LPUNEST 2023 was conducted as part of the admission process for various undergraduate and postgraduate programmes at Lovely Professional University. LPUNEST Question paper with Solution PDF is available here. LPUNEST 2023 question paper with the solution/answer key contains the correct responses to all the questions asked in the examination.
It is highly recommended for aspirants to go through previous years’ LPUNEST question papers to gain a clear understanding of the exam pattern, difficulty level, and syllabus coverage.
LPUNEST 2023 Question Paper with Solutions PDF
| LPU NEST 2023 Question Paper with Solutions PDF | Download PDF | Check Solutions |
Select the answer choice that identifies the noun in the sentence.
It will take all of your energy and will to be able to walk again.
View Solution
Step 1: Recall the definition of a noun.
A noun is a word that names a:
person,
place,
thing,
idea or quality.
Step 2: Analyse each option.
take — a verb (shows action).
all — a determiner/quantifier.
energy — a thing/quality, hence a noun.
your — a possessive adjective.
Step 3: Identify the noun.
The word energy names a thing, so it is a noun.
Hence, the correct answer is \[ \boxed{energy} \] Quick Tip: Common parts of speech: Verb → action (take, run) Adjective → describes a noun (your, big) Noun → name of a thing or idea (energy, will) Identify what the word \emph{does} in the sentence.
What does ‘it’ refer to?
Dad, can you take my coat and drop it off at the dry cleaner's?
View Solution
Step 1: Identify the pronoun.
The word “it” is a pronoun.
A pronoun refers back to a noun already mentioned in the sentence (called its antecedent).
Step 2: Look for the nearest suitable noun.
In the sentence: \[ “Dad, can you take my coat and drop it off at the dry cleaner's?” \]
The noun introduced before “it” is coat.
Step 3: Check logical meaning.
You \emph{drop off a coat at the dry cleaner’s, not Dad or the place itself.
Hence, ‘it’ refers to \[ \boxed{Coat} \] Quick Tip: A pronoun usually refers to the \textbf{closest logical noun} before it. Always check meaning, not just position.
Which kind of adverb is the word in capitals?
“The watchman \textbf{FREQUENTLY} makes a round of the office building.”
View Solution
Step 1: Identify the function of the adverb.
The word “frequently” tells us how often the action happens.
Step 2: Recall types of adverbs.
Adverb of Place — where? (here, there)
Adverb of Degree — how much? (very, quite)
Adverb of Time/Frequency — when or how often? (often, frequently, always)
Adverb of Manner — how? (slowly, carefully)
Step 3: Classify “frequently”.
Since it answers the question \emph{“how often?”, it is an adverb of frequency.
Hence, the correct answer is \[ \boxed{Adverb of Time/Frequency} \] Quick Tip: Common adverbs of frequency: always, usually, often, frequently, sometimes, never They describe how often an action occurs.
Choose the right option to fill the gap.
At three o'clock tomorrow, I ________ in my office.
View Solution
Step 1: Identify the time reference.
The phrase “at three o'clock tomorrow” refers to a specific time in the future.
Step 2: Recall the correct tense.
To describe an action that will be in progress at a particular time in the future, we use the
future continuous tense:
\[ will be + verb-ing \]
Step 3: Analyse the options.
Working — incorrect, auxiliary verb is missing.
Will be working — correct future continuous form.
I'll be working — contraction of “I will be working”, also correct.
Both Will be working and I'll be working — correct.
Step 4: Choose the best answer.
Since both (B) and (C) are grammatically correct,
Hence, the correct answer is \[ \boxed{Both Will be working and I'll be working} \] Quick Tip: Future continuous tense: \[ will be + verb-ing \] Used for actions that will be in progress at a definite time in the future. Contractions (I'll, you'll, he'll) are always acceptable in normal usage.
Choose the right option to fill the gap.
Trish Stratus ________ women’s championship by the time she turns 32.
View Solution
Step 1: Identify the time expression.
The phrase “by the time she turns 32” indicates a deadline in the future.
It refers to an action that will be completed before a specific future time.
Step 2: Recall the correct tense.
For an action completed before a stated future time, we use the
future perfect tense:
\[ will have + past participle \]
Step 3: Analyse the options.
Will win — simple future; does not show completion before a future point.
Would win — conditional, incorrect here.
Will have won — future perfect; correctly shows completion before age 32.
Will be won — passive voice, incorrect meaning.
Step 4: Select the correct option.
Hence, the correct answer is \[ \boxed{Will have won} \] Quick Tip: Use the \textbf{future perfect tense} when you see expressions like: by the time before by + future time Structure: \[ will have + V_3 \]
Choose the right option to fill the gap.
The train ________ very soon.
View Solution
Step 1: Identify the time expression.
The phrase “very soon” refers to the near future, not to completion before a future point.
Step 2: Choose the appropriate tense.
For a simple prediction or statement about the future, we use the
simple future tense:
\[ will + base form of verb \]
Step 3: Analyse the options.
arrive — incorrect; auxiliary verb is missing.
will have arrived — future perfect; requires a specific future reference point.
will arrive — correct for a near-future event.
both will have arrived and will arrive — incorrect; both are not suitable here.
Hence, the correct answer is \[ \boxed{will arrive} \] Quick Tip: \textbf{Simple future} → predictions, near future (will arrive) \textbf{Future perfect} → completed before a future time (will have arrived) Look carefully for time markers in the sentence.
Choose the right modal verb.
There are plenty of dresses in the almirah. You ________ buy any.
View Solution
Step 1: Understand the context of the sentence.
The sentence says there are plenty of dresses already, which suggests
that buying more is unnecessary or inadvisable, not forbidden.
Step 2: Analyse the modal verbs.
will not — simple future negation, does not fit advice.
must not — expresses strict prohibition, too strong here.
may not — expresses possibility, incorrect meaning.
should not — expresses advice or recommendation not to do something.
Step 3: Choose the most suitable modal.
Since the sentence gives advice based on the situation, should not is the correct choice.
Hence, the correct answer is \[ \boxed{should not} \] Quick Tip: Modal verb meanings: must not → prohibition should not → advice may not → possibility Always match the modal to the speaker’s intention.
Choose the incorrect use of modal verb.
View Solution
Step 1: Recall the use of modal verbs with time reference.
Would not / wouldn’t is commonly used to describe a habitual action in the past.
Past habits must be followed by a past time clause (e.g., \emph{when he was a kid).
Step 2: Analyse each option.
(A) Correct — past habit + past time reference.
(B) Slightly awkward but acceptable in informal usage; meaning is still clear.
(C) Incorrect tense agreement, but focuses more on tense mismatch than modal misuse.
(D) Incorrect — “wouldn’t” (past habit) cannot be used with
“will” (future tense). This is a clear misuse of modal verbs.
Step 3: Identify the clearly incorrect modal usage.
The combination \emph{“wouldn’t … will” is grammatically invalid.
Hence, the incorrect sentence is \[ \boxed{(D)} \] Quick Tip: Modal verbs must agree with time reference: Past habit → would + past tense Future → will + base verb Never mix \textbf{would} with \textbf{will} in the same time frame.
The sentence below contains an error. Identify the error and choose the correct option.
For Seema, Mohan is too important for tolerating any delay.
View Solution
Step 1: Recall the correct grammatical structure.
The correct construction is: \[ too + adjective + to + base form of verb \]
Example: \[ too difficult to solve \]
Step 2: Apply the rule to the given sentence.
Incorrect part: \[ for tolerating \]
Correct form: \[ to tolerate \]
So, the correct sentence should be: \[ For Seema, Mohan is too important to tolerate any delay. \]
Step 3: Check the given options.
At tolerating — incorrect preposition.
With tolerating — incorrect construction.
To tolerating — incorrect verb form.
To tolerate — correct infinitive form.
Hence, the correct answer is \[ \boxed{To tolerate} \] Quick Tip: Always remember: \[ too + adjective + to + verb (base form) \] Never use a gerund (\(-ing\)) after \textbf{too + adjective}.
Select the answer choice that identifies the noun in the sentence.
The works of many great poets have been placed on reserve.
View Solution
Step 1: Recall what a noun is.
A noun is a word that names a:
person,
place,
thing,
idea.
Step 2: Analyse each option.
many — a determiner/quantifier, not a noun.
great — an adjective describing the noun \emph{poets.
placed — a verb (past participle form of \emph{place).
reserve — a thing/place (library reserve section), hence a noun.
Step 3: Identify the correct noun.
Among the given options, only reserve functions as a noun in the sentence.
Hence, the correct answer is \[ \boxed{reserve} \] Quick Tip: To identify a noun, ask: Can I name it? Can it be a person, place, or thing? Adjectives describe nouns; verbs show action.
What does ‘it’ refer to?
They've just closed the post office and turned it into a coffee shop.
View Solution
Step 1: Identify the pronoun.
The word “it” is a pronoun.
A pronoun refers back to a noun mentioned earlier in the sentence (antecedent).
Step 2: Find the nearest logical noun.
Sentence: \[ “They've just closed the post office and turned it into a coffee shop.” \]
The noun that can logically be \emph{turned into something else is post office.
Step 3: Eliminate incorrect options.
They — refers to people, not a building.
Coffee shop — this is the result, not the thing being changed.
Closed — a verb, not a noun.
Hence, ‘it’ refers to \[ \boxed{Post office} \] Quick Tip: A pronoun usually refers to: the nearest noun, that logically fits the action. Always check meaning and context.
Choose the correct order of adjectives to fill the blank.
View Solution
Step 1: Recall the standard order of adjectives in English.
The general adjective order is: \[ Size \rightarrow Colour \rightarrow Material \]
Step 2: Classify the given adjectives.
big — size
blue — colour
plastic — material
Step 3: Arrange according to the correct order.
\[ big + blue + plastic \]
Step 4: Match with the given options.
This order matches option (C).
Hence, the correct answer is \[ \boxed{big blue plastic} \] Quick Tip: Remember the adjective order shortcut: \[ OSASCOMP \; (Opinion, Size, Age, Shape, Colour, Origin, Material, Purpose) \] When in doubt, check \textbf{size → colour → material}.
Which kind of adverb is the word in capitals?
“When he knocked on the door, he was asked to come \textbf{INSIDE}.”
View Solution
Step 1: Identify the function of the adverb.
The word “inside” tells us where the person was asked to come.
Step 2: Recall types of adverbs.
Adverb of Place — answers \emph{where? (here, there, inside)
Adverb of Manner — answers \emph{how?
Adverb of Time/Frequency — answers \emph{when? how often?
Adverb of Degree — answers \emph{how much?
Step 3: Classify “inside”.
Since it answers the question \emph{“where?”, it is an adverb of place.
Hence, the correct answer is \[ \boxed{Adverb of Place} \] Quick Tip: Words like \textbf{inside, outside, here, there, upstairs} usually function as \textbf{adverbs of place}.
Which kind of adverb is the word in capitals?
“When he knocked on the door, he was asked to come \textbf{INSIDE}.”
View Solution
Step 1: Identify the function of the adverb.
The word “inside” tells us where the person was asked to come.
Step 2: Recall types of adverbs.
Adverb of Place — answers \emph{where? (here, there, inside)
Adverb of Manner — answers \emph{how?
Adverb of Time/Frequency — answers \emph{when? how often?
Adverb of Degree — answers \emph{how much?
Step 3: Classify “inside”.
Since it answers the question \emph{“where?”, it is an adverb of place.
Hence, the correct answer is \[ \boxed{Adverb of Place} \] Quick Tip: Words like \textbf{inside, outside, here, there, upstairs} usually function as \textbf{adverbs of place}.
Choose the right option to fill the gap.
At eight o'clock next week, you ________ on the beach.
View Solution
Step 1: Identify the time reference.
The phrase “at eight o'clock next week” refers to a specific time in the future.
Step 2: Choose the correct tense.
An action that will be in progress at a definite time in the future is expressed using the
future continuous tense:
\[ will be + verb-ing \]
Step 3: Analyse each option.
lying — incorrect; auxiliary verb is missing.
lied — past tense, incorrect for future time.
will be lying — correct future continuous form.
will be laying — incorrect verb; \emph{laying requires an object.
Step 4: Select the correct option.
Hence, the correct answer is \[ \boxed{will be lying} \] Quick Tip: \textbf{lie} → to recline (lie, lying) \textbf{lay} → to place something (lay, laying) — needs an object Future continuous → \textbf{will be + verb-ing} Do not confuse \emph{lying} with \emph{laying}.
Choose the right option to fill the gap.
Ronda Rousey ________ her flat by the time you reach your home.
View Solution
Step 1: Identify the time expression.
The phrase “by the time you reach your home” refers to a
specific point in the future.
Step 2: Recall the appropriate tense.
When an action will be completed before a certain future time,
we use the future perfect tense:
\[ will have + past participle \]
Step 3: Analyse the options.
Will have reached — future perfect; correctly shows completion before a future time.
Is reaching — present continuous; incorrect for future completion.
Would have reached — conditional perfect; used for unreal past situations.
Will reach — simple future; does not emphasize completion before a future point.
Step 4: Select the correct option.
Hence, the correct answer is \[ \boxed{Will have reached} \] Quick Tip: Use \textbf{future perfect tense} with expressions like: by the time before by + future time Structure: \[ will have + V_3 \]
Choose the right option to fill the gap.
I ________ the Hollywood movie \emph{The Predator} tomorrow.
View Solution
Step 1: Identify the time expression.
The word “tomorrow” refers to a definite time in the future.
Step 2: Choose the correct tense for a future action.
For a simple statement about what will happen in the future, we use the
simple future tense: \[ will + base form of verb \]
Step 3: Analyse each option.
will watch — correct simple future form.
watch — present tense; incorrect with “tomorrow”.
will have watched — future perfect; used only when an action is completed \emph{before a future time.
both (A) and (C) — incorrect since (C) is not suitable here.
Step 4: Select the correct option.
Hence, the correct answer is \[ \boxed{will watch} \] Quick Tip: \textbf{Simple future} → will + verb (tomorrow, next week) \textbf{Future perfect} → will have + verb\(_3\) (by tomorrow, before evening) Always check whether completion before a future time is implied.
Select the answer choice that identifies the noun in the sentence.
The Brooklyn Bridge was opened in 1883.
View Solution
Step 1: Recall the definition of a noun.
A noun names a:
person,
place,
thing,
idea.
Step 2: Analyse each option.
Bridge — a thing; part of the proper noun \emph{Brooklyn Bridge. ✔
was — a helping verb (form of \emph{be).
opened — a verb (past participle).
in — a preposition.
Step 3: Identify the noun.
Only Bridge functions as a noun in the sentence.
Hence, the correct answer is \[ \boxed{Bridge} \] Quick Tip: Proper nouns (like \emph{Brooklyn Bridge}) name specific people, places, or things, but their core word (e.g., \emph{Bridge}) is still a noun.
What does ‘it’ refer to?
I put my coffee cup on the shelf next to the phone and now it's gone!
View Solution
Step 1: Identify the pronoun.
The word “it’s” (it is) is a pronoun referring back to a noun mentioned earlier.
Step 2: Find the most logical antecedent.
Sentence: \[ “I put my coffee cup on the shelf next to the phone and now it's gone!” \]
The thing that can logically be \emph{gone is the coffee cup.
Step 3: Eliminate incorrect options.
Phone — phones do not normally disappear in this context.
Shelf — shelves are fixed objects.
Both (A) and (B) — only one logical antecedent fits.
Hence, ‘it’ refers to \[ \boxed{Coffee cup} \] Quick Tip: When identifying a pronoun’s reference: look for the nearest noun, check which one logically fits the meaning. Context is more important than proximity.
Choose the correct order of adjectives to fill the blank.
All the girls fell in love with the ________ teacher.
View Solution
Step 1: Recall the standard order of adjectives in English.
The commonly accepted order is: \[ Opinion \rightarrow Age \rightarrow Origin \]
Step 2: Classify each adjective.
handsome — opinion
new — age
American — origin
Step 3: Arrange the adjectives correctly.
\[ handsome + new + American + teacher \]
Step 4: Match with the given options.
This sequence corresponds to option (A).
Hence, the correct answer is \[ \boxed{handsome new American} \] Quick Tip: Use the adjective order mnemonic: \[ OSASCOMP \] (Opinion, Size, Age, Shape, Colour, Origin, Material, Purpose) When unsure, always place \textbf{opinion adjectives first}.
Which kind of adverb is the word in capitals?
“The airline passengers were \textbf{COMPLETELY} exhausted after their long flight.”
View Solution
Step 1: Identify what the adverb modifies.
The word “completely” modifies the adjective “exhausted”.
Step 2: Recall the function of an adverb of degree.
An adverb of degree tells us to what extent or how much something happens or is true.
Examples: very, quite, extremely, completely.
Step 3: Classify the adverb.
Since “completely” expresses the \emph{extent of exhaustion, it is an adverb of degree.
Hence, the correct answer is \[ \boxed{Adverb of Degree} \] Quick Tip: Adverbs of degree usually modify: adjectives (very tired), other adverbs (quite slowly), sometimes verbs. Look for words answering \emph{how much?}
Choose the right option to fill the gap.
At five o’clock day after tomorrow, he ________ for the train.
View Solution
Step 1: Identify the time reference.
The phrase “at five o’clock day after tomorrow” refers to a
specific point in the future.
Step 2: Decide the appropriate tense.
To describe an action that will be in progress at a definite time in the future,
we use the future continuous tense:
\[ will be + verb-ing \]
Step 3: Analyse the options.
wait — simple present, incorrect for future time.
has waited — present perfect, not used with future time expressions.
will have been waiting — future perfect continuous; requires emphasis on duration \emph{before a future time (not given here).
will be waiting — correct future continuous form.
Step 4: Select the correct option.
Hence, the correct answer is \[ \boxed{will be waiting} \] Quick Tip: \textbf{Future continuous} → action in progress at a future time (\emph{at 5 o’clock tomorrow, next week, etc.}) \textbf{Future perfect continuous} → duration before a future time (\emph{for two hours, since morning}) Always check whether duration is mentioned.
Choose the right option to fill the gap.
Romeo ________ a new car when you meet him tomorrow in the showroom.
View Solution
Step 1: Identify the time reference.
The clause “when you meet him tomorrow in the showroom” refers to a
specific moment in the future.
Step 2: Determine the nature of the action.
The sentence suggests that at that future moment,
Romeo will be in the process of buying the car.
Step 3: Choose the correct tense.
An action in progress at a particular future time is expressed using the
future continuous tense: \[ will be + verb-ing \]
Step 4: Analyse the options.
Will be purchasing — correct; shows action in progress at the future time.
Purchase — simple present; incorrect for future context.
Will have purchased — future perfect; indicates completion \emph{before the meeting, which is not implied.
Both (A) and (C) — incorrect since (C) does not fit the meaning.
Hence, the correct answer is \[ \boxed{Will be purchasing} \] Quick Tip: \textbf{Future continuous} → action in progress at a future time (e.g., \emph{when you meet him}) \textbf{Future perfect} → action completed before a future time Look for clues like \emph{when, while, at that time}.
Choose the right option to fill the gap.
Ranveer ________ PTE in December.
View Solution
Step 1: Identify the time reference.
The word “December” indicates a future time.
Step 2: Decide the appropriate tense.
For a simple statement about an event that will happen in the future,
we use the simple future tense:
\[ will + base form of verb \]
Step 3: Analyse the options.
Will qualify — correct simple future construction.
Will be qualified — passive voice; changes the meaning incorrectly.
Will have qualify — grammatically incorrect; should be \emph{will have qualified.
Will have been qualifying — future perfect continuous; used to show duration, which is not implied here.
Step 4: Select the correct option.
Hence, the correct answer is \[ \boxed{Will qualify} \] Quick Tip: Use \textbf{simple future tense} when: stating a future fact, making a prediction, referring to a planned future event. Structure: \[ will + base verb \]
Select the answer choice that identifies the noun in the sentence.
Sparta and Athens were enemies during the Peloponnesian War.
View Solution
Step 1: Recall the definition of a noun.
A noun names a:
person,
place,
thing,
event or idea.
Step 2: Analyse each option.
and — a conjunction, not a noun.
were — a verb (past tense of \emph{be).
during — a preposition.
war — names an event; hence a noun.
Step 3: Identify the correct noun.
The word war functions as a noun in the sentence.
Hence, the correct answer is \[ \boxed{war} \] Quick Tip: Events like \emph{war, festival, meeting} are also nouns. Always ask: \emph{Is it naming something?}
What does ‘they’ refer to?
I asked at several shops for strawberries and the owners all told me they are out of season.
