1}\) and 76.3 S cm\(^2\) mol\(^{-1}\) respectively.
View Solution
Step 1: Write Kohlrausch’s equation for \( \mathrm{CaCl_2} \).
Calcium chloride dissociates as:
\[ \mathrm{CaCl_2 \rightarrow Ca^{2+} + 2Cl^-} \]
Step 2: Apply Kohlrausch’s Law.
\[ \Lambda_m^0 = \lambda^0_{\mathrm{Ca^{2+}}} + 2\lambda^0_{\mathrm{Cl^-}} \]
Step 3: Substitute the given values.
\[ \Lambda_m^0 = 119.0 + 2(76.3) \] \[ \Lambda_m^0 = 119.0 + 152.6 \] \[ \Lambda_m^0 = 271.6 \ S cm^2 mol^{-1} \]
Final Answer.
The limiting molar conductivity of \( \mathrm{CaCl_2} \) is:
\[ \boxed{271.6 \ S cm^2 mol^{-1}} \]
Quick Tip: Multiply the ionic conductivity by the number of ions produced on dissociation.
Explain linkage isomerism in coordination complexes with the help of suitable examples.
View Solution
Linkage isomerism is a type of structural isomerism shown by coordination compounds in which a ligand can coordinate to the central metal atom through two or more different donor atoms.
Such ligands are called ambidentate ligands.
In linkage isomerism, the composition of the complex remains the same, but the point of attachment of the ligand to the metal ion changes.
As a result, different isomers are formed with different physical and chemical properties.
Common ambidentate ligands that show linkage isomerism are \( NO_2^- \), \( SCN^- \), and \( CN^- \).
Example 1:
In the complex \( [Co(NH_3)_5NO_2]Cl_2 \), the nitrite ion (\( NO_2^- \)) can attach to the cobalt ion through nitrogen or oxygen.
When it attaches through nitrogen, the complex is called nitro complex:
\[ [Co(NH_3)_5(NO_2)]Cl_2 \]
When it attaches through oxygen, the complex is called nitrito complex:
\[ [Co(NH_3)_5(ONO)]Cl_2 \]
Example 2:
In thiocyanate complexes, the ligand \( SCN^- \) can coordinate through sulphur or nitrogen, forming thiocyanato and isothiocyanato complexes.
Thus, linkage isomerism arises due to the different modes of attachment of an ambidentate ligand to the same metal ion.
Quick Tip: Linkage isomerism occurs due to ambidentate ligands bonding through different donor atoms.
Draw the geometrical isomers of \( [Pt(NH_3)_2Cl_2] \).
View Solution
The complex \( [Pt(NH_3)_2Cl_2] \) is a square planar complex.
Square planar complexes show geometrical isomerism when similar ligands are arranged differently around the central metal atom.
In this complex, platinum is in the +2 oxidation state and the coordination number is 4.
The two ammonia ligands and two chloride ligands can be arranged in two different ways, giving rise to two geometrical isomers.
(a) cis-isomer:
In the cis-isomer, the two chloride ligands are adjacent to each other, and the two ammonia ligands are also adjacent.
This is called cis-\([Pt(NH_3)_2Cl_2]\).
(b) trans-isomer:
In the trans-isomer, the two chloride ligands are opposite to each other, and the two ammonia ligands are also opposite.
This is called trans-\([Pt(NH_3)_2Cl_2]\).
Thus, the complex \( [Pt(NH_3)_2Cl_2] \) shows geometrical isomerism in the form of cis and trans isomers.
Quick Tip: Square planar complexes with formula \( MA_2B_2 \) show cis–trans geometrical isomerism.
(i) Write any two differences between \( \mathrm{S_N1} \) and \( \mathrm{S_N2} \) reaction mechanisms in alkyl halides.
View Solution
Difference 1: Reaction Mechanism.
The \( \mathrm{S_N1} \) reaction proceeds in two steps. In the first step, the alkyl halide forms a carbocation intermediate by losing the halide ion. In the second step, the nucleophile attacks the carbocation.
In contrast, the \( \mathrm{S_N2} \) reaction proceeds in a single step where the nucleophile attacks the carbon atom at the same time as the leaving group departs. No intermediate is formed.