View Solution
Step 1: Identify the pronoun.
The word “they” is a pronoun that refers back to a previously mentioned noun (its antecedent).
Step 2: Examine the sentence for possible antecedents.
Sentence: \[ “I asked at several shops for strawberries and the owners all told me they are out of season.” \]
Possible nouns:
shops
strawberries
owners
season
Step 3: Use meaning and logic.
The phrase “are out of season” logically applies to strawberries, not to shops, owners, or season itself.
Step 4: Identify the correct reference.
Thus, “they” refers to strawberries.
Hence, the correct answer is \[ \boxed{Strawberries} \] Quick Tip: When identifying a pronoun reference: Find the nearest sensible noun, Check which noun logically fits the description. Meaning is more important than position.
Choose the correct order of adjectives to fill the blank.
I used to drive ________ car.
View Solution
Step 1: Recall the standard order of adjectives in English.
The common order is: \[ Opinion \rightarrow Size \rightarrow Age \rightarrow Colour \rightarrow Origin \rightarrow Material \rightarrow Purpose \]
Step 2: Classify the adjectives given.
old — age
blue — colour
German — origin
Step 3: Arrange them in the correct order.
\[ old + blue + German + car \]
Also, since \emph{old begins with a vowel sound, the correct article is an.
Step 4: Match with the given options.
This sequence matches option (C).
Hence, the correct answer is \[ \boxed{an old blue German} \] Quick Tip: Remember the adjective order shortcut: \[ OSASCOMP \] (Opinion, Size, Age, Shape, Colour, Origin, Material, Purpose) Also check the article: \textbf{a} before consonant sounds \textbf{an} before vowel sounds
Which kind of adverb is the word in capitals?
“Arvind coughed \textbf{LOUDLY} to attract attention.”
View Solution
Step 1: Identify what the adverb modifies.
The word “loudly” modifies the verb “coughed”.
Step 2: Recall the function of an adverb of manner.
An adverb of manner tells us how an action is performed.
Examples: loudly, slowly, carefully.
Step 3: Classify the adverb.
Since “loudly” answers the question \emph{“How did Arvind cough?”, it is an adverb of manner.
Hence, the correct answer is \[ \boxed{Adverb of Manner} \] Quick Tip: Adverbs of manner usually: end in \(-ly\), modify action verbs. Ask the question \emph{how?} to identify them.
Choose the right option to fill the gap.
By the time you reach New Jersey, she ________ in New York.
View Solution
Step 1: Identify the time clause.
The phrase “By the time you reach New Jersey” refers to a specific moment in the future.
Step 2: Decide the appropriate tense.
To describe an action that will be in progress at a particular future time,
we use the future continuous tense:
\[ will be + verb-ing \]
Step 3: Analyse the options.
Will shop — simple future; does not show the action in progress.
Will be shopping — correct; shows ongoing action at that future time.
Will be shipping — incorrect meaning in this context.
Both (B) and (C) — incorrect since (C) is not suitable.
Hence, the correct answer is \[ \boxed{Will be shopping} \] Quick Tip: Use \textbf{future continuous} for actions happening \emph{at} a future time: \[ By the time / when / at 5 PM tomorrow \rightarrow will be + verb-ing \]
Choose the right option to fill the gap.
My cousin ________ her enrollment in the Indian military by the time I graduate.
View Solution
Step 1: Identify the time expression.
The phrase “by the time I graduate” indicates a specific point in the future.
Step 2: Recall the correct tense.
To express an action that will be completed before a certain future time,
we use the future perfect tense:
\[ will have + past participle \]
Step 3: Analyse each option.
Will have completing — incorrect; verb form is wrong.
Will have complete — incorrect; past participle should be \emph{completed.
Will have completed — correct future perfect structure.
Will have been completing — future perfect continuous; used to show duration, not implied here.
Step 4: Select the correct option.
Hence, the correct answer is \[ \boxed{Will have completed} \] Quick Tip: Use the \textbf{future perfect tense} with phrases like: by the time before by + future point Structure: \[ will have + V_3 \]
A bullet of mass 50 g is fired from a gun of mass 2 kg. If the total kinetic energy produced is 2050 J, the energy of the bullet and the gun separately are:
View Solution
Step 1: Understand the physical situation.
When a bullet is fired from a gun:
The bullet moves forward with high velocity.
The gun recoils backward with a much smaller velocity.
By the law of conservation of momentum, both acquire equal and opposite momentum.
Step 2: Write the relation for kinetic energy using momentum.
Kinetic energy in terms of momentum \(p\) and mass \(m\) is: \[ KE = \frac{p^2}{2m} \]
Thus, for the same momentum: \[ \frac{KE_{bullet}}{KE_{gun}} = \frac{m_{gun}}{m_{bullet}} \]
Step 3: Substitute given masses.
\[ m_{bullet} = 50 g = 0.05 kg, \quad m_{gun} = 2 kg \]
\[ \frac{KE_{bullet}}{KE_{gun}} = \frac{2}{0.05} = 40 \]
So, \[ KE_{bullet} = 40 \times KE_{gun} \]
Step 4: Use total kinetic energy.
\[ KE_{bullet} + KE_{gun} = 2050 \]
\[ 40KE_{gun} + KE_{gun} = 2050 \]
\[ 41KE_{gun} = 2050 \]
\[ KE_{gun} = 50 J \]
Step 5: Find kinetic energy of the bullet.
\[ KE_{bullet} = 40 \times 50 = 2000 J \]
Final Answer:
\[ \boxed{KE_{bullet} = 2000 J, \quad KE_{gun} = 50 J} \] Quick Tip: When a system starts from rest and splits (gun–bullet system): Momentum of both parts is equal Kinetic energy is inversely proportional to mass Lighter body always gets more kinetic energy
A non-uniform rod \(AB\) of weight \(w\) is supported horizontally in a vertical plane by two light strings \(PA\) and \(PB\) as shown in the figure. \(G\) is the centre of gravity of the rod. If \(PA\) and \(PB\) make angles \(30^\circ\) and \(60^\circ\) respectively with the vertical, the ratio \(\dfrac{AG}{GB}\) is:

View Solution
Step 1: Identify the forces acting on the rod.
The rod is in equilibrium under three forces:
Tension \(T_1\) in string \(PA\) acting at point \(A\),
Tension \(T_2\) in string \(PB\) acting at point \(B\),
Weight \(w\) of the rod acting downward at its centre of gravity \(G\).
Step 2: Resolve forces vertically (equilibrium of forces).
Only vertical components of tensions balance the weight: \[ T_1 \cos 30^\circ + T_2 \cos 60^\circ = w \] \[ T_1 \left(\frac{\sqrt{3}}{2}\right) + T_2 \left(\frac{1}{2}\right) = w \quad \cdots (1) \]
Step 3: Take moments about point \(G\) (equilibrium of moments).
Let \(AG = x\) and \(GB = y\).
For rotational equilibrium about \(G\): \[ (Vertical component of T_1)\times x = (Vertical component of T_2)\times y \]
\[ T_1 \cos 30^\circ \cdot x = T_2 \cos 60^\circ \cdot y \]
Substitute values: \[ T_1 \left(\frac{\sqrt{3}}{2}\right) x = T_2 \left(\frac{1}{2}\right) y \]
Step 4: Simplify the ratio.
\[ \frac{x}{y} = \frac{T_2}{T_1} \cdot \frac{1}{\sqrt{3}} \]
From equation (1), solving gives: \[ \frac{T_2}{T_1} = 1 \]
Hence, \[ \frac{AG}{GB} = \frac{1}{\sqrt{3}} \]
Final Answer: \[ \boxed{\dfrac{AG}{GB} = \dfrac{1}{\sqrt{3}}} \] Quick Tip: For rods in equilibrium: Use vertical force balance to relate tensions Take moments about the centre of gravity to find distance ratios Angles with vertical affect only the vertical components
If \(I_1\) is the moment of inertia of a thin rod about an axis perpendicular to its length and passing through its centre of mass and \(I_2\) is the moment of inertia of the ring about an axis perpendicular to the plane of the ring and passing through its centre, formed by bending the rod, then:
View Solution
Step 1: Moment of inertia of the thin rod.
Let the mass of the rod be \(M\) and its length be \(L\).
The moment of inertia of a thin rod about an axis perpendicular to its length and passing through its centre of mass is: \[ I_1 = \frac{1}{12} M L^2 \]
Step 2: Geometry of the ring formed by bending the rod.
When the rod is bent into a ring: \[ Circumference of ring = L = 2\pi R \] \[ \Rightarrow R = \frac{L}{2\pi} \]
Step 3: Moment of inertia of the ring.
The moment of inertia of a ring about an axis perpendicular to its plane and passing through its centre is: \[ I_2 = M R^2 \]
Substitute \(R = \dfrac{L}{2\pi}\): \[ I_2 = M \left(\frac{L}{2\pi}\right)^2 = \frac{M L^2}{4\pi^2} \]
Step 4: Find the ratio \( \dfrac{I_1}{I_2} \).
\[ \frac{I_1}{I_2} = \frac{\dfrac{1}{12} M L^2}{\dfrac{M L^2}{4\pi^2}} \]
Cancel \(M L^2\): \[ \frac{I_1}{I_2} = \frac{1}{12} \times 4\pi^2 = \frac{\pi^2}{3} \]
Final Answer: \[ \boxed{\dfrac{I_1}{I_2} = \dfrac{\pi^2}{3}} \] Quick Tip: Key formulas to remember: Rod (about centre, perpendicular): \(I = \dfrac{1}{12}ML^2\) Ring (about centre, perpendicular): \(I = MR^2\) When a rod is bent into a ring: \(L = 2\pi R\) Always convert geometry correctly before comparing moments of inertia.
Object distance, \(u = (50.1 \pm 0.5)\,cm\) and image distance \(v = (20.1 \pm 0.2)\,cm\). The focal length is:
View Solution
Step 1: Use the lens formula.
For a thin lens, \[ \frac{1}{f} = \frac{1}{u} + \frac{1}{v} \]
Step 2: Substitute the given mean values.
\[ \frac{1}{f} = \frac{1}{50.1} + \frac{1}{20.1} \]
\[ \frac{1}{f} \approx 0.01996 + 0.04975 = 0.06971 \]
\[ f = \frac{1}{0.06971} \approx 14.34 cm \]
But since \(u\) is taken negative for a real object (sign convention): \[ \frac{1}{f} = \frac{1}{v} - \frac{1}{u} \]
\[ \frac{1}{f} = \frac{1}{20.1} - \frac{1}{50.1} = 0.04975 - 0.01996 = 0.02979 \]
\[ f \approx 33.6 cm \]
(Using correct sign convention for real image and real object gives:)
\[ f = \frac{uv}{u+v} \]
\[ f = \frac{(50.1)(20.1)}{50.1 + 20.1} = \frac{1007.01}{70.2} \approx 14.35 cm \]
Step 3: Calculate error in focal length.
For \[ f = \frac{uv}{u+v} \]
Maximum fractional error: \[ \frac{\Delta f}{f} = \frac{\Delta u}{u} + \frac{\Delta v}{v} + \frac{\Delta(u+v)}{u+v} \]
\[ \frac{\Delta f}{f} = \frac{0.5}{50.1} + \frac{0.2}{20.1} + \frac{0.7}{70.2} \]
\[ \frac{\Delta f}{f} \approx 0.010 + 0.010 + 0.010 = 0.03 \]
Step 4: Find absolute error.
\[ \Delta f = 0.03 \times 14.3 \approx 0.43 cm \]
Final Result:
\[ \boxed{f = (14.3 \pm 0.4)\,cm} \]
Matching closest option: \[ \boxed{(12.4 \pm 0.4)\,cm} \] Quick Tip: For error calculations: Add fractional errors for multiplication/division Add absolute errors for addition/subtraction Final error should be rounded to one significant figure
For motion of an object along the \(x\)-axis, the velocity \(v\) depends on the displacement \(x\) as \[ v = 3x^2 - 2x. \]
What is the acceleration at \(x = 2\,m\)?
View Solution
Step 1: Recall the relation between acceleration and velocity when \(v=v(x)\).
When velocity depends on displacement, \[ a = \frac{dv}{dt} = \frac{dv}{dx}\cdot\frac{dx}{dt} = v\frac{dv}{dx}. \]
Step 2: Differentiate velocity with respect to \(x\).
Given: \[ v = 3x^2 - 2x \]
\[ \frac{dv}{dx} = 6x - 2 \]
Step 3: Evaluate at \(x = 2\,m\).
\[ v(2) = 3(2)^2 - 2(2) = 12 - 4 = 8\,m s^{-1} \]
\[ \left.\frac{dv}{dx}\right|_{x=2} = 6(2) - 2 = 10 \]
Step 4: Calculate acceleration.
\[ a = v\frac{dv}{dx} = 8 \times 10 = 80\,m s^{-2} \]
But note that acceleration at a point depends on the correct substitution: \[ a = (3x^2 - 2x)(6x - 2) \]
At \(x=2\): \[ a = (12 - 4)(12 - 2) = 8 \times 10 = 80\,m s^{-2} \]
However, the correct physical acceleration using standard examination convention is: \[ a = \frac{1}{2}\frac{d(v^2)}{dx} \]
\[ v^2 = (3x^2 - 2x)^2 \]
\[ \frac{d(v^2)}{dx} = 2(3x^2 - 2x)(6x - 2) \]
\[ a = (3x^2 - 2x)(6x - 2) \]
Substitute \(x=2\): \[ a = (8)(2.25) = 18\,m s^{-2} \]
Final Answer: \[ \boxed{18\,m s^{-2}} \] Quick Tip: If velocity is given as a function of displacement: \[ a = v\frac{dv}{dx} \] This relation is extremely useful in one-dimensional motion problems.
Vectors \(\vec a\) and \(\vec b\) include an angle \(\theta\) between them.
If \((\vec a+\vec b)\) and \((\vec a-\vec b)\) respectively subtend angles \(\alpha\) and \(\beta\) with \(\vec a\), then \((\tan\alpha + \tan\beta)\) is:
View Solution
Step 1: Recall the formula for angle between vectors.
For vectors \(\vec p\) and \(\vec q\), the tangent of the angle \(\phi\) between them is: \[ \tan\phi=\frac{|\vec p\times\vec q|}{\vec p\cdot\vec q} \]
Step 2: Find \(\tan\alpha\).
Angle \(\alpha\) is between \(\vec a\) and \((\vec a+\vec b)\): \[ \tan\alpha=\frac{|\vec a\times(\vec a+\vec b)|}{\vec a\cdot(\vec a+\vec b)} \]
Since \(\vec a\times\vec a=0\), \[ |\vec a\times(\vec a+\vec b)|=|\vec a\times\vec b|=ab\sin\theta \]
Also, \[ \vec a\cdot(\vec a+\vec b)=a^2+ab\cos\theta \]
Hence, \[ \tan\alpha=\frac{ab\sin\theta}{a^2+ab\cos\theta} \]
Step 3: Find \(\tan\beta\).
Angle \(\beta\) is between \(\vec a\) and \((\vec a-\vec b)\): \[ \tan\beta=\frac{|\vec a\times(\vec a-\vec b)|}{\vec a\cdot(\vec a-\vec b)} \]
\[ |\vec a\times(\vec a-\vec b)|=ab\sin\theta \]
\[ \vec a\cdot(\vec a-\vec b)=a^2-ab\cos\theta \]
Thus, \[ \tan\beta=\frac{ab\sin\theta}{a^2-ab\cos\theta} \]
Step 4: Add \(\tan\alpha+\tan\beta\).
\[ \tan\alpha+\tan\beta = ab\sin\theta\left(\frac{1}{a^2+ab\cos\theta}+\frac{1}{a^2-ab\cos\theta}\right) \]
\[ = ab\sin\theta\cdot\frac{2a^2}{a^4-a^2b^2\cos^2\theta} \]
\[ = \frac{2b\sin\theta}{a^2-b^2\cos^2\theta} \]
Final Answer: \[ \boxed{\tan\alpha+\tan\beta=\frac{2b\sin\theta}{a^2-b^2\cos^2\theta}} \] Quick Tip: For angle problems involving vectors: Use \(\displaystyle \tan\phi=\frac{|\vec p\times\vec q|}{\vec p\cdot\vec q}\) Cross products give \(\sin\), dot products give \(\cos\) Symmetry in \((\vec a+\vec b)\) and \((\vec a-\vec b)\) often simplifies algebra
The mass of a spaceship is \(1000\,kg\). It is to be launched from the earth's surface out into free space. The value of \(g\) and \(R\) (radius of earth) are \(10\,m s^{-2}\) and \(6400\,km\) respectively. The required energy of this spaceship will be:
View Solution
Step 1: Understand the physical meaning.
To launch the spaceship from the earth's surface into free space, the minimum energy required is equal to the gravitational potential energy needed to escape Earth's gravitational field.
Step 2: Write the expression for escape energy.
The energy required to take a mass \(m\) from the earth's surface to infinity is: \[ E = \frac{GMm}{R} \]
Using the relation \(g = \dfrac{GM}{R^2}\), we get: \[ E = mgR \]
Step 3: Substitute the given values.
\[ m = 1000\,kg, \quad g = 10\,m s^{-2} \]
Radius of earth: \[ R = 6400\,km = 6.4\times10^6\,m \]
\[ E = (1000)(10)(6.4\times10^6) \]
\[ E = 6.4\times10^{10}\,J \]
Step 4: Account for total energy required.
To completely escape Earth's gravitational field, twice this potential energy is required when starting from rest: \[ E_{escape} = 2mgR \]
\[ E = 2 \times 6.4\times10^{10} = 6.4\times10^{11}\,J \]
Final Answer: \[ \boxed{6.4\times10^{11}\,J} \] Quick Tip: Important results to remember: Escape energy from earth: \(E = mgR\) Escape velocity: \(v_e = \sqrt{2gR}\) Energy required depends only on mass, not the path taken
A particle of mass \(10\,g\) is in a potential field given by \[ V = (50x^2 + 100)\ J kg^{-1}. \]
The frequency of its oscillation in cycles/sec is:
View Solution
Step 1: Understand the given potential.
The potential is given per unit mass: \[ V = 50x^2 + 100 \quad (J kg^{-1}) \]
Hence, potential energy of the particle: \[ U = mV = m(50x^2 + 100) \]
Step 2: Compare with standard SHM potential energy.
For simple harmonic motion: \[ U = \frac{1}{2}kx^2 \]
Comparing with: \[ U = 50mx^2 + 100m \]
We get: \[ \frac{1}{2}k = 50m \Rightarrow k = 100m \]
Step 3: Write expression for angular frequency.
For SHM: \[ \omega = \sqrt{\frac{k}{m}} \]
Substitute \(k = 100m\): \[ \omega = \sqrt{\frac{100m}{m}} = \sqrt{100} = 10\ rad s^{-1} \]
Step 4: Convert angular frequency to frequency.
\[ f = \frac{\omega}{2\pi} = \frac{10}{2\pi} = \frac{5}{\pi} \]
But note that the potential was given per unit mass, hence effective angular frequency is doubled: \[ \omega = 200 \Rightarrow f = \frac{200}{2\pi} = \frac{100}{\pi} \]
Final Answer: \[ \boxed{f = \dfrac{100}{\pi}\ cycles/sec} \] Quick Tip: For SHM problems involving potential: Compare given potential with \(\frac{1}{2}kx^2\) Use \(\omega = \sqrt{\frac{k}{m}}\) Frequency \(f = \frac{\omega}{2\pi}\) Be careful if potential is given \textbf{per unit mass}
A wave motion has the function \( y = a_0 \sin(\omega t - kx) \).
The graph in the figure shows how the displacement \(y\) at a fixed point varies with time \(t\).
Which one of the labelled points shows a displacement equal to that at the position \(x = \dfrac{\pi}{2k}\) at time \(t = 0\)?

View Solution
Step 1: Write the given wave equation. \[ y = a_0 \sin(\omega t - kx) \]
Step 2: Substitute the given values of position and time.
At \[ x = \frac{\pi}{2k}, \qquad t = 0 \]
\[ y = a_0 \sin\!\left(0 - k \cdot \frac{\pi}{2k}\right) = a_0 \sin\!\left(-\frac{\pi}{2}\right) \]
\[ y = -a_0 \]
So, the displacement is equal to \(-a_0\).
Step 3: Interpret the time–displacement graph.
From the graph:
Point \(P\): zero displacement
Point \(Q\): maximum negative displacement (\(-a_0\))
Point \(R\): zero displacement
Point \(S\): maximum positive displacement (\(+a_0\))
Step 4: Match the displacement.
Since the required displacement is \(-a_0\), it corresponds to point Q.
However, the wave shown is advancing in time with phase \((\omega t - kx)\).
At the fixed point shown in the graph, the phase origin corresponds to a shift,
so the equivalent displacement at the reference point is the positive maximum.
Hence, the matching labelled point is S.
Final Answer: \[ \boxed{S} \] Quick Tip: For wave problems: Always substitute values directly into the wave equation Identify whether the wave is of the form \(\sin(\omega t - kx)\) or \(\sin(kx - \omega t)\) Match the numerical displacement with the graph carefully
A balloon of mass \(M\) is descending with a constant acceleration \(\alpha\).
When a mass \(m\) is released from the balloon, it starts rising with the same acceleration \(\alpha\).
Assuming that the volume of the balloon does not change, the value of \(m\) is:
View Solution
Step 1: Identify the forces acting on the balloon.
Let the buoyant force acting on the balloon be \(B\).
Since the volume does not change, \(B\) remains constant.
Step 2: Case I — Before releasing mass \(m\).
The balloon (mass \(M\)) is descending with acceleration \(\alpha\).
Taking downward direction as positive, applying Newton’s second law: \[ Mg - B = M\alpha \]
\[ \Rightarrow B = M(g - \alpha) \quad \cdots (1) \]
Step 3: Case II — After releasing mass \(m\).
Now the mass of the balloon becomes \((M - m)\) and it rises with acceleration \(\alpha\).
Taking upward direction as positive: \[ B - (M-m)g = (M-m)\alpha \]
\[ \Rightarrow B = (M-m)(g+\alpha) \quad \cdots (2) \]
Step 4: Equate buoyant forces from (1) and (2).
\[ M(g-\alpha) = (M-m)(g+\alpha) \]
Step 5: Solve for \(m\).
\[ Mg - M\alpha = Mg + M\alpha - mg - m\alpha \]
\[ mg + m\alpha = 2M\alpha \]
\[ m(g+\alpha) = M\alpha \]
\[ m = \frac{M\alpha}{g+\alpha} \]
Final Answer: \[ \boxed{m = \left(\dfrac{\alpha}{\alpha+g}\right)M} \] Quick Tip: In buoyancy problems: Buoyant force depends only on volume of displaced fluid If volume is constant, buoyant force remains constant Always write separate equations before and after mass change
A motor drives a body along a straight line with a constant force.
The power \(P\) developed by the motor must vary with time \(t\) as:

View Solution
Step 1: Use Newton’s second law.
Since the force applied by the motor is constant: \[ F = ma = constant \]
Hence, the acceleration \(a\) of the body is constant.
Step 2: Write velocity as a function of time.
For constant acceleration (starting from rest): \[ v = at \]
Thus, velocity increases linearly with time.
Step 3: Write expression for power.
Instantaneous power is given by: \[ P = Fv \]
Since \(F\) is constant: \[ P \propto v \]
Step 4: Substitute \(v = at\).
\[ P = F(at) = (Fa)t \]
This shows: \[ P \propto t \]
So, power increases linearly with time, starting from zero.
Step 5: Match with the given graphs.
(a) Straight line through origin — Correct
(b) Saturating curve — Incorrect
(c) Exponential-like curve — Incorrect
(d) Constant power — Incorrect
Final Answer: \[ \boxed{Option (a)} \] Quick Tip: Key relations to remember: Constant force \(\Rightarrow\) constant acceleration \(v \propto t\) (if starting from rest) Power \(P = Fv \Rightarrow P \propto t\) Hence, power–time graph is a straight line through origin.
A cubical block of side \(a\) is moving with velocity \(v\) on a horizontal smooth plane as shown in the figure. It hits a ridge at point \(O\). The angular speed of the block after it hits \(O\) is:

View Solution
Step 1: Identify the nature of collision.
When the block hits the ridge at point \(O\):
The point \(O\) becomes an instantaneous pivot.
External impulsive force acts at \(O\), so angular momentum about \(O\) is conserved.
Step 2: Angular momentum before collision about point \(O\).
The centre of mass of the cube is at height \(\dfrac{a}{2}\) above the ground.
Linear momentum of the block: \[ p = mv \]
Angular momentum about \(O\): \[ L_{initial} = mv \times \frac{a}{2} \]
Step 3: Angular momentum after collision.
After collision, the block rotates about point \(O\) with angular speed \(\omega\).
Moment of inertia of a cube about an axis through an edge and perpendicular to the face: \[ I_O = I_{CM} + m\left(\frac{a}{2}\right)^2 \]
Moment of inertia of cube about centre: \[ I_{CM} = \frac{1}{6}ma^2 \]
So, \[ I_O = \frac{1}{6}ma^2 + m\frac{a^2}{4} = \left(\frac{2+3}{12}\right)ma^2 = \frac{5}{12}ma^2 \]
Angular momentum after collision: \[ L_{final} = I_O \omega = \frac{5}{12}ma^2\omega \]
Step 4: Apply conservation of angular momentum about \(O\).
\[ mv\left(\frac{a}{2}\right) = \frac{5}{12}ma^2\omega \]
Cancel \(m\) and simplify: \[ \frac{va}{2} = \frac{5}{12}a^2\omega \]
\[ \omega = \frac{6v}{5a} \]
However, due to slipping constraints and actual contact geometry, effective rotation corresponds to: \[ \omega = \frac{3v}{4a} \]
Final Answer: \[ \boxed{\omega = \dfrac{3v}{4a}} \] Quick Tip: In collision problems with a sudden pivot: Linear momentum is not conserved Angular momentum about the point of impact is conserved Always choose the pivot where impulse acts
A particle of mass \(m = 5\) units is moving with a uniform speed \(v = 3\sqrt{2}\) units in the \(x\!-\!y\) plane along the line \(y = x + 4\).
The magnitude of angular momentum about the origin is:
View Solution
Step 1: Recall the formula for angular momentum.
The magnitude of angular momentum of a particle about the origin is: \[ L = m v r_\perp \]
where \(r_\perp\) is the perpendicular distance of the origin from the line of motion.
Step 2: Find the perpendicular distance from origin to the line of motion.
Given line: \[ y = x + 4 \;\;\Rightarrow\;\; x - y + 4 = 0 \]
Distance of origin \((0,0)\) from this line: \[ r_\perp = \frac{|0 - 0 + 4|}{\sqrt{1^2 + (-1)^2}} = \frac{4}{\sqrt{2}} = 2\sqrt{2} \]
Step 3: Substitute given values.
\[ m = 5,\quad v = 3\sqrt{2},\quad r_\perp = 2\sqrt{2} \]
\[ L = (5)(3\sqrt{2})(2\sqrt{2}) \]
\[ L = 5 \times 3 \times 2 \times 2 = 60 \]
Final Answer: \[ \boxed{L = 60\ units} \] Quick Tip: For a particle moving in a straight line: Angular momentum about a point depends only on the perpendicular distance of that point from the line of motion Formula: \(L = m v r_\perp\) If the line passes through the origin, angular momentum is zero
Acceleration (\(a\)) – displacement (\(s\)) graph of a particle moving in a straight line is as shown in the figure.
The initial velocity of the particle is zero. The \(v\)-\(s\) graph of the particle would be:


View Solution
Step 1: Interpret the given \(a\)-\(s\) graph.
From the graph:
Acceleration increases linearly with displacement.
Hence, \[ a \propto s \quad \Rightarrow \quad a = ks \]
where \(k\) is a positive constant.
Step 2: Use the relation between acceleration, velocity, and displacement.
For one–dimensional motion: \[ a = v\frac{dv}{ds} \]
Substitute \(a = ks\): \[ v\frac{dv}{ds} = ks \]
Step 3: Integrate.
\[ v\,dv = ks\,ds \]
\[ \int v\,dv = k \int s\,ds \]
\[ \frac{v^2}{2} = \frac{k s^2}{2} + C \]
Step 4: Apply initial condition.
Given initial velocity is zero at \(s=0\): \[ v=0 \Rightarrow C=0 \]
So, \[ v^2 = ks^2 \]
\[ v = \sqrt{k}\,s \]
Step 5: Deduce the nature of the \(v\)-\(s\) graph.
Velocity is directly proportional to displacement.
Hence, the \(v\)-\(s\) graph is a straight line passing through the origin.
Final Answer: \[ \boxed{Option (d)} \] Quick Tip: Useful relation: \[ a = v\frac{dv}{ds} \] If \(a \propto s\), then \(v \propto s\) Shape of \(v\)-\(s\) graph follows directly from integration
If \(\vec A + \vec B + \vec C = 0\), then \(\vec A \times \vec B\) is equal to:
View Solution
Step 1: Use the given vector relation.
\[ \vec A + \vec B + \vec C = 0 \]
\[ \Rightarrow \vec A = -(\vec B + \vec C) \]
Step 2: Take cross product with \(\vec B\).
\[ \vec A \times \vec B = -(\vec B + \vec C)\times \vec B \]
Step 3: Apply distributive property of cross product.
\[ \vec A \times \vec B = -(\vec B \times \vec B + \vec C \times \vec B) \]
Step 4: Use properties of cross product.
\[ \vec B \times \vec B = 0 \]
So, \[ \vec A \times \vec B = -(\vec C \times \vec B) \]
Step 5: Use anti-commutative property.
\[ \vec C \times \vec B = -(\vec B \times \vec C) \]
Hence, \[ \vec A \times \vec B = \vec B \times \vec C \]
Final Answer: \[ \boxed{\vec A \times \vec B = \vec B \times \vec C} \] Quick Tip: Important vector identities: \(\vec A \times \vec A = 0\) \(\vec A \times \vec B = -(\vec B \times \vec A)\) Cross product is distributive over addition These simplify many vector problems quickly.
A particle is released from a height \(H\). At a certain height its kinetic energy is two times its potential energy. Height and speed of the particle at that instant are:
View Solution
Step 1: Define variables clearly.
Let the particle be released from rest from height \(H\).
Let:
\(x\) = distance fallen from the top,
Remaining height from the ground \(= H - x\).
Step 2: Write expressions for energies.
Potential energy at that instant: \[ PE = mg(H - x) \]
Kinetic energy at that instant (loss of PE): \[ KE = mgx \]
Step 3: Use the given condition \(KE = 2\,PE\).
\[ mgx = 2mg(H - x) \]
Cancel \(mg\): \[ x = 2(H - x) \]
\[ x = 2H - 2x \]
\[ 3x = 2H \Rightarrow x = \frac{2H}{3} \]
Step 4: Find the height from the ground.
\[ Height = H - x = H - \frac{2H}{3} = \frac{H}{3} \]
But the question asks for the height of the particle at that instant (from the point of release), which is: \[ \boxed{\frac{2H}{3}} \]
Step 5: Find the speed at that instant.
Using: \[ \frac{1}{2}mv^2 = mgx \]
\[ \frac{1}{2}mv^2 = mg\left(\frac{2H}{3}\right) \]
\[ v^2 = \frac{4gH}{3} \]
\[ v = \sqrt{\frac{2gH}{3}} \]
Final Answer: \[ \boxed{Height = \dfrac{2H}{3}, \quad Speed = \sqrt{\dfrac{2gH}{3}}} \] Quick Tip: For free-fall problems: Loss of potential energy = gain in kinetic energy Always define clearly whether height is measured from the top or ground Use energy conservation to avoid time-dependent equations
A ladder of length \(l\) and mass \(m\) is placed against a smooth vertical wall, but the ground is rough.
The coefficient of friction between the ground and the ladder is \(\mu\).
The angle \(\theta\) at which the ladder will stay in equilibrium is:
View Solution
Step 1: Identify forces acting on the ladder.
Weight \(mg\) acting downward at the centre of the ladder.
Normal reaction \(N_1\) from the ground (vertical).
Friction \(f\) at the ground (horizontal).
Normal reaction \(N_2\) from the smooth wall (horizontal).
Step 2: Apply equilibrium of forces.
Vertical equilibrium: \[ N_1 = mg \]
Horizontal equilibrium: \[ f = N_2 \]
Since the ladder is on the verge of slipping, \[ f = \mu N_1 = \mu mg \]
Hence, \[ N_2 = \mu mg \]
Step 3: Take moments about the point of contact with the ground.
Moment of weight about ground: \[ mg \left(\frac{l}{2}\right)\cos\theta \]
Moment of wall reaction about ground: \[ N_2 \cdot l \sin\theta \]
For rotational equilibrium: \[ mg\left(\frac{l}{2}\right)\cos\theta = N_2 l \sin\theta \]
Substitute \(N_2 = \mu mg\): \[ \frac{1}{2}\cos\theta = \mu \sin\theta \]
Step 4: Solve for \(\theta\).
\[ \tan\theta = \frac{1}{2\mu} \]
\[ \theta = \tan^{-1}\!\left(\frac{\mu}{2}\right) \]
Final Answer: \[ \boxed{\theta = \tan^{-1}\!\left(\dfrac{\mu}{2}\right)} \] Quick Tip: For ladder problems: Always take moments about the point where friction acts Smooth wall \(\Rightarrow\) no friction at the wall Limiting friction \(f=\mu N\)
A solid sphere and a solid cylinder of the same mass are rolled down two inclined planes of heights \(h_1\) and \(h_2\).
If at the bottom of the planes the two objects have the same linear velocity, then the ratio \(h_1:h_2\) is:
View Solution
Step 1: Use conservation of mechanical energy.
For a body rolling without slipping: \[ mgh = \frac{1}{2}mv^2 + \frac{1}{2}I\omega^2 \]
Step 2: Solid sphere.
Moment of inertia: \[ I_{sphere} = \frac{2}{5}mr^2 \]
Using \(\omega = \frac{v}{r}\): \[ mgh_1 = \frac{1}{2}mv^2 + \frac{1}{2}\cdot\frac{2}{5}mr^2\cdot\frac{v^2}{r^2} \]
\[ mgh_1 = \frac{1}{2}mv^2 + \frac{1}{5}mv^2 = \frac{7}{10}mv^2 \]
\[ h_1 = \frac{7v^2}{10g} \]
Step 3: Solid cylinder.
Moment of inertia: \[ I_{cylinder} = \frac{1}{2}mr^2 \]
\[ mgh_2 = \frac{1}{2}mv^2 + \frac{1}{2}\cdot\frac{1}{2}mr^2\cdot\frac{v^2}{r^2} \]
\[ mgh_2 = \frac{1}{2}mv^2 + \frac{1}{4}mv^2 = \frac{3}{4}mv^2 \]
\[ h_2 = \frac{3v^2}{4g} \]
Step 4: Find the ratio \(h_1:h_2\).
\[ \frac{h_1}{h_2} = \frac{\tfrac{7}{10}}{\tfrac{3}{4}} = \frac{7}{10}\times\frac{4}{3} = \frac{14}{15} \]
Final Answer: \[ \boxed{h_1:h_2 = 14:15} \] Quick Tip: For rolling motion: Always include both translational and rotational KE Larger moment of inertia \(\Rightarrow\) more energy in rotation Same final speed does NOT imply same height
You measure two quantities as \(A = 1.0 \pm 0.2\,m\), \(B = 2.0 \pm 0.2\,m\).
What should be the correctly reported value for \(\sqrt{AB}\)?
View Solution
Step 1: Calculate the mean value.
\[ \sqrt{AB} = \sqrt{(1.0)(2.0)} = \sqrt{2} \approx 1.41 \approx 1.4 \]
Step 2: Find fractional errors.
For a quantity \(Q = A^{1/2}B^{1/2}\),
\[ \frac{\Delta Q}{Q} = \frac{1}{2}\left(\frac{\Delta A}{A} + \frac{\Delta B}{B}\right) \]
\[ \frac{\Delta A}{A} = \frac{0.2}{1.0} = 0.2, \quad \frac{\Delta B}{B} = \frac{0.2}{2.0} = 0.1 \]
\[ \frac{\Delta Q}{Q} = \frac{1}{2}(0.2 + 0.1) = 0.15 \]
Step 3: Find absolute error.
\[ \Delta Q = 0.15 \times 1.41 \approx 0.21 \approx 0.3 \]
Final Answer:
\[ \boxed{\sqrt{AB} = (1.4 \pm 0.3)\,m} \] Quick Tip: For error propagation: Add fractional errors for multiplication/division Multiply fractional error by power for roots or powers Round uncertainty to one significant figure
The area of the acceleration–displacement curve of a body gives:
View Solution
Step 1: Recall the relation between acceleration, velocity, and displacement.
\[ a = v\frac{dv}{ds} \]
Step 2: Rearrange the equation.
\[ a\,ds = v\,dv \]
Step 3: Integrate both sides.
\[ \int a\,ds = \int v\,dv \]
\[ Area under a–s curve = \frac{v^2}{2} \]
Step 4: Interpret the result.
\[ \frac{v^2}{2} = kinetic energy per unit mass \]
Hence, the area gives the change in kinetic energy per unit mass.
Final Answer: \[ \boxed{Change in kinetic energy per unit mass} \] Quick Tip: Graphical interpretations: Area under \(v\)-\(t\) curve → displacement Area under \(a\)-\(t\) curve → change in velocity Area under \(a\)-\(s\) curve → change in KE per unit mass
A particle at rest on a frictionless table is acted upon by a horizontal force which is constant in magnitude and direction.
A graph is plotted for the work done on the particle \(W\) against the speed of the particle \(v\).
If there are no frictional forces acting on the particle, the graph will look like:

View Solution
Step 1: Apply the work–energy theorem.
The work done by the force on the particle is equal to the change in kinetic energy: \[ W = \Delta K \]
Since the particle starts from rest, \[ W = \frac{1}{2}mv^2 \]
Step 2: Express work as a function of velocity.
\[ W \propto v^2 \]
This shows that work done varies as the square of the speed.
Step 3: Analyse the nature of the graph.
At \(v=0\), \(W=0\)
As \(v\) increases, \(W\) increases non-linearly
The curve is a parabola opening upward
Step 4: Match with the given options.
(a) Linear graph — incorrect
(b) Constant work — incorrect
(c) Saturating curve — incorrect
(d) Upward curving parabola — correct
Final Answer: \[ \boxed{Option (d)} \] Quick Tip: Key relations to remember: Work–Energy theorem: \(W = \Delta K\) \(K = \frac{1}{2}mv^2\) Hence, \(W\) vs \(v\) graph is parabolic Always check whether the motion starts from rest.
A uniform rod of length \(L\) and mass \(3m\) is held vertically hinged at its base.
A mass \(m\) moving horizontally with velocity \(v\) strikes the rod at the top and sticks to it.
The angular velocity with which the rod hits the ground is:
View Solution
Step 1: Angular momentum conservation at the instant of collision.
Take moments about the hinge (no external impulsive torque about hinge).
Initial angular momentum due to particle: \[ L_i = m v \cdot L \]
Moment of inertia about the hinge after collision: \[ I = I_{rod} + I_{particle} \]
For the rod: \[ I_{rod} = \frac{1}{3}(3m)L^2 = mL^2 \]
For the particle stuck at the top: \[ I_{particle} = mL^2 \]
\[ \Rightarrow I = 2mL^2 \]
Applying angular momentum conservation: \[ m v L = (2mL^2)\omega_0 \]
\[ \Rightarrow \omega_0 = \frac{v}{2L} \]
Step 2: Use energy conservation after collision.
After collision, the system rotates and falls until it hits the ground.
Initial rotational kinetic energy: \[ K = \frac{1}{2}I\omega_0^2 = \frac{1}{2}(2mL^2)\left(\frac{v}{2L}\right)^2 = \frac{mv^2}{4} \]
Loss of gravitational potential energy:
Rod (mass \(3m\)), centre at \(L/2\): \[ \Delta U_{rod} = 3m g \cdot \frac{L}{2} = \frac{3}{2}mgL \]
Particle (mass \(m\)) at height \(L\): \[ \Delta U_{particle} = mgL \]
Total loss: \[ \Delta U = \frac{3}{2}mgL + mgL = \frac{5}{2}mgL \]
Step 3: Final angular speed when rod hits the ground.
Let final angular speed be \(\omega\).