Difference 2: Rate of Reaction.
The rate of an \( \mathrm{S_N1} \) reaction depends only on the concentration of the alkyl halide and follows first-order kinetics.
The rate of an \( \mathrm{S_N2} \) reaction depends on the concentration of both the alkyl halide and the nucleophile and follows second-order kinetics.
Quick Tip: \( \mathrm{S_N1} \) reactions involve carbocation formation, while \( \mathrm{S_N2} \) reactions occur in a single concerted step.
(ii) Write the equation for the dehydrohalogenation reaction of 2-bromobutane using alcoholic KOH and predict the major product.
View Solution
Step 1: Nature of the Reaction.
Dehydrohalogenation is an elimination reaction in which a hydrogen atom and a halogen atom are removed from adjacent carbon atoms of an alkyl halide. Alcoholic KOH acts as a strong base and favors elimination reactions.
Step 2: Application to 2-Bromobutane.
When 2-bromobutane is treated with alcoholic KOH, elimination of hydrogen bromide takes place. According to Saytzeff’s rule, the more substituted alkene is formed as the major product.
Step 3: Reaction Equation.
\[ CH_3CHBrCH_2CH_3 + KOH (alc) \rightarrow CH_3CH=CHCH_3 + KBr + H_2O \]
Step 4: Major Product.
The major product formed is but-2-ene, as it is the more stable, more substituted alkene.
Quick Tip: Alcoholic KOH favors elimination reactions, and the major product follows Saytzeff’s rule.
Aldol condensation
View Solution
Step 1: Definition of Aldol Condensation.
Aldol condensation is a reaction in which aldehydes or ketones containing at least one \(\alpha\)-hydrogen atom react in the presence of a dilute base to form \(\beta\)-hydroxy aldehydes or \(\beta\)-hydroxy ketones, collectively called aldols.
Step 2: Reaction Conditions.
The reaction is carried out in the presence of dilute alkali such as NaOH or KOH. The base removes an \(\alpha\)-hydrogen atom, forming an enolate ion which acts as a nucleophile.
Step 3: Example.
When acetaldehyde undergoes aldol condensation in the presence of dilute NaOH:
\[ 2\,\mathrm{CH_3CHO} \xrightarrow{dil. NaOH} \mathrm{CH_3CH(OH)CH_2CHO} \]
Step 4: Dehydration of Aldol.
On heating, the aldol loses a molecule of water to form an \(\alpha,\beta\)-unsaturated aldehyde:
\[ \mathrm{CH_3CH(OH)CH_2CHO} \xrightarrow{\Delta} \mathrm{CH_3CH=CHCHO + H_2O} \]
Step 5: Conclusion.
Thus, aldol condensation involves formation of an aldol followed by dehydration to give an unsaturated carbonyl compound.
Quick Tip: Aldol condensation occurs only in aldehydes or ketones having \(\alpha\)-hydrogen atoms.
Cannizzaro reaction
View Solution
Step 1: Definition of Cannizzaro Reaction.
Cannizzaro reaction is a disproportionation reaction in which aldehydes that do not contain \(\alpha\)-hydrogen atoms react with concentrated alkali to produce an alcohol and a salt of carboxylic acid.
Step 2: Reaction Conditions.
The reaction takes place in the presence of concentrated NaOH or KOH. One molecule of aldehyde undergoes oxidation, while another undergoes reduction.
Step 3: Example.
When benzaldehyde reacts with concentrated NaOH, the reaction is:
\[ 2\,\mathrm{C_6H_5CHO} + \mathrm{NaOH} \rightarrow \mathrm{C_6H_5CH_2OH} + \mathrm{C_6H_5COONa} \]
Step 4: Explanation.
In this reaction, one molecule of benzaldehyde is reduced to benzyl alcohol, while the other molecule is oxidised to sodium benzoate.
Step 5: Conclusion.
Thus, Cannizzaro reaction is shown by aldehydes lacking \(\alpha\)-hydrogen atoms and involves simultaneous oxidation and reduction.
Quick Tip: Aldehydes without \(\alpha\)-hydrogen atoms undergo Cannizzaro reaction.






















Comments