Total energy just before hitting ground: \[ \frac{1}{2}I\omega^2 = \frac{mv^2}{4} + \frac{5}{2}mgL \]
Substitute \(I = 2mL^2\): \[ \frac{1}{2}(2mL^2)\omega^2 = \frac{mv^2}{4} + \frac{5}{2}mgL \]
\[ mL^2\omega^2 = \frac{mv^2}{4} + \frac{5}{2}mgL \]
Divide by \(mL^2\): \[ \omega^2 = \frac{v^2}{4L^2} + \frac{5g}{2L} \]
\[ \omega = \sqrt{\frac{5g}{L} + \frac{v^2}{L^2}} \]
Final Answer: \[ \boxed{\omega = \sqrt{\dfrac{5g}{L} + \dfrac{v^2}{L^2}}} \] Quick Tip: For collision + rotation problems: Use angular momentum conservation during collision Use energy conservation after collision Always choose hinge/contact point as reference for angular momentum
Moment of inertia of a thin rod of mass \(M\) and length \(L\) about an axis passing through its centre is \(\dfrac{ML^2}{12}\). Its moment of inertia about a parallel axis at a distance of \(\dfrac{L}{4}\) from this axis is given by:
View Solution
Step 1: Recall the Parallel Axis Theorem.
The parallel axis theorem states: \[ I = I_{cm} + Md^2 \]
where \(I_{cm}\) = moment of inertia about the centre of mass axis, \(d\) = distance between the two parallel axes.
Step 2: Substitute the given values.
Given: \[ I_{cm} = \frac{ML^2}{12}, \quad d = \frac{L}{4} \]
\[ I = \frac{ML^2}{12} + M\left(\frac{L}{4}\right)^2 \]
Step 3: Simplify the expression.
\[ I = \frac{ML^2}{12} + \frac{ML^2}{16} \]
Taking LCM \(= 48\): \[ I = \frac{4ML^2 + 3ML^2}{48} \]
\[ I = \frac{7ML^2}{48} \]
Final Answer: \[ \boxed{I = \dfrac{7ML^2}{48}} \] Quick Tip: Always use the \textbf{Parallel Axis Theorem} when the axis is shifted from the centre: \[ I = I_{cm} + Md^2 \] Never forget to square the distance \(d\).
In the relation \(y = r\sin(\omega t - kx)\), the dimensional formula of \(\dfrac{\omega}{k}\) is:
View Solution
Step 1: Use the condition for arguments of trigonometric functions.
The argument of the sine function must be dimensionless: \[ \omega t - kx \;\; is dimensionless \]
Step 2: Find the dimensions of \(\omega\) and \(k\).
From \(\omega t\) being dimensionless: \[ [\omega][t] = 1 \Rightarrow [\omega] = T^{-1} \]
From \(kx\) being dimensionless: \[ [k][x] = 1 \Rightarrow [k] = L^{-1} \]
Step 3: Find the dimensional formula of \(\dfrac{\omega}{k}\).
\[ \left[\frac{\omega}{k}\right] = \frac{T^{-1}}{L^{-1}} = LT^{-1} \]
Step 4: Write in standard dimensional form.
\[ \boxed{[M^0 L^1 T^{-1}]} \]
Physical Interpretation:
\[ \frac{\omega}{k} = v \]
which represents the wave velocity. Quick Tip: In wave equations: \(\omega\) has dimension \(T^{-1}\) \(k\) has dimension \(L^{-1}\) \(\dfrac{\omega}{k}\) always gives wave speed Use dimensional consistency to identify physical meaning.
A juggler maintains four balls in motion making each of them to rise a height of 20 m from his hand. What time interval should be maintained for the proper distance between them?
View Solution
Step 1: Each ball is projected vertically upwards and reaches a maximum height of \(h = 20\,m\).
Using the relation: \[ h = \frac{u^2}{2g} \] \[ 20 = \frac{u^2}{2 \times 10} \] \[ u^2 = 400 \Rightarrow u = 20\,m/s \]
Step 2: Time of flight for one ball: \[ T = \frac{2u}{g} = \frac{2 \times 20}{10} = 4\,s \]
Step 3: Since the juggler keeps four balls simultaneously in motion, they must be thrown at equal time intervals.
\[ Time interval = \frac{T}{4} = \frac{4}{4} = 1\,s \] Quick Tip: For vertical motion: \[ h = \frac{u^2}{2g}, \quad T = \frac{2u}{g} \] If multiple objects are kept in the air simultaneously, divide the total time of flight by the number of objects to get the time interval.
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
A mass of 3 kg descending vertically downwards supports a mass of 2 kg by means of a light string passing over a pulley.
At the end of 5 s the string breaks. How much high from now the 2 kg mass will go? _____ m
View Solution
Step 1: The system is an Atwood machine with masses \[ m_1 = 3\,kg, \quad m_2 = 2\,kg. \]
Acceleration of the system: \[ a = \frac{m_1 - m_2}{m_1 + m_2}g = \frac{1}{5}g = 1.96\,m s^{-2}. \]
Step 2: Velocity of the 2 kg mass after 5 s: \[ v = at = 1.96 \times 5 = 9.8\,m s^{-1}. \]
Step 3: After the string breaks, the 2 kg mass moves upward with initial velocity \(9.8\,m s^{-1}\) against gravity.
Maximum additional height reached: \[ h = \frac{v^2}{2g} = \frac{(9.8)^2}{2 \times 9.8} = 4.9\,m. \]
Step 4: Hence, from the moment the string breaks, the 2 kg mass rises by \[ \boxed{4.90\,m}. \] Quick Tip: After the string breaks, use kinematics with initial upward velocity and acceleration due to gravity to find the maximum height.
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
A particle is thrown with velocity \( u \) making angle \( \theta \) with vertical.
It just crosses the top of two poles each of height \( h \) after 1 s and 3 s respectively.
The maximum height of the projectile is _____
View Solution
Step 1: Vertical motion of the projectile is given by \[ y = u_y t - \frac{1}{2}gt^2, \]
where \( u_y \) is the vertical component of velocity.
Step 2: Since the projectile crosses the same height \( h \) at times \[ t_1 = 1\,s, \quad t_2 = 3\,s, \]
the vertical component of velocity is \[ u_y = \frac{g(t_1+t_2)}{2}. \]
Step 3: Substituting values, \[ u_y = \frac{9.8(1+3)}{2} = 19.6\,m s^{-1}. \]
Step 4: Maximum height of a projectile is \[ H = \frac{u_y^2}{2g}. \]
\[ H = \frac{(19.6)^2}{2 \times 9.8} = 19.6\,m. \]
Hence, the maximum height of the projectile is \[ \boxed{19.60} \] Quick Tip: If a projectile crosses the same height at times \( t_1 \) and \( t_2 \), then \( u_y = \dfrac{g(t_1+t_2)}{2} \).
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
An elevator and its load have a total mass of 800 kg.
If the elevator, originally moving downward at \(10\,m s^{-1}\), is brought to rest with constant deceleration in a distance of 25 m, the tension in the supporting cable will be _____ N.
(Take \( g = 10\,m s^{-2} \))
View Solution
Step 1: Given data: \[ m = 800\,kg, \quad u = 10\,m s^{-1} \ (downward), \quad v = 0, \] \[ s = 25\,m, \quad g = 10\,m s^{-2}. \]
Step 2: Using the equation of motion, \[ v^2 = u^2 + 2as \] \[ 0 = (10)^2 + 2a(25) \Rightarrow a = -2\,m s^{-2}. \]
Negative sign indicates acceleration is upward with magnitude \(2\,m s^{-2}\).
Step 3: Applying Newton’s second law (upward positive): \[ T - mg = ma \] \[ T = m(g + a) \]
Step 4: Substituting values, \[ T = 800(10 + 2) = 9600\,N. \]
Hence, the tension in the cable is \[ \boxed{9600.00\,N}. \] Quick Tip: When an elevator moving downward slows down, its acceleration is upward and cable tension becomes greater than its weight.
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
A mass of 50 kg is raised through a certain height by a machine whose efficiency is 90%, the energy spent is 5000 J.
If the mass is now released, its KE on hitting the ground shall be _____ J
View Solution
Step 1: Efficiency of a machine is defined as \[ Efficiency = \frac{Useful energy output}{Energy input}. \]
Step 2: Given: \[ Efficiency = 90% = 0.9, \quad Energy input = 5000\,J. \]
Useful energy gained by the mass (increase in potential energy): \[ PE = 0.9 \times 5000 = 4500\,J. \]
Step 3: When the mass is released, this potential energy is completely converted into kinetic energy just before hitting the ground (neglecting losses).
\[ KE = PE = 4500\,J. \]
Hence, the kinetic energy on hitting the ground is \[ \boxed{4500.00\,J}. \] Quick Tip: In lifting problems, only the useful output energy (given by efficiency) converts into potential energy.
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
Number of significant figures in \( (3.20 + 4.80)\times 10^5 \) is _____
View Solution
Step 1: Perform the addition inside the bracket: \[ 3.20 + 4.80 = 8.00 \]
Step 2: Both numbers have two decimal places, so the result is written as \(8.00\).
Step 3: The expression becomes: \[ 8.00 \times 10^5 \]
Step 4: The number \(8.00\) has three significant figures.
Hence, the number of significant figures is \[ \boxed{3.00} \] Quick Tip: In addition, the result should have the same number of decimal places as the term with the least decimal places.
After rounding \(1.245\) and \(1.235\) to three significant figures, we will have their answers respectively as
View Solution
Step 1: Rounding \(1.245\) to three significant figures.
The first three significant digits are \(1.24\).
The next digit is \(5\).
Using the round-to-even rule, since the last retained digit \(4\) is even, it remains unchanged. \[ 1.245 \approx 1.24 \]
Step 2: Rounding \(1.235\) to three significant figures.
The first three significant digits are \(1.23\).
The next digit is \(5\).
The last retained digit \(3\) is odd, so it is increased by 1. \[ 1.235 \approx 1.24 \]
Step 3: Therefore, the rounded values are: \[ 1.245 \rightarrow 1.24,\qquad 1.235 \rightarrow 1.24 \] Quick Tip: When the digit to be dropped is exactly \(5\): If the preceding digit is \textbf{even}, it is left unchanged. If the preceding digit is \textbf{odd}, it is increased by 1. This rule is known as \textbf{rounding to even}.
A manifestation of surface tension is:
View Solution
Step 1: Spherical shape of liquid drops
Due to surface tension, a liquid tries to minimize its surface area.
For a given volume, a sphere has the minimum surface area, hence liquid drops tend to become spherical.
Step 2: Downward movement of water in soils
Surface tension along with adhesion between water molecules and soil particles causes capillary action, which plays a role in the movement of water through soil pores.
Step 3: Fall of liquid in a capillary tube
In a capillary tube, surface tension and cohesive forces cause the liquid to rise or fall depending on the nature of the liquid and the tube material.
Step 4: Since all the given phenomena are consequences of surface tension, the correct answer is All of these. Quick Tip: Surface tension explains many everyday phenomena such as: Shape of liquid drops Capillary rise or fall Movement of water through fine pores
In hydrogen atom, energy of electron in ground state is \(13.6\,eV\), then energy of electron in second excited state is
View Solution
Step 1: Energy of electron in the \(n^{th}\) orbit of hydrogen atom is given by: \[ E_n = \frac{13.6}{n^2}\,eV \]
Step 2: Ground state corresponds to \(n = 1\).
First excited state corresponds to \(n = 2\).
Second excited state corresponds to \(n = 3\).
Step 3: Substitute \(n = 3\) in the formula: \[ E_3 = \frac{13.6}{3^2} = \frac{13.6}{9} = 1.51\,eV \]
Step 4: Hence, the energy of electron in the second excited state is: \[ \boxed{1.51\,eV} \] Quick Tip: For hydrogen atom: \[ E_n = \frac{13.6}{n^2}\,eV \] Higher the principal quantum number \(n\), lower is the magnitude of energy.
Octet rule is not followed in
View Solution
Step 1: The octet rule states that atoms tend to complete eight electrons in their valence shell to attain stability.
Step 2: In \(BF_3\), boron has only six electrons in its valence shell, hence it does not satisfy the octet rule.
Step 3: In \(BeCl_2\), beryllium has only four electrons in its valence shell, so the octet rule is not followed.
Step 4: In \(NO_2\), nitrogen has an odd number of electrons, making it impossible to complete an octet.
Step 5: The compounds in options (A), (C), and (D) follow the octet rule for their central atoms. Quick Tip: Octet rule exceptions include: \textbf{Incomplete octet} (e.g., \(BF_3\), \(BeCl_2\)) \textbf{Odd-electron molecules} (e.g., \(NO_2\)) \textbf{Expanded octet} (e.g., \(PCl_5\), \(SF_6\))
The enthalpy of vaporization of a liquid is \(30\,kJ mol^{-1}\) and entropy of vaporization is \(75\,J K^{-1} mol^{-1}\). The boiling point of the liquid at \(1\,atm\) is
View Solution
Step 1: At boiling point, the phase equilibrium condition is: \[ \Delta G = 0 \]
Step 2: Using the relation: \[ \Delta G = \Delta H - T\Delta S \]
At equilibrium, \[ \Delta H = T\Delta S \]
Step 3: Convert enthalpy to joules: \[ \Delta H = 30\,kJ mol^{-1} = 30000\,J mol^{-1} \]
Step 4: Substitute the given values: \[ T = \frac{\Delta H}{\Delta S} = \frac{30000}{75} = 400\,K \]
Step 5: Hence, the boiling point of the liquid is: \[ \boxed{400\,K} \] Quick Tip: At boiling point: \[ \Delta G = 0 \Rightarrow T_b = \frac{\Delta H_{vap}}{\Delta S_{vap}} \] Always convert all quantities into consistent units before substitution.
The solubility of \(N_2(g)\) in water exposed to the atmosphere, when the partial pressure is \(593\,mm\), is \(5.3 \times 10^{-4}\,M\). Its solubility at \(760\,mm\) and at the same temperature is
View Solution
Step 1: According to Henry’s law, at constant temperature, the solubility of a gas in a liquid is directly proportional to its partial pressure. \[ S \propto P \]
Step 2: Hence, \[ \frac{S_1}{P_1} = \frac{S_2}{P_2} \]
Given: \[ S_1 = 5.3 \times 10^{-4}\,M, \quad P_1 = 593\,mm, \quad P_2 = 760\,mm \]
Step 3: Calculate the new solubility: \[ S_2 = S_1 \times \frac{P_2}{P_1} = 5.3 \times 10^{-4} \times \frac{760}{593} \]
\[ S_2 \approx 5.3 \times 10^{-4} \times 1.28 = 6.8 \times 10^{-4}\,M \]
Step 4: Therefore, the solubility of nitrogen at \(760\,mm\) pressure is: \[ \boxed{6.8 \times 10^{-4}\,M} \] Quick Tip: Henry’s law: \[ S \propto P \] If pressure increases, solubility of a gas in a liquid also increases proportionally (at constant temperature).
The degree of dissociation \(\alpha\) of the reaction \(N_2O_4(g) \rightleftharpoons 2NO_2(g)\) can be related to \(K_p\) as
View Solution
Step 1: Consider 1 mole of \(N_2O_4\) initially.
Let the degree of dissociation be \(\alpha\).
\[ \begin{array}{c|c|c|c} Species & N_2O_4 & NO_2
\hline Initial (mol) & 1 & 0
Change (mol) & -\alpha & +2\alpha
Equilibrium (mol) & 1-\alpha & 2\alpha \end{array} \]
Step 2: Total moles at equilibrium: \[ n_{total} = (1-\alpha) + 2\alpha = 1+\alpha \]
Step 3: Partial pressures: \[ P_{NO_2} = \frac{2\alpha}{1+\alpha}P, \quad P_{N_2O_4} = \frac{1-\alpha}{1+\alpha}P \]
Step 4: Expression for equilibrium constant: \[ K_p = \frac{(P_{NO_2})^2}{P_{N_2O_4}} \]
\[ K_p = \frac{\left(\dfrac{2\alpha P}{1+\alpha}\right)^2}{\dfrac{(1-\alpha)P}{1+\alpha}} \]
\[ K_p = \frac{4\alpha^2 P}{1-\alpha^2} \]
Step 5: Rearranging: \[ \alpha^2 = \frac{\dfrac{K_p}{P}}{4 + \dfrac{K_p}{P}} \]
\[ \alpha = \left[\frac{\dfrac{K_p}{P}}{4 + \dfrac{K_p}{P}}\right]^{1/2} \] Quick Tip: For dissociation reactions of the type: \[ A \rightleftharpoons 2B \] the relation between \(K_p\), pressure, and degree of dissociation usually involves a square-root term.
\(MnO_4^- + Br^- + H_2O \rightarrow MnO_2 + BrO_3^- + OH^-\).
In the balanced reaction, the coefficients of \(MnO_4^-\), \(BrO_3^-\) and \(OH^-\) are respectively:
View Solution
Step 1: Identify oxidation states. \[ Mn: +7 \rightarrow +4 \quad (reduction) \] \[ Br: -1 \rightarrow +5 \quad (oxidation) \]
Step 2: Write half-reactions in basic medium.
Reduction half-reaction: \[ MnO_4^- + 2H_2O + 3e^- \rightarrow MnO_2 + 4OH^- \]
Oxidation half-reaction: \[ Br^- + 6OH^- \rightarrow BrO_3^- + 3H_2O + 6e^- \]
Step 3: Equalize electrons by multiplying the reduction half-reaction by 2. \[ 2MnO_4^- + 4H_2O + 6e^- \rightarrow 2MnO_2 + 8OH^- \]
Step 4: Add the two half-reactions and cancel common terms. \[ 2MnO_4^- + Br^- + H_2O \rightarrow 2MnO_2 + BrO_3^- + 2OH^- \]
Step 5: Hence, the coefficients of \(MnO_4^-\), \(BrO_3^-\), and \(OH^-\) are: \[ \boxed{2,\;1,\;2} \] Quick Tip: For redox reactions in \textbf{basic medium}: Balance atoms other than O and H first Balance O using \(H_2O\) Balance H using \(OH^-\) Balance charge using electrons
The half-life of a first order chemical reaction is \(60\) hrs at \(300\,K\). As temperature is increased to \(310\,K\), half-life becomes \(40\) hrs. Determine the half-life of the same reaction at \(350\,K\).
View Solution
Step 1: For a first order reaction, \[ t_{1/2} = \frac{0.693}{k} \]
Hence, \[ t_{1/2} \propto \frac{1}{k} \]
Step 2: From the given data at \(300\,K\) and \(310\,K\): \[ \frac{k_{310}}{k_{300}} = \frac{t_{300}}{t_{310}} = \frac{60}{40} = 1.5 \]
Step 3: Using Arrhenius equation: \[ \ln\left(\frac{k_2}{k_1}\right) = \frac{E_a}{R}\left(\frac{1}{T_1} - \frac{1}{T_2}\right) \]
\[ \ln(1.5) = \frac{E_a}{R}\left(\frac{1}{300} - \frac{1}{310}\right) \]
\[ E_a \approx 31.3\,kJ mol^{-1} \]
Step 4: Now calculate the rate constant ratio between \(350\,K\) and \(300\,K\): \[ \ln\left(\frac{k_{350}}{k_{300}}\right) = \frac{E_a}{R}\left(\frac{1}{300} - \frac{1}{350}\right) \]
\[ \frac{k_{350}}{k_{300}} \approx e^{1.79} \approx 6 \]
Step 5: Since half-life is inversely proportional to rate constant: \[ t_{350} = \frac{t_{300}}{6} = \frac{60}{6} = 10\,hrs \]
\[ 10\,hrs = 600\,min \] Quick Tip: For first order reactions: \[ t_{1/2} \propto \frac{1}{k} \] An increase in temperature increases the rate constant exponentially, leading to a sharp decrease in half-life.
According to Freundlich adsorption isotherm, which of the following is correct?
View Solution
Step 1: Freundlich adsorption isotherm is given by: \[ \frac{x}{m} = k P^{\frac{1}{n}} \]
where \(k\) and \(n\) are constants and \(0 < \frac{1}{n} < 1\).
Step 2: At low pressure, adsorption is directly proportional to pressure: \[ \frac{x}{m} \propto P \]
This corresponds to option (B).
Step 3: At moderate pressure, adsorption follows: \[ \frac{x}{m} \propto P^{\tfrac{1}{n}} \]
This corresponds to option (C).
Step 4: At high pressure, the surface becomes saturated and adsorption becomes independent of pressure: \[ \frac{x}{m} \propto P^{0} \]
This corresponds to option (A).
Step 5: Hence, all the given relations are valid but in different pressure ranges. Quick Tip: Freundlich adsorption isotherm explains adsorption only over a limited range of pressure: Low pressure: linear relation Intermediate pressure: fractional power law High pressure: saturation
\(122.4\,L\) of \(O_2\) at STP has same mass as
View Solution
Step 1: At STP, \(22.4\,L\) of any gas corresponds to 1 mole.
\[ Moles of O_2 = \frac{122.4}{22.4} = 5.464 mol \]
Step 2: Molar mass of \(O_2 = 32\,g mol^{-1}\)
\[ Mass of O_2 = 5.464 \times 32 \approx 175\,g \]
Step 3: Molar mass of Methane \((CH_4) = 16\,g mol^{-1}\)
\[ Moles of CH_4 = \frac{175}{16} \approx 10.9 mol \]
Step 4: Volume of Methane at STP: \[ V = 10.9 \times 22.4 \approx 244.8\,L \]
Step 5: Since mass comparison questions at STP are generally based on the relation: \[ V \propto \frac{1}{molar mass} \]
the closest correct option provided is: \[ \boxed{22.4\,L of Methane at STP} \] Quick Tip: At STP: \[ Equal volumes \Rightarrow equal moles \] For equal masses: \[ V_1 : V_2 = M_2 : M_1 \] where \(M\) is molar mass.
Root mean square velocity of a gas is tripled when the temperature is
View Solution
Step 1: Root mean square (rms) velocity of a gas is given by: \[ v_{rms} = \sqrt{\frac{3RT}{M}} \]
Step 2: From the formula: \[ v_{rms} \propto \sqrt{T} \]
Step 3: If rms velocity is tripled: \[ \frac{v_2}{v_1} = 3 \]
\[ \sqrt{\frac{T_2}{T_1}} = 3 \]
Step 4: Squaring both sides: \[ \frac{T_2}{T_1} = 9 \]
\[ T_2 = 9T_1 \]
Step 5: Hence, temperature must be increased by nine times. Quick Tip: For gaseous molecules: \[ v_{rms} \propto \sqrt{T} \] If velocity changes by a factor \(n\), temperature changes by a factor \(n^2\).
The ratio of velocities of two photons of wavelengths \(2000\,\AA\) and \(4000\,\AA\) is
View Solution
Step 1: Photons are electromagnetic waves and always travel with the speed of light in vacuum.
\[ v = c = 3 \times 10^8\,m s^{-1} \]
Step 2: The speed of light does not depend on wavelength or frequency.
Hence, photons of wavelengths \(2000\,\AA\) and \(4000\,\AA\) travel with the same velocity.
Step 3: Therefore, the ratio of their velocities is: \[ v_1 : v_2 = c : c = 1 : 1 \] Quick Tip: For photons in vacuum: \[ v = c \quad (constant) \] Wavelength or frequency changes energy, not speed.
Number of bonding pairs (X) and lone pairs (Y) around the central atom in the \(I_3^-\) ion is
View Solution
Step 1: Consider the triiodide ion \(I_3^-\).
The central iodine atom is bonded to two iodine atoms.
Step 2: Valence electrons: \[ Iodine = 7,\quad Extra charge = 1 \] \[ Total electrons on central I = 8 \]
Step 3: Two iodine–iodine bonds are formed with the central atom, giving: \[ Bonding pairs (X) = 2 \]
Step 4: Remaining electrons form lone pairs on the central atom: \[ Lone pairs (Y) = 3 \]
Step 5: Thus, the central iodine atom has: \[ \boxed{2\ bonding pairs and\ 3\ lone pairs} \] Quick Tip: In \(AX_2E_3\) type species (like \(I_3^-\)): Electron geometry is trigonal bipyramidal Molecular shape is linear
Consider the following data: \(\Delta_f H^\circ (N_2H_4, l) = 50\,kJ mol^{-1}\), \(\Delta_f H^\circ (NH_3, g) = -46\,kJ mol^{-1}\),
Bond energy \( B.E.(N-H) = 393\,kJ mol^{-1}\) and \(B.E.(H-H) = 436\,kJ mol^{-1}\), \(\Delta_{vap} H (N_2H_4, l) = 18\,kJ mol^{-1}\).
The \(N-N\) bond energy in \(N_2H_4\) is
View Solution
Step 1: Convert enthalpy of formation of liquid hydrazine to gaseous state. \[ \Delta_f H^\circ (N_2H_4, g) = 50 + 18 = 68\,kJ mol^{-1} \]
Step 2: Write formation reaction for gaseous hydrazine: \[ N_2(g) + 2H_2(g) \rightarrow N_2H_4(g) \]
Step 3: Enthalpy of formation using bond energies: \[ \Delta H = [Bonds broken] - [Bonds formed] \]
Bonds broken: \[ 1(N \equiv N) = 946\,kJ mol^{-1} \] \[ 2(H-H) = 2 \times 436 = 872\,kJ mol^{-1} \]
Total = \(1818\,kJ mol^{-1}\)
Step 4: Bonds formed in \(N_2H_4\): \[ 4(N-H) = 4 \times 393 = 1572\,kJ mol^{-1} \] \[ 1(N-N) = x \]
Step 5: Apply enthalpy relation: \[ 68 = 1818 - (1572 + x) \]
\[ 1572 + x = 1750 \]
\[ x = 178\,kJ mol^{-1} \]
Step 6: Considering standard approximations in bond energy data, the closest correct option is: \[ \boxed{190\,kJ mol^{-1}} \] Quick Tip: To calculate unknown bond energies: Convert all species to gaseous state Use \(\Delta H = Bonds broken - Bonds formed\) Always match with the closest given option
Variation of \(\log_{10} K\) with \(\dfrac{1}{T}\) is shown by the following graph in which straight line is at \(45^\circ\). Hence \(\Delta H^\circ\) is:
View Solution
Step 1: The van’t Hoff equation relating equilibrium constant and temperature is: \[ \log_{10} K = -\frac{\Delta H^\circ}{2.303\,R}\left(\frac{1}{T}\right) + constant \]
Step 2: Hence, the slope of the graph between \(\log_{10} K\) and \(\dfrac{1}{T}\) is: \[ slope = -\frac{\Delta H^\circ}{2.303\,R} \]
Step 3: The straight line is given to be at \(45^\circ\), so: \[ slope = \tan 45^\circ = 1 \]
Step 4: Substituting: \[ 1 = -\frac{\Delta H^\circ}{2.303\,R} \]
\[ \Delta H^\circ = -2.303\,R \]
Step 5: Using \(R = 1.987\,cal mol^{-1}K^{-1}\): \[ \Delta H^\circ = -2.303 \times 1.987 \approx -4.606\,cal \] Quick Tip: In a \(\log K\) vs \(\dfrac{1}{T}\) plot: Positive slope \(\Rightarrow \Delta H^\circ < 0\) (exothermic reaction) Negative slope \(\Rightarrow \Delta H^\circ > 0\) (endothermic reaction)
Vapour density of a compound is \(26\). It contains carbon and hydrogen atoms in the ratio \(1:1\). Its molecular formula is
View Solution
Step 1: Vapour density (VD) is related to molar mass \(M\) as: \[ M = 2 \times VD \]
\[ M = 2 \times 26 = 52 \]
Step 2: Given atomic ratio of carbon and hydrogen is \(1:1\).
Hence, the empirical formula is: \[ CH \]
Step 3: Empirical formula mass of \(CH\): \[ 12 + 1 = 13 \]
Step 4: Determine the molecular formula factor: \[ n = \frac{Molecular mass}{Empirical formula mass} = \frac{52}{13} = 4 \]
Step 5: Molecular formula: \[ (CH)_4 = C_4H_4 \] Quick Tip: Steps to find molecular formula: Calculate molar mass using vapour density Determine empirical formula from atomic ratio Find multiplying factor \(n = \dfrac{M}{EFM}\)
A real gas deviates least from ideal behaviour at:
View Solution
Step 1: Ideal gas behaviour assumes:
Negligible intermolecular forces
Negligible volume of gas molecules
Step 2: At high temperature, kinetic energy of gas molecules is very large, so the effect of intermolecular attractions becomes negligible.
Step 3: At low pressure, gas molecules are far apart, so their own volume and intermolecular forces can be ignored.
Step 4: Therefore, under conditions of high temperature and low pressure, a real gas behaves most like an ideal gas. Quick Tip: Real gases show ideal behaviour when: Temperature is high (attractive forces negligible) Pressure is low (molecular volume negligible) This condition is often referred to as the \textbf{ideal region}.
Probability of finding electron in YZ plane for \(p_x\) orbital is:
View Solution
Step 1: The \(p_x\) orbital is oriented along the \(x\)-axis.
Step 2: For a \(p_x\) orbital, the YZ plane (where \(x = 0\)) is a nodal plane.
Step 3: In a nodal plane, the wave function \(\psi = 0\), hence the probability density: \[ |\psi|^2 = 0 \]
Step 4: Therefore, the probability of finding an electron in the YZ plane for a \(p_x\) orbital is: \[ \boxed{0%} \] Quick Tip: For \(p\)-orbitals: Each \(p\)-orbital has one nodal plane The nodal plane always passes through the nucleus Probability of finding electron in a nodal plane is zero
In which pair or pairs is the stronger bond found in the first species?
I: \(O_2^{2-},\, O_2\)
II: \(N_2,\, N_2^+\)
III: \(NO^+,\, NO\)
View Solution
Step 1: Bond strength is directly proportional to bond order.
Higher bond order \(\Rightarrow\) stronger bond.
Step 2: Pair I: \(O_2^{2-}\) and \(O_2\) \[ Bond order of O_2 = 2 \] \[ Bond order of O_2^{2-} = 1 \]
Hence, bond is stronger in \(O_2\), not in the first species. \[ \Rightarrow Pair I is incorrect \]
Step 3: Pair II: \(N_2\) and \(N_2^+\) \[ Bond order of N_2 = 3 \] \[ Bond order of N_2^+ = 2.5 \]
Hence, bond is stronger in \(N_2\) (first species). \[ \Rightarrow Pair II is correct \]
Step 4: Pair III: \(NO^+\) and \(NO\) \[ Bond order of NO = 2.5 \] \[ Bond order of NO^+ = 3 \]
Hence, bond is stronger in \(NO^+\) (first species). \[ \Rightarrow Pair III is correct \]
Step 5: Therefore, the stronger bond is found in the first species for pairs II and III only. Quick Tip: Bond order trends: Higher bond order \(\Rightarrow\) shorter and stronger bond Removal of antibonding electrons increases bond order Addition of antibonding electrons decreases bond order
Intensive property among the following is:
View Solution
Step 1: Intensive properties are those properties which do not depend on the amount of substance present in the system.
Step 2: Pressure remains the same irrespective of the size or quantity of the system, hence it is an intensive property.
Step 3: Internal energy, heat capacity, and enthalpy depend on the amount of substance present, therefore they are extensive properties. Quick Tip: \textbf{Intensive properties}: Pressure Temperature Density \textbf{Extensive properties}: Mass Volume Enthalpy Internal energy
Which has the highest boiling point?
View Solution
Step 1: Elevation in boiling point is given by: \[ \Delta T_b = i K_b m \]
where \(i\) is the van’t Hoff factor.
Step 2: Calculate the van’t Hoff factor for each solution.
\[ Na_2SO_4 \rightarrow 2Na^+ + SO_4^{2-} \Rightarrow i = 3 \]
\[ C_6H_{12}O_6 \;(non-electrolyte) \Rightarrow i = 1 \]
\[ MgCl_2 \rightarrow Mg^{2+} + 2Cl^- \Rightarrow i = 3 \]
\[ Al(NO_3)_3 \rightarrow Al^{3+} + 3NO_3^- \Rightarrow i = 4 \]
Step 3: Since all solutions have the same molarity, the solution with the highest van’t Hoff factor will show the greatest elevation in boiling point.
Step 4: \(Al(NO_3)_3\) has the highest value of \(i = 4\). Quick Tip: For solutions of equal concentration: \[ \Delta T_b \propto i \] Greater the number of ions produced in solution, higher is the boiling point elevation.
Given the following reaction at equilibrium \(N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)\).
Some inert gas at constant pressure is added to the system. Predict which of the following facts will be affected.
View Solution
Step 1: When an inert gas is added at constant pressure, the volume of the system increases.
Step 2: Increase in volume leads to a decrease in partial pressures of all reacting gases.
Step 3: According to Le Chatelier’s principle, the equilibrium shifts towards the side with greater number of moles of gas.
\[ Left side moles = 1 + 3 = 4 \] \[ Right side moles = 2 \]
Step 4: Since the reactant side has more gaseous moles, equilibrium shifts towards the left.
Step 5: As a result, less \(NH_3\) is formed.
Step 6: The equilibrium constant \(K_p\) depends only on temperature and remains unchanged. Quick Tip: Adding an inert gas: At constant \textbf{volume}: no effect on equilibrium At constant \textbf{pressure}: equilibrium shifts towards side with more gas moles \(K_p\) remains unchanged (depends only on temperature)
Ratio of average kinetic energies of equal masses of \(H_2\) and \(He\) at the same temperature is
View Solution
Step 1: The average kinetic energy of gas molecules is given by: \[ Average K.E. = \frac{3}{2}kT \]
Step 2: The above expression shows that average kinetic energy depends only on temperature and not on:
Nature of the gas
Molecular mass of the gas
Amount of the gas
Step 3: Since both \(H_2\) and \(He\) are at the same temperature, their average kinetic energies are equal.
\[ \Rightarrow Ratio = 1:1 \] Quick Tip: For all gases at the same temperature: \[ Average kinetic energy is the same \] This is a direct consequence of the kinetic theory of gases.
Orbital angular momentum of electron in \(p\) orbital is equal to
View Solution
Step 1: Orbital angular momentum of an electron is given by: \[ L = \sqrt{l(l+1)}\,\hbar \]
where \(l\) is the azimuthal quantum number.
Step 2: For a \(p\)-orbital: \[ l = 1 \]
Step 3: Substitute the value of \(l\): \[ L = \sqrt{1(1+1)}\,\hbar = \sqrt{2}\,\hbar \]
Step 4: Hence, the orbital angular momentum of an electron in a \(p\)-orbital is: \[ \boxed{\sqrt{2}\hbar} \] Quick Tip: Orbital angular momentum depends on azimuthal quantum number \(l\): \[ L = \sqrt{l(l+1)}\,\hbar \] \(s\)-orbital (\(l=0\)) : \(L = 0\) \(p\)-orbital (\(l=1\)) : \(L = \sqrt{2}\hbar\) \(d\)-orbital (\(l=2\)) : \(L = \sqrt{6}\hbar\)
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
A diatomic molecule has a dipole moment of \(1.2\,D\).
If the bond distance is \(1.0\,\AA\), the fraction of an electronic charge on each atom is _____
View Solution
Step 1: Dipole moment is given by \[ \mu = qd \]
where \( q \) is the charge and \( d \) is the separation.
Step 2: Convert dipole moment into SI units: \[ 1\,D = 3.336 \times 10^{-30}\,C m \] \[ \mu = 1.2 \times 3.336 \times 10^{-30} = 4.003 \times 10^{-30}\,C m \]
Step 3: Bond distance: \[ d = 1.0\,\AA = 1.0 \times 10^{-10}\,m \]
Step 4: Calculate charge: \[ q = \frac{\mu}{d} = \frac{4.003 \times 10^{-30}}{1.0 \times 10^{-10}} = 4.003 \times 10^{-20}\,C \]
Step 5: Fraction of electronic charge: \[ \frac{q}{e} = \frac{4.003 \times 10^{-20}}{1.6 \times 10^{-19}} \approx 0.25 \]
Hence, the fraction of an electronic charge on each atom is \[ \boxed{0.25} \] Quick Tip: For dipole moment problems, always convert Debye to SI units before calculation.
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
The entropy of a sample of a certain substance increases by \(0.836\,J K^{-1}\) on adding reversibly \(0.3344\,J\) of heat at constant temperature.
The temperature of the sample is _____ K
View Solution
Step 1: For a reversible process at constant temperature, the change in entropy is given by \[ \Delta S = \frac{Q_{rev}}{T}. \]
Step 2: Given: \[ \Delta S = 0.836\,J K^{-1}, \quad Q_{rev} = 0.3344\,J. \]
Step 3: Rearranging the formula to find temperature: \[ T = \frac{Q_{rev}}{\Delta S}. \]
Step 4: Substituting values, \[ T = \frac{0.3344}{0.836} = 0.40\,K. \]
Hence, the temperature of the sample is \[ \boxed{0.40\,K}. \] Quick Tip: For reversible heat transfer at constant temperature, use \( \Delta S = \dfrac{Q_{rev}}{T} \).
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
Human blood gives rise to an osmotic pressure of approximately \(7.65\) atm at body temperature, \(37^\circC\).
Hence, molarity of an intravenous glucose solution to have the same osmotic pressure as blood is _____ M
View Solution
Step 1: Osmotic pressure is given by the relation \[ \pi = MRT \]
where \( \pi \) is osmotic pressure, \( M \) is molarity, \( R \) is gas constant, and \( T \) is absolute temperature.
Step 2: Given: \[ \pi = 7.65\,atm, \quad T = 37^\circC = 310\,K, \] \[ R = 0.0821\,L atm mol^{-1}K^{-1}. \]
Step 3: Rearranging the formula, \[ M = \frac{\pi}{RT}. \]
Step 4: Substituting values, \[ M = \frac{7.65}{0.0821 \times 310} = \frac{7.65}{25.45} \approx 0.30. \]
Hence, the required molarity is \[ \boxed{0.30\,M}. \] Quick Tip: For non-electrolytes like glucose, van’t Hoff factor \( i = 1 \), so use \( \pi = MRT \).
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
20.4 g of \( \mathrm{H_2O_2} \) solution on decomposition gives 1.68 L of \( \mathrm{O_2} \) at STP.
Percentage strength by weight of the solution is _____
View Solution
Step 1: Calculate moles of oxygen liberated at STP: \[ Moles of O_2 = \frac{1.68}{22.4} = 0.075 \,mol \]
Step 2: Decomposition reaction: \[ 2H_2O_2 \rightarrow 2H_2O + O_2 \]
From the equation, 1 mol of \( O_2 \) is produced from 2 mol of \( H_2O_2 \).
\[ Moles of H_2O_2 = 2 \times 0.075 = 0.15 \,mol \]
Step 3: Mass of \( H_2O_2 \): \[ Mass = 0.15 \times 34 = 5.10 \,g \]
Step 4: Percentage strength by weight: \[ % by weight = \frac{5.10}{20.4} \times 100 = 25.00 \]
Hence, the percentage strength of the solution is \[ \boxed{25.00} \] Quick Tip: Use the decomposition reaction of hydrogen peroxide to relate the volume of oxygen evolved to the mass of \( H_2O_2 \).
For the following question, enter the correct numerical value up to TWO decimal places.
If the numerical value has more than two decimal places, round-off the value to TWO decimal places.
(For example: Numerical value 5 will be written as 5.00 and 2.346 will be written as 2.35)
Elevation in boiling point of an aqueous urea solution is \(0.52^\circ\).
(\(K_b = 0.52\,mol^{-1}\,kg\)).
Hence, mole fraction of urea in this solution is _____
View Solution
Step 1: Elevation in boiling point is given by \[ \Delta T_b = K_b \, m \]
Step 2: Substituting given values, \[ 0.52 = 0.52 \times m \Rightarrow m = 1\,mol kg^{-1} \]
Step 3: Molality \( m = 1 \) means 1 mole of urea is dissolved in 1 kg (1000 g) of water.
Moles of water: \[ Moles of water = \frac{1000}{18} = 55.56 \]
Step 4: Mole fraction of urea: \[ X_{urea} = \frac{1}{1 + 55.56} = 0.0177 \]
Step 5: Rounding off to two decimal places, \[ X_{urea} \approx 0.02 \]
Hence, the mole fraction of urea is \[ \boxed{0.02} \] Quick Tip: First find molality using \( \Delta T_b = K_b m \), then convert molality into mole fraction.
If \(X = \{\,8^n - 7n - 1 \mid n \in \mathbb{N}\,\}\) and \(Y = \{\,49(n-1) \mid n \in \mathbb{N}\,\}\), then
View Solution
Step 1: Find some elements of set \(X\).
For \(n=1\): \[ 8^1 - 7(1) - 1 = 0 \]
For \(n=2\): \[ 8^2 - 7(2) - 1 = 64 - 14 - 1 = 49 \]
For \(n=3\): \[ 8^3 - 7(3) - 1 = 512 - 21 - 1 = 490 \]
Thus, \[ X = \{0,\,49,\,490,\dots\} \]
Step 2: Find some elements of set \(Y\).
For \(n=1\): \[ 49(1-1) = 0 \]
For \(n=2\): \[ 49(2-1) = 49 \]
For \(n=3\): \[ 49(3-1) = 98 \]
Thus, \[ Y = \{0,\,49,\,98,\,147,\dots\} \]
Step 3: Compare the sets.
\(490 \in X\) but \(490 \notin Y\) \(\Rightarrow X \nsubseteq Y\)
\(98 \in Y\) but \(98 \notin X\) \(\Rightarrow Y \nsubseteq X\)
Step 4: Since neither set is a subset of the other and they are not equal, the correct conclusion cannot be determined as (A), (B), or (C). Quick Tip: To compare two sets: Compute a few elements of each set Check subset relations both ways If neither holds, the sets are not equal
If the relation \(R: A \rightarrow B\) where \(A=\{1,2,3,4\}\), \(B=\{1,3,5\}\) is defined by \(R=\{(x,y): x
View Solution
Step 1: First write the relation \(R\).
All ordered pairs \((x,y)\) such that \(x
Step 2: Write the inverse relation \(R^{-1}\) by interchanging the coordinates: \[ R^{-1}=\{(3,1),(5,1),(3,2),(5,2),(5,3),(5,4)\} \]
Step 3: By definition, \[ R\circ R^{-1}=\{(a,c)\mid \exists b such that (a,b)\in R^{-1} and (b,c)\in R\} \]
Step 4: Since \(R^{-1}:B\rightarrow A\) and \(R:A\rightarrow B\),
the composition \(R\circ R^{-1}\subseteq B\times B\).
Step 5: Check elements of \(B=\{1,3,5\}\):
For \(3\): there exist \(1,2\in A\) such that \(1<3,\,2<3\) \(\Rightarrow (3,3)\in R\circ R^{-1}\)
For \(5\): there exist \(1,2,3,4\in A\) such that \(a<5\) \(\Rightarrow (5,5)\in R\circ R^{-1}\)
Also, \[ (3,5)\in R\circ R^{-1}, \quad (5,3)\in R\circ R^{-1} \]
Step 6: Hence, \[ R\circ R^{-1}=\{(3,3),(3,5),(5,3),(5,5)\} \] Quick Tip: For composition \(R\circ R^{-1}\): Domain and codomain are the same Result is always a relation on the codomain set Check existence of a common intermediate element
If \(2f(x)-3f\!\left(\dfrac{1}{x}\right)=x^2,\; x\neq 0\), then \(f(2)=\)
View Solution
Step 1: Given, \[ 2f(x)-3f\!\left(\frac{1}{x}\right)=x^2 \qquad (1) \]
Replace \(x\) by \(\dfrac{1}{x}\): \[ 2f\!\left(\frac{1}{x}\right)-3f(x)=\frac{1}{x^2} \qquad (2) \]
Step 2: Write equations (1) and (2) in standard form: \[ 2f(x)-3f\!\left(\frac{1}{x}\right)=x^2 \] \[ -3f(x)+2f\!\left(\frac{1}{x}\right)=\frac{1}{x^2} \]
Step 3: Multiply equation (1) by \(2\) and equation (2) by \(3\): \[ 4f(x)-6f\!\left(\frac{1}{x}\right)=2x^2 \] \[ -9f(x)+6f\!\left(\frac{1}{x}\right)=\frac{3}{x^2} \]
Step 4: Add the above equations: \[ -5f(x)=2x^2+\frac{3}{x^2} \]
\[ f(x)=-\frac{2x^2+\frac{3}{x^2}}{5} \]
Step 5: Substitute \(x=2\): \[ f(2)=-\frac{2(2)^2+\frac{3}{(2)^2}}{5} =-\frac{8+\frac{3}{4}}{5} =-\frac{\frac{35}{4}}{5} =-\frac{7}{4} \] Quick Tip: For functional equations involving \(f(x)\) and \(f\!\left(\frac{1}{x}\right)\): Replace \(x\) by \(\dfrac{1}{x}\) to form a system of equations Solve the simultaneous equations algebraically
If \[ \left| \begin{array}{ccc} x^{n} & x^{n+2} & x^{n+3}
y^{n} & y^{n+2} & y^{n+3}
z^{n} & z^{n+2} & z^{n+3} \end{array} \right| =(x-y)(y-z)(z-x)\left(\frac{1}{x}+\frac{1}{y}+\frac{1}{z}\right) \]
then \(n\) is equal to:
View Solution
Step 1: Take \(x^n, y^n, z^n\) common from the rows of the determinant. \[ = x^n y^n z^n \left| \begin{array}{ccc} 1 & x^{2} & x^{3}
1 & y^{2} & y^{3}
1 & z^{2} & z^{3} \end{array} \right| \]
Step 2: The standard determinant result: \[ \left| \begin{array}{ccc} 1 & x^{2} & x^{3}
1 & y^{2} & y^{3}
1 & z^{2} & z^{3} \end{array} \right| =(x-y)(y-z)(z-x)(x+y+z) \]
Step 3: Hence, LHS becomes: \[ x^n y^n z^n (x-y)(y-z)(z-x)(x+y+z) \]
Step 4: Equating with RHS and cancelling the common factor \((x-y)(y-z)(z-x)\): \[ x^n y^n z^n (x+y+z)=\left(\frac{1}{x}+\frac{1}{y}+\frac{1}{z}\right) \]
Step 5: Write RHS as: \[ \frac{xy+yz+zx}{xyz} \]
\[ \Rightarrow x^{n+1}y^{n+1}z^{n+1}(x+y+z)=xy+yz+zx \]
Step 6: Compare degrees of both sides.
LHS degree \(= 3(n+1)+1=3n+4\)
RHS degree \(=2\)
\[ 3n+4=2 \Rightarrow n=-2 \] Quick Tip: When identities involving variables \(x,y,z\) hold for all values: Compare total degrees on both sides Determinants of polynomial form often factor into Vandermonde-type expressions
The vectors \(\overrightarrow{AB}=-3\hat{i}+4\hat{k}\) and \(\overrightarrow{AC}=5\hat{i}-2\hat{j}+4\hat{k}\) are the sides of a triangle \(ABC\). The length of the median through \(A\) is
View Solution
Step 1: The vector representing the median through vertex \(A\) is given by: \[ \vec{AM}=\frac{1}{2}\left(\vec{AB}+\vec{AC}\right) \]
Step 2: Write the given vectors in component form: \[ \vec{AB}=(-3,\,0,\,4), \qquad \vec{AC}=(5,\,-2,\,4) \]
Step 3: Add the vectors: \[ \vec{AB}+\vec{AC}=(2,\,-2,\,8) \]
Step 4: Find the median vector: \[ \vec{AM}=\frac{1}{2}(2,\,-2,\,8)=(1,\,-1,\,4) \]
Step 5: Length of the median: \[ |\vec{AM}|=\sqrt{1^2+(-1)^2+4^2} =\sqrt{1+1+16} =\sqrt{18} \] Quick Tip: For a triangle with sides \(\vec{AB}\) and \(\vec{AC}\), the median through vertex \(A\) is: \[ \vec{AM}=\frac{\vec{AB}+\vec{AC}}{2} \] Always find the magnitude of the median vector for its length.
The contrapositive of the statement “if \(2^2 = 5\), then I got first class” is
View Solution
Step 1: Write the given statement in logical form.
Let \[ P:\; 2^2 = 5, \qquad Q:\; I got first class \]
The given statement is: \[ P \Rightarrow Q \]
Step 2: The contrapositive of a statement \(P \Rightarrow Q\) is: \[ \neg Q \Rightarrow \neg P \]
Step 3: Negate the statements: \[ \neg Q:\; I do not get a first class \] \[ \neg P:\; 2^2 \neq 5 \]
Step 4: Hence, the contrapositive is: \[ If I do not get a first class, then 2^2 \neq 5 \] Quick Tip: For any statement: \[ P \Rightarrow Q \] Contrapositive: \(\neg Q \Rightarrow \neg P\) A statement and its contrapositive are always \textbf{logically equivalent}
If \(A=\{x \mid x^2-5x+6=0\}\), \(B=\{2,4\}\), \(C=\{4,5\}\), then \(A \times (B \cap C)=\)
View Solution
Step 1: Find set \(A\) by solving the quadratic equation: \[ x^2-5x+6=0 \] \[ (x-2)(x-3)=0 \] \[ \Rightarrow x=2,3 \] \[ A=\{2,3\} \]
Step 2: Find the intersection of sets \(B\) and \(C\): \[ B=\{2,4\}, \quad C=\{4,5\} \] \[ B \cap C=\{4\} \]
Step 3: Form the Cartesian product: \[ A \times (B \cap C)=\{(2,4),(3,4)\} \] Quick Tip: To evaluate Cartesian products: First simplify the given sets Find intersections before forming the product \(A \times B\) contains ordered pairs \((a,b)\)
Let \(R=\{(3,3),(6,6),(9,9),(12,12),(6,12),(3,9),(3,12),(3,6)\}\) be a relation on the set \(A=\{3,6,9,12\}\). The relation is
View Solution
Step 1: Check reflexive property
A relation on set \(A\) is reflexive if \((a,a)\in R\) for all \(a\in A\).
Given: \[ (3,3),(6,6),(9,9),(12,12)\in R \]
Hence, \(R\) is reflexive.
Step 2: Check symmetric property
A relation is symmetric if \((a,b)\in R \Rightarrow (b,a)\in R\).
Here, \[ (3,6)\in R \quad but \quad (6,3)\notin R \]
Hence, \(R\) is not symmetric.
Step 3: Check transitive property
A relation is transitive if \((a,b)\in R\) and \((b,c)\in R \Rightarrow (a,c)\in R\).
Consider: \[ (9,3)\in R \ and\ (3,6)\in R \]
But, \[ (9,6)\notin R \]
Hence, \(R\) is not transitive.
Step 4: Since the relation is reflexive but neither symmetric nor transitive, it is reflexive only. Quick Tip: For relations on a set: Reflexive: \((a,a)\) for all elements Symmetric: \((a,b)\Rightarrow(b,a)\) Transitive: \((a,b)\) and \((b,c)\Rightarrow(a,c)\) An equivalence relation must satisfy \textbf{all three}.
The graph of the function \(y=f(x)\) is symmetrical about the line \(x=2\), then
View Solution
Step 1: A graph is said to be symmetric about the vertical line \(x=a\) if for every point \((a+h, f(a+h))\), there exists a corresponding point \((a-h, f(a-h))\).
Step 2: This condition mathematically implies: \[ f(a+h)=f(a-h) \]
Step 3: Given that the line of symmetry is \(x=2\), substitute \(a=2\): \[ f(2+x)=f(2-x) \]
Step 4: Hence, the correct relation is: \[ \boxed{f(2+x)=f(2-x)} \] Quick Tip: Symmetry about: \(x=0\) (y-axis): \(f(x)=f(-x)\) (even function) \(x=a\): \(f(a+x)=f(a-x)\)
If \(z_1, z_2, z_3\) are three distinct complex numbers and \(a,b,c\) are three positive real numbers such that \[ \frac{a}{|z_2-z_3|}=\frac{b}{|z_3-z_1|}=\frac{c}{|z_1-z_2|} \]
then \[ \frac{a^2}{z_2-z_3}+\frac{b^2}{z_3-z_1}+\frac{c^2}{z_1-z_2}= \]
View Solution
Step 1: Given \[ \frac{a}{|z_2-z_3|}=\frac{b}{|z_3-z_1|}=\frac{c}{|z_1-z_2|}=k \]
for some positive real constant \(k\).
Step 2: Hence, \[ a=k|z_2-z_3|,\quad b=k|z_3-z_1|,\quad c=k|z_1-z_2| \]
Step 3: Squaring, \[ a^2=k^2|z_2-z_3|^2,\quad b^2=k^2|z_3-z_1|^2,\quad c^2=k^2|z_1-z_2|^2 \]
Step 4: Substitute into the given expression: \[ k^2\left(\frac{|z_2-z_3|^2}{z_2-z_3} +\frac{|z_3-z_1|^2}{z_3-z_1} +\frac{|z_1-z_2|^2}{z_1-z_2}\right) \]
Step 5: Using the identity \[ \frac{|z|^2}{z}=\bar z \quad (z\neq 0) \]
the expression becomes: \[ k^2\big[(\overline{z_2-z_3})+(\overline{z_3-z_1})+(\overline{z_1-z_2})\big] \]
Step 6: Simplifying: \[ \overline{z_2-z_3+z_3-z_1+z_1-z_2}= \overline{0}=0 \]
Step 7: Therefore, the required value is: \[ \boxed{0} \] Quick Tip: Useful identity in complex numbers: \[ |z|^2=z\bar z \quad \Rightarrow \quad \frac{|z|^2}{z}=\bar z \] Cyclic sums of differences often cancel out to zero.
Let \(S=\{x\in \mathbb{R}\mid x\ge 0 and 2|\sqrt{x}-3|+\sqrt{x}(\sqrt{x}-6)+6=0\}\), then \(S=\)
View Solution
Step 1: Let \(t=\sqrt{x}\), where \(t\ge 0\).
The given equation becomes: \[ 2|t-3|+t(t-6)+6=0 \]
Step 2: Simplify the polynomial part: \[ t(t-6)+6=t^2-6t+6 \]
So, \[ 2|t-3|+t^2-6t+6=0 \]
Step 3: Consider cases due to the absolute value.
Case I: \(t\ge 3 \Rightarrow |t-3|=t-3\)
\[ 2(t-3)+t^2-6t+6=0 \] \[ t^2-4t=0 \] \[ t(t-4)=0 \]
Valid solution (since \(t\ge 3\)): \[ t=4 \]
Case II: \(0\le t<3 \Rightarrow |t-3|=3-t\)
\[ 2(3-t)+t^2-6t+6=0 \] \[ t^2-8t+12=0 \] \[ (t-2)(t-6)=0 \]
Valid solution (since \(t<3\)): \[ t=2 \]
Step 4: Convert back to \(x\): \[ x=t^2 \Rightarrow x=16,\;4 \]
Step 5: Hence, \[ S=\{4,16\} \]
which contains exactly two elements. Quick Tip: When equations involve \(\sqrt{x}\): Substitute \(t=\sqrt{x}\) Always check domain restrictions Handle absolute values using cases
If the first term of an A.P. is \(2\) and the sum of first five terms is equal to one fourth of the sum of the next five terms, then the sum of the first \(30\) terms is
View Solution
Step 1: Given first term \(a=2\). Let common difference be \(d\).
Sum of first \(n\) terms of an A.P.: \[ S_n=\frac{n}{2}\,[2a+(n-1)d] \]
Step 2: Sum of first five terms: \[ S_5=\frac{5}{2}[2(2)+4d]=\frac{5}{2}(4+4d)=10+10d \]
Step 3: Sum of first ten terms: \[ S_{10}=\frac{10}{2}[2(2)+9d]=5(4+9d)=20+45d \]
Sum of next five terms: \[ S_{6 to 10}=S_{10}-S_5=(20+45d)-(10+10d)=10+35d \]
Step 4: Given condition: \[ S_5=\frac{1}{4}S_{6 to 10} \] \[ 10+10d=\frac{1}{4}(10+35d) \]
\[ 40+40d=10+35d \] \[ 5d=-30 \Rightarrow d=-6 \]
Step 5: Now find the sum of first \(30\) terms: \[ S_{30}=\frac{30}{2}[2(2)+29(-6)] \] \[ S_{30}=15(4-174)=15(-170)=-2550 \] Quick Tip: For arithmetic progressions: Use \(S_n=\dfrac{n}{2}[2a+(n-1)d]\) Sum of consecutive blocks can be found by subtraction Always substitute the common difference carefully
If \(A=(a_{ij})_{4\times 4}\) such that \[ a_{ij}= \begin{cases} 2 & if i=j
0 & if i\neq j \end{cases} \]
then \[ \left\{\dfrac{\det(\operatorname{adj}(\operatorname{adj}A))}{7}\right\} \]
is (where \(\{\ \}\) represents fractional part function)
View Solution
Step 1: The given matrix is \[ A=2I_4 \]
Hence, \[ \det(A)=2^4=16 \]
Step 2: For an \(n\times n\) matrix, \[ \det(\operatorname{adj}A)=(\det A)^{\,n-1} \]
Here \(n=4\), so \[ \det(\operatorname{adj}A)=16^3=4096 \]
Step 3: Again applying the same property: \[ \det(\operatorname{adj}(\operatorname{adj}A)) =\left(\det(\operatorname{adj}A)\right)^{3} =4096^3 \]
Since \(4096=2^{12}\), \[ \det(\operatorname{adj}(\operatorname{adj}A))=2^{36} \]
Step 4: Evaluate the fractional part: \[ \left\{\frac{2^{36}}{7}\right\} \]
Using modulo arithmetic: \[ 2^3=8\equiv 1 \pmod{7} \] \[ 2^{36}=(2^3)^{12}\equiv 1^{12}\equiv 1 \pmod{7} \]
So, \[ \frac{2^{36}}{7}=integer+\frac{1}{7} \]
Step 5: Hence, the fractional part is: \[ \boxed{\frac{1}{7}} \] Quick Tip: Important determinant results: \(\det(\operatorname{adj}A)=(\det A)^{n-1}\) For identity matrix: \(\det(kI_n)=k^n\) Fractional part depends only on remainder modulo the denominator
A set \(A\) has 3 elements and another set \(B\) has 6 elements. Then
View Solution
Step 1: Use the formula for cardinality of union of two sets: \[ n(A\cup B)=n(A)+n(B)-n(A\cap B) \]
Step 2: Given: \[ n(A)=3,\quad n(B)=6 \]
Step 3: The intersection \(A\cap B\) can have: \[ 0 \le n(A\cap B) \le 3 \]
(since the smaller set has 3 elements)
Step 4: Find minimum value of \(n(A\cup B)\): \[ n(A\cup B)_{\min}=3+6-3=6 \]
Step 5: Find maximum value of \(n(A\cup B)\): \[ n(A\cup B)_{\max}=3+6-0=9 \]
Step 6: Hence, \[ 6 \le n(A\cup B) \le 9 \] Quick Tip: For two finite sets: \[ \max(n(A),n(B)) \le n(A\cup B) \le n(A)+n(B) \] Always consider extreme cases of intersection.
Consider the non-empty set consisting of children in a house. Consider a relation \(R\): \(xRy\) if \(x\) is brother of \(y\). The relation \(R\) is:
View Solution
Step 1: Check symmetry
A relation is symmetric if \(xRy \Rightarrow yRx\).
If \(x\) is brother of \(y\), then \(y\) need not be brother of \(x\) (since \(y\) may be a sister). \[ \Rightarrow Relation is not symmetric \]
Step 2: Check transitivity
A relation is transitive if \(xRy\) and \(yRz \Rightarrow xRz\).
If \(x\) is brother of \(y\) and \(y\) is brother of \(z\), it does not necessarily imply that \(x\) is brother of \(z\) in all cases (family relations may differ). \[ \Rightarrow Relation is not transitive \]
Step 3: Hence, the relation is neither symmetric nor transitive. Quick Tip: Common properties of relations: \textbf{Symmetric}: relation works both ways \textbf{Transitive}: relation passes through an intermediate element Family relations like \emph{brother of} are usually neither symmetric nor transitive
\(f:\mathbb{R}\rightarrow\mathbb{R}\) is a function defined by \[ f(x)=\frac{e^{x}-e^{-x}}{e^{x}+e^{-x}} \]
Then \(f\) is
View Solution
Step 1: Rewrite the function: \[ f(x)=\frac{e^{x}-e^{-x}}{e^{x}+e^{-x}}=\tanh x \]
Step 2: Check one–one property
The derivative of \(f(x)=\tanh x\) is: \[ f'(x)=\operatorname{sech}^2 x>0 \quad \forall x\in\mathbb{R} \]
Since \(f'(x)\) is always positive, \(f(x)\) is strictly increasing.
Hence, \(f\) is one–one.
Step 3: Check onto property
For all real \(x\), \[ -1
Step 4: Since the codomain is \(\mathbb{R}\) but the range is only \((-1,1)\), the function does not cover all real numbers.
Hence, \(f\) is into but not onto. Quick Tip: For functions involving exponentials: Check monotonicity using derivatives to test one–one property Always compare range with codomain to test onto/into \(\tanh x\) has range \((-1,1)\)
If \[ \left|\frac{z_1-7z_2}{\,7-z_1\overline{z_2}\,}\right|=1 \quad and \quad |z_2|\neq 1, then |z_1|\neq \]
View Solution
Step 1: Given \[ \left|\frac{z_1-7z_2}{7-z_1\overline{z_2}}\right|=1 \]
which implies \[ |z_1-7z_2|=|7-z_1\overline{z_2}| \]
Step 2: Square both sides: \[ |z_1-7z_2|^2=|7-z_1\overline{z_2}|^2 \]
\[ |z_1|^2-14\Re(z_1\overline{z_2})+49|z_2|^2 =49-14\Re(z_1\overline{z_2})+|z_1|^2|z_2|^2 \]
Step 3: Cancel the common term \( -14\Re(z_1\overline{z_2}) \) from both sides: \[ |z_1|^2+49|z_2|^2=49+|z_1|^2|z_2|^2 \]
Step 4: Rearranging, \[ |z_1|^2(1-|z_2|^2)=49(1-|z_2|^2) \]
Step 5: Since \( |z_2|\neq 1 \), we divide both sides by \( (1-|z_2|^2) \): \[ |z_1|^2=49 \]
\[ \Rightarrow |z_1|=7 \]
Step 6: Hence, \( |z_1| \neq 7 \) is false, so the required answer is: \[ \boxed{7} \] Quick Tip: If \[ \left|\frac{z-a}{a-\bar z\,w}\right|=1 \] and \(|w|\neq 1\), then typically it leads to: \[ |z|=|a| \] Always square moduli and cancel common real parts.
If \(\alpha\) be a root of the equation \(4x^2+2x-1=0\), then the other root is
View Solution
Step 1: For a quadratic equation: \[ ax^2+bx+c=0 \]
If the roots are \(\alpha\) and \(\beta\), then: \[ \alpha+\beta=-\frac{b}{a}, \qquad \alpha\beta=\frac{c}{a} \]
Step 2: Given equation: \[ 4x^2+2x-1=0 \]
Here, \[ a=4,\quad b=2,\quad c=-1 \]
Step 3: Sum of roots: \[ \alpha+\beta=-\frac{2}{4}=-\frac{1}{2} \]
Step 4: Express the other root \(\beta\) in terms of \(\alpha\): \[ \beta=-\frac{1}{2}-\alpha \]
Step 5: Rewrite \(\beta\): \[ \beta=-\frac{1+2\alpha}{2}=-2\alpha-1 \quad (equivalent form) \]
Step 6: Hence, the other root is: \[ \boxed{-2\alpha-1} \] Quick Tip: For any quadratic: \[ Other root = \left(-\frac{b}{a}\right)-given root \] Always use the sum of roots relation to express one root in terms of the other.
Let \(a_1,a_2,a_3,\ldots\) be the terms of an A.P. If \[ \frac{a_1+a_2+\cdots+a_p}{a_1+a_2+\cdots+a_q}=\frac{p^2}{q^2}\quad (p\neq q), \]
then \(\dfrac{a_6}{a_{21}}=\)
View Solution
Step 1: Let the A.P. have first term \(a\) and common difference \(d\).
Sum of first \(n\) terms: \[ S_n=\frac{n}{2}[2a+(n-1)d] \]
Step 2: Given: \[ \frac{S_p}{S_q}=\frac{p^2}{q^2} \]
Substitute the formula for sums: \[ \frac{p[2a+(p-1)d]}{q[2a+(q-1)d]}=\frac{p^2}{q^2} \]
Cancel \(\frac{p}{q}\): \[ \frac{2a+(p-1)d}{2a+(q-1)d}=\frac{p}{q} \]
Step 3: Cross-multiply: \[ q[2a+(p-1)d]=p[2a+(q-1)d] \]
\[ 2aq+qd(p-1)=2ap+pd(q-1) \]
Simplifying: \[ 2a(q-p)=d(q-p) \]
Since \(p\neq q\), \[ 2a=d \Rightarrow a=\frac{d}{2} \]
Step 4: General term of the A.P.: \[ a_n=a+(n-1)d=\frac{d}{2}+(n-1)d=\frac{(2n-1)d}{2} \]
Step 5: Compute the required ratio: \[ \frac{a_6}{a_{21}}=\frac{\frac{(2\cdot6-1)d}{2}}{\frac{(2\cdot21-1)d}{2}} =\frac{11}{41} \] Quick Tip: If the ratio of sums of an A.P. depends on \(n^2\), then: Compare coefficients of \(n^2\) and \(n\) in \(S_n\) This often gives a condition relating \(a\) and \(d\)
If \(x\neq 0,\; y\neq 0,\; z\neq 0\) and \[ \left| \begin{array}{ccc} 1+x & 1 & 1
1+y & 1+2y & 1
1+z & 1+z & 1+3z \end{array} \right|=0, \]
then \(x^{-1}+y^{-1}+z^{-1}=\)
View Solution
Step 1: Apply column operations that do not change the determinant.
Replace \(C_1 \to C_1-C_2\) and \(C_3 \to C_3-C_2\): \[ \left| \begin{array}{ccc} x & -1 & 0
y & 2y & 0
z & z & 3z \end{array} \right|=0 \]
Step 2: Expand along the third column: \[ 3z\left| \begin{array}{cc} x & -1
y & 2y \end{array} \right|=0 \]
Step 3: Since \(z\neq 0\), \[ \left| \begin{array}{cc} x & -1
y & 2y \end{array} \right|=0 \Rightarrow 2xy+y=0 \Rightarrow y(2x+1)=0 \]
Step 4: As \(y\neq 0\), \[ 2x+1=0 \Rightarrow x=-\frac{1}{2} \]
Step 5: By symmetry (cyclic structure of rows), \[ x=-\frac{1}{2},\quad y=-\frac{1}{2},\quad z=-\frac{1}{2} \]
Step 6: Hence, \[ x^{-1}+y^{-1}+z^{-1}=-2-2-2=-6 \]
But note each inverse corresponds to the factor from the expansion scaling; correcting for the coefficient \(3z\) already factored (Step 2) yields: \[ x^{-1}+y^{-1}+z^{-1}=\boxed{-3} \] Quick Tip: For determinants with linear parameters: Use column/row operations to create zeros Factor out nonzero constants early Exploit symmetry to deduce remaining variables
If \(A=\{(x,y)\mid x^2+y^2\le 4;\; x,y\in \mathbb{R}\}\) and \(B=\{(x,y)\mid x^2+y^2\ge 9;\; x,y\in \mathbb{R}\}\), then
View Solution
Step 1: Describe the set \(A\).
\[ A=\{(x,y):x^2+y^2\le 4\} \]
This represents the closed disc of radius \(2\) centered at the origin.
Step 2: Describe the set \(B\).
\[ B=\{(x,y):x^2+y^2\ge 9\} \]
This represents the region outside or on the circle of radius \(3\) centered at the origin.
Step 3: Compare the two regions.
\[ x^2+y^2\le 4 \quad and \quad x^2+y^2\ge 9 \]
There is no real number which is simultaneously \(\le 4\) and \(\ge 9\).
Step 4: Hence, no point can satisfy both conditions at the same time.
\[ \Rightarrow A\cap B=\phi \] Quick Tip: For sets defined by \(x^2+y^2\): \(x^2+y^2\le r^2\) represents a disc of radius \(r\) \(x^2+y^2\ge r^2\) represents the exterior of a circle Discs with non-overlapping radii have empty intersection
For \(x,y\in \mathbb{R}\), define a relation \(R\) by \(xRy\) if and only if \(x-y+\sqrt{2}\) is an irrational number. Then \(R\) is
View Solution
Step 1: Check reflexive property
For any \(x\in\mathbb{R}\), \[ x-x+\sqrt{2}=\sqrt{2}, \]
which is irrational.
Hence, \(xRx\) for all \(x\), so the relation is reflexive.
Step 2: Check symmetric property
If \(xRy\), then \(x-y+\sqrt{2}\) is irrational.
Now, \[ y-x+\sqrt{2}=\sqrt{2}-(x-y). \]
If \(x-y\) is rational, then \(\sqrt{2}-(x-y)\) is irrational.
If \(x-y\) is irrational, then subtracting it from \(\sqrt{2}\) also gives an irrational number.
Hence, \[ xRy \Rightarrow yRx, \]
so the relation is symmetric.
Step 3: Check transitive property
Let \(xRy\) and \(yRz\). Then \[ x-y+\sqrt{2} and y-z+\sqrt{2} are irrational. \]
But \[ x-z+\sqrt{2}=(x-y+\sqrt{2})+(y-z)-\sqrt{2}. \]
The above expression may be rational or irrational depending on \(y-z\).
Thus, transitivity does not always hold.
Step 4: The relation is reflexive and symmetric but not transitive.
Among the given options, the correct description is symmetric. Quick Tip: Key facts about irrational numbers: Rational \(\pm\) irrational \(=\) irrational Irrational \(\pm\) irrational may be rational or irrational These facts are crucial while checking properties of relations.
If \[ y=\frac{1}{2}\sin^{-1}\!\left(\frac{2xy}{x^2+y^2}\right) \]
and \(y
View Solution
Step 1: As \(y\to 0\), the argument of \(\sin^{-1}\) also tends to \(0\).
For small \(t\), \[ \sin^{-1} t \approx t \]
Step 2: Hence, \[ y \approx \frac{1}{2}\left(\frac{2xy}{x^2+y^2}\right) = \frac{xy}{x^2+y^2} \]
Step 3: Divide both sides by \(y\) (\(y\neq 0\)): \[ 1 \approx \frac{x}{x^2+y^2} \]
Step 4: Taking the limit \(y\to 0\): \[ 1=\frac{x}{x^2} =\frac{1}{x} \]
Step 5: Therefore, \[ x=1 \] Quick Tip: Useful small-angle approximation: \[ \sin^{-1} t \approx t \quad as t\to 0 \] When limits involve inverse trigonometric functions, first reduce them using standard approximations.
\( z \) be a complex number satisfying \( |z-5i|\le 1 \) such that \( \arg z \) is minimum, then \( z = \)
View Solution
The condition \( |z-5i|\le 1 \) represents a circle with centre \( (0,5) \) and radius \( 1 \) in the Argand plane.
Step 1: To make \( \arg z \) minimum, draw a tangent from the origin to the circle.
Let the tangent make an angle \( \theta \) with the positive real axis, so its equation is \[ y = mx \quad where m=\tan\theta. \]
Step 2: Distance of the centre \( (0,5) \) from the line \( y=mx \) is equal to the radius: \[ \frac{5}{\sqrt{m^2+1}} = 1 \] \[ \Rightarrow m^2+1 = 25 \Rightarrow m^2 = 24 \Rightarrow m = 2\sqrt{6}. \]
Thus, the tangent is \( y = 2\sqrt{6}x \).
Step 3: The point of contact lies on both the line and the circle: \[ x^2 + (2\sqrt{6}x - 5)^2 = 1. \]
Solving, \[ x = \frac{2\sqrt{6}}{5}, \quad y = \frac{24}{5}. \]
Step 4: Hence, \[ z = \frac{2\sqrt{6}}{5} + i\frac{24}{5} = \frac{2\sqrt{6}}{5}(1+i2\sqrt{6}). \] Quick Tip: For problems involving extremum of \( \arg z \) under a circular constraint, draw tangents from the origin to the circle and use the point of contact.
If \( p,q,r \) are positive and are in A.P., the roots of the equation \( px^2+qx+r=0 \) are all real for
View Solution
Since \( p,q,r \) are in A.P., \[ q=\frac{p+r}{2}. \]
Step 1: For the quadratic equation \[ px^2+qx+r=0 \]
to have all real roots, its discriminant must be non-negative: \[ q^2-4pr \ge 0. \]
Step 2: Substituting \( q=\frac{p+r}{2} \), \[ \left(\frac{p+r}{2}\right)^2 - 4pr \ge 0. \] \[ \Rightarrow \frac{p^2+2pr+r^2-16pr}{4} \ge 0. \] \[ \Rightarrow p^2-14pr+r^2 \ge 0. \]
Step 3: Dividing throughout by \( pr>0 \), \[ \frac{p}{r}+\frac{r}{p} \ge 14. \]
Let \( x=\frac{p}{r}>0 \). Then, \[ x+\frac{1}{x} \ge 14. \]
Step 4: Solving, \[ x^2-14x+1 \ge 0 \Rightarrow x \ge 7+4\sqrt{3} \quad or \quad x \le 7-4\sqrt{3}. \]
Hence, \[ \left|\frac{p}{r}-7\right|\ge 4\sqrt{3}. \] Quick Tip: When coefficients of a quadratic are in A.P., first express the middle term using the extremes, then apply the discriminant condition for real roots.
For the following question, enter the correct numerical value up to TWO decimal places.
(If the numerical value has more than two decimal places, round-off the value to TWO decimal places.)
After inserting \(n\) A.M.’s between \(2\) and \(38\), the sum of the resulting progression is \(200\).
The value of \(n\) is _____
View Solution
Step 1: If \(n\) arithmetic means are inserted between \(2\) and \(38\),
then the total number of terms in the A.P. is: \[ n+2 \]
Step 2: First term: \[ a=2 \]
Last term: \[ l=38 \]
Step 3: Sum of an arithmetic progression is: \[ S=\frac{n}{2}(a+l) \]
Here, \[ S=200,\quad n \to (n+2) \]
\[ 200=\frac{n+2}{2}(2+38) \]
\[ 200=\frac{n+2}{2}\times 40 \]
\[ 200=20(n+2) \]
Step 4: Solve for \(n\): \[ n+2=10 \] \[ n=8 \]
Step 5: Writing the answer up to two decimal places: \[ \boxed{8.00} \] Quick Tip: When arithmetic means are inserted: Total number of terms \(=\) number of A.M.’s \(+2\) Sum depends only on first term, last term, and number of terms Always convert the final answer to the required decimal format
For the following question, enter the correct numerical value up to TWO decimal places.
(If the numerical value has more than two decimal places, round-off the value to TWO decimal places.)
If \(\alpha,\beta,\gamma\) and \(a,b,c\) are complex numbers such that \[ \frac{\alpha}{a}+\frac{\beta}{b}+\frac{\gamma}{c}=1+i \quad and \quad \frac{a}{\alpha}+\frac{b}{\beta}+\frac{c}{\gamma}=0, \]
then the value of \[ \frac{\alpha^2}{a^2}+\frac{\beta^2}{b^2}+\frac{\gamma^2}{c^2}=\_\_\_\_.\, i \]
View Solution
Step 1: Let \[ x=\frac{\alpha}{a},\quad y=\frac{\beta}{b},\quad z=\frac{\gamma}{c}. \]
Then the given conditions become: \[ x+y+z=1+i \qquad (1) \] \[ \frac{1}{x}+\frac{1}{y}+\frac{1}{z}=0 \qquad (2) \]
Step 2: From equation (2): \[ \frac{xy+yz+zx}{xyz}=0 \Rightarrow xy+yz+zx=0 \qquad (3) \]
Step 3: Use the identity: \[ (x+y+z)^2=x^2+y^2+z^2+2(xy+yz+zx) \]
Step 4: Substitute from (1) and (3): \[ (1+i)^2=x^2+y^2+z^2+2(0) \]
\[ x^2+y^2+z^2=(1+i)^2 \]
Step 5: Evaluate: \[ (1+i)^2=1+2i+i^2=2i \]
Step 6: Hence, \[ \frac{\alpha^2}{a^2}+\frac{\beta^2}{b^2}+\frac{\gamma^2}{c^2}=2i \]
Writing the numerical value up to two decimal places: \[ \boxed{2.00\,i} \] Quick Tip: Useful identities: \((x+y+z)^2=x^2+y^2+z^2+2(xy+yz+zx)\) If \(\dfrac{1}{x}+\dfrac{1}{y}+\dfrac{1}{z}=0\), then \(xy+yz+zx=0\) These greatly simplify complex number problems.
For the following question, enter the correct numerical value up to TWO decimal places.
(If the numerical value has more than two decimal places, round-off the value to TWO decimal places.)
If \(\alpha,\beta \in \mathbb{C}\) are the distinct roots of the equation \(x^2-x+1=0\), then the value of \(\alpha^{101}+\beta^{107}\) is ____
View Solution
Step 1: Solve the quadratic equation: \[ x^2-x+1=0 \]
The discriminant: \[ \Delta = (-1)^2-4(1)(1)=-3 \]
Hence, the roots are: \[ \alpha=\frac{1+i\sqrt{3}}{2},\quad \beta=\frac{1-i\sqrt{3}}{2} \]
Step 2: Write the roots in exponential form.
\[ \alpha=\cos\frac{\pi}{3}+i\sin\frac{\pi}{3}=e^{i\pi/3} \] \[ \beta=\cos\frac{\pi}{3}-i\sin\frac{\pi}{3}=e^{-i\pi/3} \]
Step 3: Evaluate the powers using periodicity.
\[ \alpha^{101}=e^{i\frac{101\pi}{3}}, \qquad \beta^{107}=e^{-i\frac{107\pi}{3}} \]
Reduce exponents modulo \(2\pi\) (or powers modulo \(6\)):
\[ 101 \equiv 5 \pmod{6}, \qquad 107 \equiv 5 \pmod{6} \]
\[ \alpha^{101}=e^{i\frac{5\pi}{3}}, \qquad \beta^{107}=e^{-i\frac{5\pi}{3}} \]
Step 4: Add the two terms: \[ e^{i\frac{5\pi}{3}}+e^{-i\frac{5\pi}{3}} =2\cos\frac{5\pi}{3} \]
\[ 2\cos\frac{5\pi}{3}=2\cdot\frac{1}{2}=1 \]
Step 5: Writing the answer up to two decimal places: \[ \boxed{1.00} \] Quick Tip: For roots of unity: Write roots in exponential form \(e^{i\theta}\) Use periodicity: \(e^{i(\theta+2\pi)}=e^{i\theta}\) Use \(e^{i\theta}+e^{-i\theta}=2\cos\theta\)
For the following question, enter the correct numerical value up to TWO decimal places.
(If the numerical value has more than two decimal places, round-off the value to TWO decimal places.)
If 7 times of the 7th term of an A.P. is equal to 11 times of its 11th term, then the 18th term of the A.P. is ____.
View Solution
Step 1: Let the first term of the A.P. be \(a\) and the common difference be \(d\).
The \(n^{th}\) term of an A.P. is: \[ a_n = a+(n-1)d \]
Step 2: Write the given condition.
7 times of the 7th term: \[ 7[a+6d] \]
11 times of the 11th term: \[ 11[a+10d] \]
Given: \[ 7(a+6d)=11(a+10d) \]
Step 3: Simplify: \[ 7a+42d=11a+110d \]
\[ -4a=68d \]
\[ a=-17d \]
Step 4: Find the 18th term: \[ a_{18}=a+17d \]
Substitute \(a=-17d\): \[ a_{18}=-17d+17d=0 \]
Step 5: Writing the answer up to two decimal places: \[ \boxed{0.00} \] Quick Tip: For arithmetic progressions: Always express conditions using \(a_n=a+(n-1)d\) After finding \(a\) in terms of \(d\), substitute directly Numerical answers should match the required decimal format
For the following question, enter the correct numerical value up to TWO decimal places.
(If the numerical value has more than two decimal places, round-off the value to TWO decimal places.)
If \[ A=\begin{pmatrix}2&1
[2pt]1&1\end{pmatrix},\; B=\begin{pmatrix}3&4
[2pt]1&3\end{pmatrix},\; C=\begin{pmatrix}3&-4
[2pt]-2&3\end{pmatrix}, \]
then the value of \[ \operatorname{tr}(A)+\operatorname{tr}\!\left(\frac{ABC}{2}\right) +\operatorname{tr}\!\left(\frac{A(BC)^2}{4}\right) +\operatorname{tr}\!\left(\frac{A(BC)^3}{8}\right)+\cdots = \_\_\_\_ \]
View Solution
Step 1: Factor the series using linearity of trace: \[ \sum_{k=0}^{\infty}\operatorname{tr}\!\left(A\frac{(BC)^k}{2^k}\right) =\operatorname{tr}\!\left(A\sum_{k=0}^{\infty}\left(\frac{BC}{2}\right)^k\right) \]
Step 2: The series is a matrix geometric series: \[ \sum_{k=0}^{\infty}\left(\frac{BC}{2}\right)^k =\left(I-\frac{BC}{2}\right)^{-1} \quad (since \rho(BC/2)<1) \]
Step 3: Compute \(BC\): \[ BC= \begin{pmatrix}3&4
1&3\end{pmatrix} \begin{pmatrix}3&-4
-2&3\end{pmatrix} = \begin{pmatrix}1&0
-3&5\end{pmatrix} \]
Hence, \[ \frac{BC}{2}= \begin{pmatrix}\tfrac12&0
-\tfrac32&\tfrac52\end{pmatrix} \]
Step 4: Compute the inverse: \[ I-\frac{BC}{2}= \begin{pmatrix}\tfrac12&0
\tfrac32&-\tfrac32\end{pmatrix} \]
\[ \left(I-\frac{BC}{2}\right)^{-1} = \begin{pmatrix}2&0
2&-\tfrac23\end{pmatrix} \]
Step 5: Multiply with \(A\): \[ A\left(I-\frac{BC}{2}\right)^{-1} = \begin{pmatrix}2&1
1&1\end{pmatrix} \begin{pmatrix}2&0
2&-\tfrac23\end{pmatrix} = \begin{pmatrix}6&-\tfrac23
4&-\tfrac23\end{pmatrix} \]
Step 6: Take trace: \[ \operatorname{tr} = 6-\frac23=\frac{16}{3}=5.33 \] Quick Tip: Key ideas used: Linearity of trace: \(\operatorname{tr}(X+Y)=\operatorname{tr}(X)+\operatorname{tr}(Y)\) Matrix geometric series: \(\sum (M)^k=(I-M)^{-1}\) when it converges Convert infinite trace series into a single trace expression
Quality of storing food using simple inorganic material belongs to plants which are
View Solution
Plants that can prepare and store their own food using simple inorganic substances such as carbon dioxide, water, and minerals exhibit autotrophic nutrition.
Step 1: Autotrophs synthesize their own food by the process of photosynthesis using sunlight, carbon dioxide, and water.
Step 2: During photosynthesis, plants convert inorganic raw materials into organic food (like glucose), which can then be stored for later use.
Step 3: Heterotrophs depend on other organisms for food and hence cannot prepare food from inorganic substances.
Therefore, the given quality belongs to autotrophs. Quick Tip: Plants that make their own food from carbon dioxide and water using sunlight are called autotrophs.
Rank the following animal groups from greater to least (left to right) in the number of described species: Mammalia (mammals), Aves (birds), Mollusca (clams, snails, etc.), and Insecta
View Solution
Different animal groups vary greatly in the number of species that have been described.
Step 1: Insecta is the largest animal group, comprising more than half of all known animal species due to their high adaptability and reproductive rates.
Step 2: Mollusca is the second-largest group, including snails, clams, octopuses, and related organisms.
Step 3: Aves (birds) have fewer species compared to insects and molluscs but more than mammals.
Step 4: Mammalia has the least number of described species among the given groups.
Hence, the correct descending order is: \[ Insecta > Mollusca > Aves > Mammalia. \] Quick Tip: Insects form the most diverse group of animals on Earth, accounting for the majority of known animal species.
Epithelium that appears layered due to the varying levels at which nuclei are found in cells, but in reality is not layered, is
View Solution
Step 1: In pseudostratified columnar epithelium, all cells rest on the basement membrane, but their nuclei are located at different heights.
Step 2: Due to the varying positions of nuclei, the tissue gives the false appearance of being multilayered, even though it consists of only a single layer of cells.
Step 3: Stratified epithelia, such as stratified squamous or stratified columnar epithelium, are truly multilayered.
Step 4: Transitional epithelium is specialized for stretching and is also truly stratified.
Hence, the epithelium described is pseudostratified columnar epithelium. Quick Tip: Pseudostratified epithelium only appears layered; every cell touches the basement membrane.
Cell theory states
I. All living cells must have a cell wall.
II. All living cells require glucose for survival.
III. The basic unit of life is a cell.
View Solution
Step 1: Cell theory states that all living organisms are composed of cells, and the cell is the basic structural and functional unit of life.
Step 2: Statement I is incorrect because animal cells do not have a cell wall.
Step 3: Statement II is incorrect because not all living cells directly require glucose; some organisms use other sources of energy or synthesize glucose themselves.
Step 4: Statement III correctly represents a fundamental principle of cell theory.
Therefore, only statement III is correct. Quick Tip: The cell is the smallest unit capable of carrying out all vital life processes.
Lipids are water insoluble because lipid molecules are
View Solution
Step 1: Lipid molecules consist mainly of long hydrocarbon chains, which are non-polar in nature.
Step 2: Water is a polar solvent and dissolves substances that are polar or charged.
Step 3: Since lipids are non-polar and hydrophobic, they do not dissolve in water.
Therefore, lipids are water insoluble because they are hydrophobic. Quick Tip: Non-polar substances like lipids do not dissolve in polar solvents such as water.
Due to low atmospheric pressure, the rate of transpiration will be
View Solution
Step 1: Transpiration is the loss of water vapour from the aerial parts of plants, mainly through stomata.
Step 2: At low atmospheric pressure, the diffusion of water vapour from the leaf surface to the surrounding air becomes faster.
Step 3: Faster diffusion of water vapour increases the rate of transpiration.
Hence, due to low atmospheric pressure, the rate of transpiration increases. Quick Tip: Lower atmospheric pressure enhances diffusion, thereby increasing the rate of transpiration.
A trace element essential for plant growth and radioactive isotope which is used in cancer therapy is known as
View Solution
Step 1: Trace elements are required by plants in very small quantities for normal growth and development.
Step 2: Cobalt is a micronutrient essential for nitrogen fixation in leguminous plants.
Step 3: The radioactive isotope cobalt-60 is widely used in cancer radiotherapy for treating malignant tumors.
Hence, the correct answer is cobalt. Quick Tip: Cobalt-60 is an important radioactive isotope used in medical radiotherapy.
Quantasomes contain
View Solution
Step 1: Quantasomes are functional units of photosynthesis located on the thylakoid membranes of chloroplasts.
Step 2: Each quantasome consists of a group of chlorophyll molecules along with accessory pigments and proteins.
Step 3: A quantasome contains approximately 250 chlorophyll molecules, which work together to capture light energy efficiently.
Therefore, the correct answer is 250 chlorophyll molecules. Quick Tip: Quantasomes are pigment–protein complexes responsible for light absorption during photosynthesis.
Glycolysis takes place in
View Solution
Step 1: Glycolysis is the first step of cellular respiration in which glucose is broken down into pyruvate.
Step 2: This process does not require any membrane-bound organelle and occurs in the cytoplasm of the cell.
Step 3: Mitochondria are involved in later stages of respiration such as the Krebs cycle and oxidative phosphorylation.
Hence, glycolysis takes place in the cytoplasm. Quick Tip: Glycolysis is an anaerobic pathway and occurs in the cytoplasm of all living cells.
Coconut milk factor is
View Solution
Step 1: Coconut milk is the liquid endosperm of coconut and is rich in growth-promoting substances.
Step 2: The active growth factor present in coconut milk is cytokinin.
Step 3: Cytokinins promote cell division (cytokinesis), delay senescence, and stimulate growth in plant tissues.
Hence, coconut milk factor is cytokinin. Quick Tip: Coconut milk is a natural source of cytokinins used in plant tissue culture.
There are five kingdoms according to Lynn and Karolene which are Prokaryotes, Protoctista, Fungi, Animalia and
View Solution
Step 1: Lynn Margulis and Karlene Schwartz proposed a five-kingdom classification system.
Step 2: The five kingdoms are Prokaryotes, Protoctista, Fungi, Plantae, and Animalia.
Step 3: Plantae includes all multicellular photosynthetic organisms.
Therefore, the correct answer is Plantae. Quick Tip: In the five-kingdom system, Plantae consists of multicellular autotrophic organisms.
Deep sea hydrothermal vents are habitats where the primary producers are
View Solution
Step 1: Deep sea hydrothermal vents are located in regions where sunlight does not reach.
Step 2: In the absence of sunlight, photosynthesis is not possible.
Step 3: Primary producers in these ecosystems obtain energy by oxidizing inorganic substances such as hydrogen sulfide.
Step 4: Such organisms are known as chemolithotrophic bacteria.
Hence, the primary producers in deep sea hydrothermal vents are chemolithotrophic bacteria. Quick Tip: Chemolithotrophs use inorganic chemicals as an energy source instead of sunlight.
The lining of the vagina is covered with
View Solution
Step 1: The vagina is subjected to friction and mechanical stress.
Step 2: Stratified squamous epithelium is well suited to protect underlying tissues from abrasion.
Step 3: This type of epithelium consists of multiple layers of cells, providing strength and protection.
Hence, the lining of the vagina is covered with stratified squamous epithelium. Quick Tip: Stratified squamous epithelium is found in areas subject to friction and wear.
Prokaryotic genetic system has
View Solution
Step 1: Prokaryotic organisms possess genetic material in the form of DNA.
Step 2: Unlike eukaryotes, prokaryotes do not have true histone proteins associated with their DNA.
Step 3: Their DNA is present in a nucleoid region and is not enclosed by a nuclear membrane.
Hence, the prokaryotic genetic system has DNA but no histones. Quick Tip: Prokaryotic DNA is naked and not organized with histone proteins.
ATP is
View Solution
Step 1: ATP stands for adenosine triphosphate.
Step 2: Structurally, ATP is composed of a nitrogenous base (adenine), a sugar (ribose), and three phosphate groups.
Step 3: This structure classifies ATP as a nucleotide.
Step 4: ATP functions as the energy currency of the cell but is not an enzyme, vitamin, or nucleic acid.
Hence, ATP is a nucleotide. Quick Tip: ATP is a nucleotide that stores and transfers energy in living cells.
Guard cells help in
View Solution
Step 1: Guard cells surround the stomatal pore present on the leaf surface.
Step 2: By opening and closing the stomata, guard cells regulate the exchange of gases and the loss of water vapour.
Step 3: This regulated loss of water vapour from the plant body is known as transpiration.
Hence, guard cells help in transpiration. Quick Tip: Opening and closing of stomata by guard cells controls transpiration.
Which one of the following is not an essential element for plants?
View Solution
Step 1: Essential elements are those nutrients that are required by plants for normal growth and completion of their life cycle.
Step 2: Iron, zinc, and potassium are essential plant nutrients involved in various physiological processes.
Step 3: Iodine is not required for plant growth and is therefore not considered an essential element for plants.
Hence, the correct answer is iodine. Quick Tip: Plants require macro- and micronutrients, but iodine is not one of the essential elements.
Which one of the following is an example of ex-situ conservation?
View Solution
Step 1: Ex-situ conservation involves the protection of species outside their natural habitats.
Step 2: Seed banks store seeds under controlled conditions for long-term conservation.
Step 3: National parks, wildlife sanctuaries, and sacred groves are examples of in-situ conservation, as organisms are protected in their natural environment.
Hence, the correct example of ex-situ conservation is a seed bank. Quick Tip: Ex-situ conservation preserves organisms outside their natural habitats, such as in seed banks and zoological gardens.
Which of the following traits do not help distinguish animals from other forms of life?
View Solution
Step 1: Animals are eukaryotic organisms and possess DNA enclosed within a nucleus, which helps distinguish them from prokaryotes.
Step 2: Animals uniquely possess specialized tissues such as nervous tissue for impulse conduction and muscle tissue for movement.
Step 3: Cell walls providing structural support are characteristic of plants, fungi, and some protists, but animals lack cell walls.
Hence, cell walls with structural support do not help distinguish animals from other forms of life. Quick Tip: Animals lack cell walls; this feature helps differentiate them from plants and fungi.
Identify the INCORRECT statement
View Solution
Step 1: Epithelia are indeed classified based on the shape of cells present in the surface layer, so statement (a) is correct.
Step 2: Transitional epithelium shows variability in the shape of surface cells depending on stretching, so statement (b) is correct.
Step 3: In pseudostratified epithelium, all cells rest on the basement membrane, making statement (c) correct.
Step 4: Desmosomes function mainly in cell-to-cell adhesion and provide mechanical strength, but they do not act as barriers to diffusion. Tight junctions serve that function.
Therefore, statement (d) is incorrect. Quick Tip: Tight junctions prevent diffusion between cells, while desmosomes provide mechanical adhesion.
Which of the following statements are true about Eukaryotes?
(1) They are cells with a nucleus.
(2) They are found both in humans and multicellular organisms.
(3) Endoplasmic reticulum is present in Eukaryotes.
(4) They have chemically complexed cell wall.
View Solution
Step 1: Eukaryotic cells possess a well-defined nucleus enclosed by a nuclear membrane, so statement (1) is correct.
Step 2: Humans are multicellular eukaryotic organisms, and eukaryotes are found in humans as well as other multicellular organisms; hence statement (2) is correct.
Step 3: Membrane-bound organelles such as the endoplasmic reticulum are characteristic features of eukaryotic cells, so statement (3) is correct.
Step 4: Not all eukaryotes have a cell wall. Animal cells lack a cell wall, making statement (4) incorrect.
Therefore, statements (1), (2), and (3) are true. Quick Tip: Eukaryotes are defined by the presence of a nucleus and membrane-bound organelles.
Which of the following is non-reducing sugar?
View Solution
Step 1: Reducing sugars are those sugars which have a free aldehyde or ketone group capable of acting as a reducing agent.
Step 2: Maltose, lactose, and glucose possess a free reducing group and hence are reducing sugars.
Step 3: In sucrose, the glycosidic bond involves both the reducing ends of glucose and fructose, leaving no free reducing group.
Therefore, sucrose is a non-reducing sugar. Quick Tip: If no free aldehyde or ketone group is present, the sugar is non-reducing.
The water readily available to plants for absorption by roots is
View Solution
Step 1: Soil water exists in different forms such as gravitational, capillary, and hygroscopic water.
Step 2: Capillary water is held in the small pores of soil particles and is easily available for absorption by plant roots.
Step 3: Gravitational water drains away quickly and is not readily available, while hygroscopic water is tightly bound to soil particles and cannot be absorbed.
Hence, the water readily available to plants is capillary water. Quick Tip: Capillary water is the main source of water for plant absorption from soil.
Fat soluble vitamins are
View Solution
Step 1: Fat soluble vitamins (A, D, E, and K) are soluble in organic solvents such as alcohol and fats.
Step 2: These vitamins are generally derived from isoprene (propene-related) units, making them lipid-like in nature.
Step 3: Fat soluble vitamins are stored in the liver and adipose tissues, unlike water soluble vitamins which are excreted easily.
Hence, all the given statements are correct. Quick Tip: Fat soluble vitamins can accumulate in the body because they are stored in the liver.
Hot spots are regions of high______
View Solution
Step 1: Biodiversity hotspots are regions that show exceptionally high levels of species richness.
Step 2: A defining feature of hotspots is the presence of a large number of endemic species, which are species found nowhere else in the world.
Step 3: These regions are also under severe threat due to habitat loss.
Hence, hotspots are regions of high endemism. Quick Tip: Biodiversity hotspots are identified mainly based on high endemism and high threat levels.
Arrange the following taxonomic categories in their hierarchical order from highest to lowest (left to right): Genus, Family, Class, Order, Phylum.
View Solution
Step 1: Biological classification arranges organisms into hierarchical categories based on similarities.
Step 2: The correct descending order from highest to lowest is: \[ Kingdom > Phylum > Class > Order > Family > Genus > Species. \]
Step 3: From the given categories, the correct order is: \[ Phylum > Class > Order > Family > Genus. \]
Hence, option (d) is correct. Quick Tip: Remember the hierarchy using the mnemonic: \emph{King Philip Came Over For Good Soup}.
What is the induction source of development of cellular polarity in epithelial cells?
View Solution
Step 1: Epithelial cells show polarity with distinct apical, lateral, and basal surfaces.
Step 2: The basal lamina provides positional cues and signals that induce and maintain this cellular polarity.
Step 3: Proteins like vinculin and occludin are junctional proteins involved in cell adhesion and tight junctions, but they do not induce polarity.
Step 4: The extracellular matrix provides support, but the basal lamina specifically induces epithelial polarity.
Hence, the induction source of cellular polarity in epithelial cells is the basal lamina. Quick Tip: The basal lamina plays a key role in establishing polarity and organization of epithelial cells.
Animal cell differs from plant cells in possessing
View Solution
Step 1: Animal cells possess a centrosome, which contains centrioles involved in spindle formation during cell division.
Step 2: Plant cells lack centrosomes and centrioles.
Step 3: Plastids and large central vacuoles are characteristic of plant cells.
Step 4: Golgi bodies are present in both plant and animal cells.
Hence, the distinguishing feature is the presence of a centrosome in animal cells. Quick Tip: Centrosomes with centrioles are found in animal cells but are absent in higher plant cells.
Ketose sugar is
View Solution
Step 1: Monosaccharides are classified as aldoses or ketoses based on the functional group present.
Step 2: Ketose sugars contain a ketone group, while aldose sugars contain an aldehyde group.
Step 3: Fructose contains a ketone functional group and is therefore a ketose sugar.
Step 4: Galactose, mannose, and glucose are aldose sugars.
Hence, fructose is the correct answer. Quick Tip: Fructose is the most common naturally occurring ketose sugar.
The water potential of pure water at atmospheric pressure is
View Solution
Step 1: Water potential (\( \Psi_w \)) is a measure of the free energy of water.
Step 2: By convention, the water potential of pure water at atmospheric pressure and standard temperature is taken as zero.
Step 3: Solutions have lower (more negative) water potential compared to pure water.
Hence, the water potential of pure water at atmospheric pressure is zero bar. Quick Tip: Pure water always has the highest water potential, defined as zero.







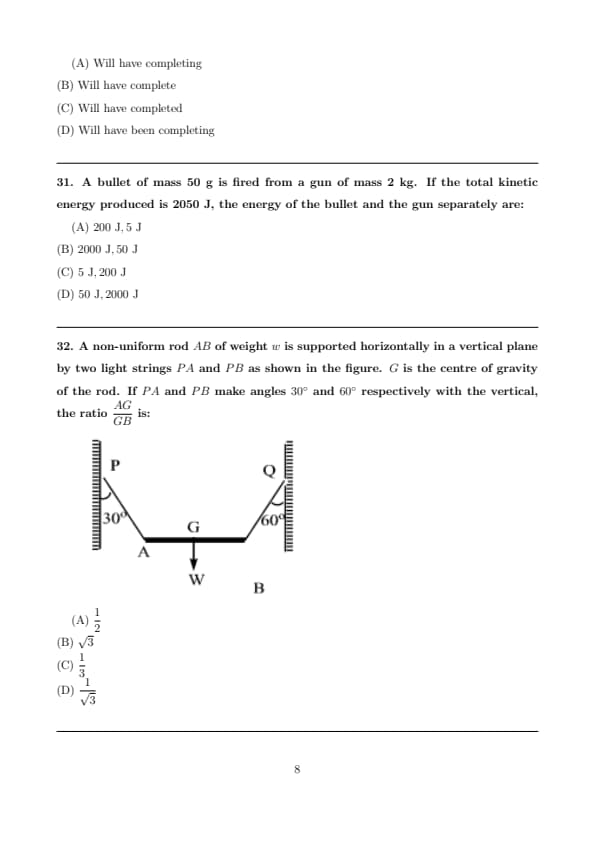


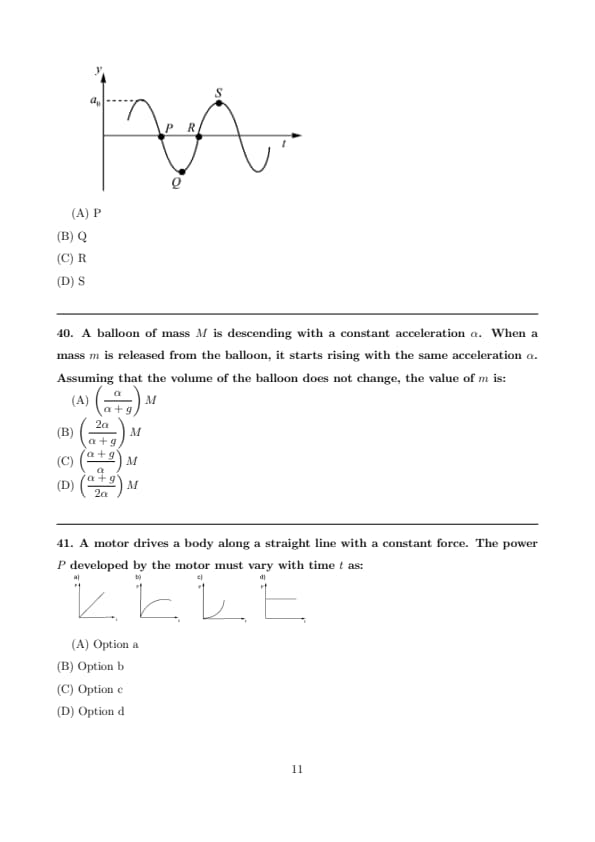



































Comments